| Cadogan–Sundberg indole synthesis | |
|---|---|
| Named after | John Cadogan Richard J. Sundberg |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000509 |
The Cadogan–Sundberg indole synthesis, or simply Cadogan indole synthesis, is a name reaction in organic chemistry that allows for the generation of indoles from o-nitrostyrenes with the use of trialkyl phosphites, such as triethyl phosphite.
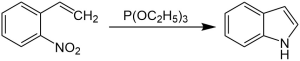
Mechanism
o-nitrostyrene first reacts with triethyl phosphite, and the nitro group is converted to a nitroso group. The nitroso group then reacts with the alkene, and N-hydroxylindole is formed, which reacts again with triethyl phosphite to form the indole.

Application
The Cadogan–Sundberg indole synthesis has been used as an intermediate step in the total synthesis of Tjipanazole E, transforming 2--5-chloro-1H-indole to 5,5’-Dichloro-2,2’-biindole.
References
- ^ Cadogan, John Ivan George; Mackie, R. K. (1974). "Tervalent phosphorus compounds in organic synthesis". Chemical Society Reviews. 3 (1): 87–137. doi:10.1039/CS9740300087.
- ^ Wang, Zerong (15 September 2010). Comprehensive Organic Name Reactions and Reagents. doi:10.1002/9780470638859.conrr128. ISBN 9780470638859.
- ^ Sundberg, Richard J.; Yamazaki, Toshio (1 February 1967). "Rearrangements and ring expansions during the deoxygenation of β,β-disubstituted o-nitrostyrenes". The Journal of Organic Chemistry. 32 (2): 290–294. doi:10.1021/jo01288a009.
- ^ Sundberg, Richard J. (1 November 1965). "Deoxygenation of Nitro Groups by Trivalent Phosphorus. Indoles from o-Nitrostyrenes". The Journal of Organic Chemistry. 30 (11): 3604–3610. doi:10.1021/jo01022a006.
- Gribble, Gordon W. (17 June 2016). "Cadogan–Sundberg Indole Synthesis". Indole Ring Synthesis: From Natural Products to Drug Discovery. pp. 266–277. doi:10.1002/9781118695692.ch26. ISBN 9781118695692.
- Majgier-Baranowska, Helena; Williams, John D.; Li, Bing; Peet, Norton P. (1 February 1967). "Studies on the mechanism of the Cadogan–Sundberg indole synthesis". Tetrahedron Letters. 53 (35): 4785–4788. doi:10.1016/j.tetlet.2012.06.146.
- Li, Jie Jack (4 January 2014). "Cadogan–Sundberg indole synthesis". Indole Ring Synthesis: From Natural Products to Drug Discovery. Springer. p. 102-103. doi:10.1007/978-3-319-03979-4_49. ISBN 978-3-319-03979-4.
- Kuethe, Jeffrey T.; Wong, Audrey; Davies, Iaan W. (3 September 2003). "Effective Strategy for the Preparation of Indolocarbazole Aglycons and Glycosides: Total Synthesis of Tjipanazoles B, D, E, and I". Organic Letters. 5 (20): 3721–3723. doi:10.1021/ol035418r. PMID 14507214.
This chemical reaction article is a stub. You can help Misplaced Pages by expanding it. |