 | |
| Names | |
|---|---|
| Preferred IUPAC name Formyl cyanide | |
| Systematic IUPAC name Methanoyl cyanide | |
| Other names
Cyanoformaldehyde Glyoxylonitrile 2-oxo-acetonitrile oxo-acetonitrile | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| PubChem CID | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C2HNO |
| Molar mass | 55.036 g·mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa). Infobox references | |
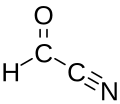
Formyl cyanide is a simple organic compound with the formula HCOCN and structure HC(=O)−C≡N. It is simultaneously a nitrile (R−C≡N) and an aldehyde (R−CH=O). Formyl cyanide is the simplest member of the acyl cyanide family. It is known to occur in space in the Sgr B2 molecular cloud.
Production
Formyl cyanide was first made through methoxyacetonitrile flash vacuum pyrolysis at 600 °C. The same technique with cinnamyloxyacetonitrile or allyloxyacetonitrile also generates formyl cyanide.
In molecular clouds, formation of formyl cyanide is speculated to result from formaldehyde and the cyanide radical:
- CH2O + CN• → HCOCN + H•
In Earth's atmosphere, the pollutant acrylonitrile reacts with hydroxyl radical forming formyl cyanide, hydroperoxyl and formaldehyde:
- CH2=CHCN + HO• + 3⁄2 O2 → HOO• + HCOCN + CH2O
Reactions
Formyl cyanide reacts rapidly with even trace quantities of water to form formic acid and hydrogen cyanide. In scrupulously dry conditions, the compound instead releases carbon monoxide, with a half-life exceeding 45 h.
Related
By formally substituting the hydrogen atom, cyanoformyl chloride, ClC(O)CN, and cyanoformyl bromide, BrC(O)CN are obtained.
References
- Gronowski, Marcin; Eluszkiewicz, Piotr; Custer, Thomas Gage (12 April 2017). "Structure and Spectroscopy of C2HNO Isomers". The Journal of Physical Chemistry A. 121 (17): 3263–3273. Bibcode:2017JPCA..121.3263G. doi:10.1021/acs.jpca.6b12609. PMID 28402122.
- ^ Lewis-Bevan, Wyn; Gaston, Rick D.; Tyrrell, James; Stork, Wilmer D.; Salmon, Gary L. (March 1992). "Formyl cyanide: a stable species. Experimental and theoretical studies". Journal of the American Chemical Society. 114 (6): 1933–1938. doi:10.1021/ja00032a001.
- Bogey, M.; Destombes, J.L.; Vallee, Y.; Ripoll, J.L. (May 1988). "Formyl cyanide: Efficient production from allyloxyacetonitrile and its millimeter-wave spectrum". Chemical Physics Letters. 146 (3–4): 227–229. Bibcode:1988CPL...146..227B. doi:10.1016/0009-2614(88)87435-9.
- Bogey, M.; Demuynck, C.; Destombes, J.L.; Vallee, Y. (August 1995). "Millimeter-Wave Spectrum of Formyl Cyanide, HCOCN: Centrifugal Distortion and Hyperfine Structure Analysis". Journal of Molecular Spectroscopy. 172 (2): 344–351. Bibcode:1995JMoSp.172..344B. doi:10.1006/jmsp.1995.1183.
- Remijan, Anthony J.; Hollis, J. M.; Lovas, F. J.; Stork, Wilmer D.; Jewell, P. R.; Meier, D. S. (10 March 2008). "Detection of Interstellar Cyanoformaldehyde (CNCHO)". The Astrophysical Journal. 675 (2): L85 – L88. Bibcode:2008ApJ...675L..85R. doi:10.1086/533529. S2CID 19005362.
- Grosjean, Daniel (December 1990). "Atmospheric Chemistry of Toxic Contaminants. 3. Unsaturated Aliphatics: Acrolein, Acrylonitrile, Maleic Anhydride". Journal of the Air & Waste Management Association. 40 (12): 1664–1669. Bibcode:1990JAWMA..40.1664G. doi:10.1080/10473289.1990.10466814.
- Pasinszki, Tibor; Vass, Gábor; Klapstein, Dieter; Westwood, Nicholas P. C. (5 April 2012). "Generation, Spectroscopy, and Structure of Cyanoformyl Chloride and Cyanoformyl Bromide, XC(O)CN". The Journal of Physical Chemistry A. 116 (13): 3396–3403. Bibcode:2012JPCA..116.3396P. doi:10.1021/jp301528q. PMID 22409314.