| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name N-Ethyl-N-hydroxyethanamine | |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| Beilstein Reference | 1731349 | ||
| ChemSpider | |||
| ECHA InfoCard | 100.020.960 | ||
| EC Number |
| ||
| MeSH | N,N-diethylhydroxylamine | ||
| PubChem CID | |||
| RTECS number |
| ||
| UNII | |||
| UN number | 1993 | ||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
| Chemical formula | C4H11NO | ||
| Molar mass | 89.138 g·mol | ||
| Appearance | Colorless liquid | ||
| Odor | Ammoniacal | ||
| Density | 867 mg mL | ||
| Melting point | −26 to −25 °C (−15 to −13 °F; 247 to 248 K) | ||
| Boiling point | 127.6 °C; 261.6 °F; 400.7 K | ||
| Solubility in water | Miscible | ||
| Vapor pressure | 500 Pa (at 0 °C) | ||
| Acidity (pKa) | 5.67 (est) | ||
| Thermochemistry | |||
| Heat capacity (C) | 370.8 J K mol | ||
| Std enthalpy of formation (ΔfH298) |
−175.47–−174.03 kJ mol | ||
| Std enthalpy of combustion (ΔcH298) |
−2.97201–−2.97069 MJ mol | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms |  
| ||
| Signal word | Warning | ||
| Hazard statements | H226, H312, H315, H319, H332 | ||
| Precautionary statements | P280, P305+P351+P338 | ||
| Explosive limits | 1.9–10% | ||
| Lethal dose or concentration (LD, LC): | |||
| LD50 (median dose) |
| ||
| Related compounds | |||
| Related alkanols | |||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
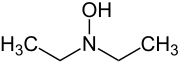
Diethylhydroxylamine (DEHA) is an organic compound with the formula (C2H5)2NOH. Strictly, this is N,N-diethylhydroxylamine. It has an isomer, N,O-diethylhydroxylamine with the formula EtNHOEt. Pure N,N-diethylhydroxylamine is a colorless liquid, although it is usually encountered as a colourless-to-yellow solution in water with an amine-like odor.
DEHA can be synthesised from a reaction between triethylamine and a peroxide.
Applications
DEHA is largely used as an oxygen scavenger in water treatment.
It is a volatile oxygen scavenger and reacts in a ratio of 2.8/1 DEHA/O2. It is employed in high pressure (>70 bar) boiler systems due to a very low rate of reaction at low temperatures and pressures. Due to its volatility, it acts as an oxygen scavenger throughout the entire boiler system due to steam carryover.
DEHA also reacts with ferrous metals to form a passivized film of magnetite throughout the boiler system. The reduction of toxic heavy metals, such as hexavalent chromium to their more environmentally-friendly counterparts like trivalent chromium, is also performed using aqueous solutions containing DEHA.
Several other applications include its use as:
- Polymerisation inhibitor
- Color stabilizer (photographics)
- Corrosion inhibitor
- Discoloration inhibitor (phenolics)
- Antiozonant
- Radical scavenger
References
- Hilal SH et al; pp. 291-353 in Quantitative Treatments of Solute/Solvent Interactions: Theoretical and Computational Chemistry Vol. 1 NY, NY: Elsevier (1994). SPARC (Software Process Automation Reaction Chemistry) Available from, as of Dec 7, 2007: http://ibmlc2.chem.uga.edu/sparc/
- Cáceres, T.; Lissi, E. A.; Sanhueza, E. (November 1978). "Autooxidation of diethyl hydroxylamine". International Journal of Chemical Kinetics. 10 (11): 1167–1182. doi:10.1002/kin.550101107.
- Shaffer, Dean; Heicklen, Julian (August 1986). "Oxidation of diethylhydroxylamine in water solution at 25-80.degree". The Journal of Physical Chemistry. 90 (18): 4408–4413. doi:10.1021/j100409a039.
- Abuin, E.; Encina, M. V.; Diaz, S.; Lissi, E. A. (July 1978). "On the reactivity of diethyl hydroxyl amine toward free radicals". International Journal of Chemical Kinetics. 10 (7): 677–686. doi:10.1002/kin.550100704.