| Flavonol 3-O-glucosyltransferase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 Flavonol 3-O-glucosyltransferase Flavonol 3-O-glucosyltransferase | |||||||||
| Identifiers | |||||||||
| EC no. | 2.4.1.91 | ||||||||
| CAS no. | 50812-18-5 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
In enzymology, a flavonol 3-O-glucosyltransferase (EC 2.4.1.91) is an enzyme that catalyzes the chemical reaction
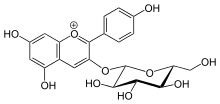
- UDP-glucose + a flavonol UDP + a flavonol 3-O-beta-D-glucoside
Thus, the two substrates of this enzyme are UDP-glucose and flavonol, whereas its two products are UDP and flavonol 3-O-beta-D-glucoside. The flavonoids that can act as substrates within this reaction include quercetin, kaempferol, dihydrokaempferol, kaempferid, fisetin, and isorhamnetin. Flavonol 3-O-glucosyltransferase is a hexosyl group transfer enzyme.
This enzyme is known by the systematic name UPD-glucose:flavonol 3-O-D glucosyltransferase, and it participates in flavonoid biosynthesis and causes the formation of anthocyanins. Anthocyanins produce a purple color in the plant tissues that they are present in.
It is an enzyme found most notably in grapes (Vitis vinifera). This enzyme is found within a number of other plants as well—such as snapdragons (Antirrhinum majus), kale (Brassica oleracea), and grapefruit (Citrus x paradisi).
Pathways
This enzyme is involved in the biosynthesis of secondary metabolites. The primary function of this enzyme within its pathway is binding a glucoside onto a flavonol molecule, forming a flavonol 3-O-glucoside. It is through this mechanism that the enzyme converts anthocyanidins to anthocyanins as a part of the phenylpropanoid pathway. One specific example would be this enzymes actions on pelargonidin. Flavonol 3-O-glucosyltransferase binds the glucoside to this protein, making pelargonidin 3-O-glucoside. This enzyme is also involved in the flavone glycoside pathway, and daphnetin modification in some organisms. The role of the enzyme in these pathways is, again, to bind a glucoside to the substrate to construct a flavonol 3-O-glucoside.

Nomenclature
This enzyme belongs to the family of glycosyltransferases, specifically the hexosyltransferases. The systematic name of this enzyme class is UDP-glucose:flavonol 3-O-D-glucosyltransferase. Other names in common use include:
- GTI,
- uridine diphosphoglucose-flavonol 3-O-glucosyltransferase,
- UDP-glucose:flavonol 3-O-glucosyltransferase, and
- UDP-glucose:flavonoid 3-O-glucosyltransferase (UFGT).
Among those, UFGT is divided into UDP-glucose: Flavonoid 3-O-glucosyltransferase (UF3GT) and UDP-glucose: Flavonoid 5-O-glucosyltransferase (UF5GT), which are responsible for the glucosylation of anthocyanins to produce stable molecules.
Inhibitors and Structure of the Enzyme
Some of the inhibitors of this enzyme include CaCl2, CoCl2, Cu, CuCl2, KCl, Mg, and Mn. The primary active site residue of this enzyme is Asp181, as determined by studies of how mutations affect enzyme capacity. There are several documentations of the crystalline structure of flavonol 3-O-glucosyltransferase (2C1X, 2C1Z, and 2C9Z), and, based on these renderings of the enzyme, there is only one subunit in the quaternary structure of the molecule.
References
- http://brenda-enzymes.info/enzyme.php?ecno=2.4.1.91&Suchword=&reference=&organism%5B%5D=Vitis+vinifera&show_tm=0#REACTION TYPE
- Dooner, H.K. & Nelson, O.E. Biochem Genet (1977) 15: 509. doi:10.1007/BF00520194
- Kobayashi S, Ishimaru M, Ding CK, Yakushiji H, Goto N (February 2001). "Comparison of UDP-glucose:flavonoid 3-O-glucosyltransferase (UFGT) gene sequences between white grapes (Vitis vinifera) and their sports with red skin". Plant Sci. 160 (3): 543–550. Bibcode:2001PlnSc.160..543K. doi:10.1016/S0168-9452(00)00425-8. PMID 11166442.
- "BRENDA - Information on EC 2.4.1.91 - flavonol 3-O-glucosyltransferase".
- http://brenda-enzymes.info/enzyme.php?ecno=2.4.1.91&Suchword=&reference=&organism%5B%5D=Vitis+vinifera&show_tm=0#SOURCE TISSUE
- "KEGG ENZYME: 2.4.1.115". www.genome.jp. Retrieved 2016-12-06.
- Zhao DQ, Han CX, Ge JT, Tao J (November 2012). "Isolation of a UDP-glucose: Flavonoid 5-O-glucosyltransferase gene and expression analysis of anthocyanin biosynthetic genes in herbaceous peony (Paeonia lactiflora Pall.)". Electronic Journal of Biotechnology. 15 (6). doi:10.2225/vol15-issue6-fulltext-7.
- Schomburg, Professor Dietmar; Schomburg, Dr Ida, eds. (1 January 2006). "Flavonol 3-O-glucosyltransferase". Springer Handbook of Enzymes. Vol. 32. Springer Berlin Heidelberg. pp. 21–29. doi:10.1007/978-3-540-49534-5_2. ISBN 978-3-540-32591-8.
- Hiromoto, Takeshi; Honjo, Eijiro; Noda, Naonobu; Tamada, Taro; Kazuma, Kohei; Suzuki, Masahiko; Blaber, Michael; Kuroki, Ryota (2016-12-06). "Structural basis for acceptor-substrate recognition of UDP-glucose: anthocyanidin 3-O-glucosyltransferase from Clitoria ternatea". Protein Science. 24 (3): 395–407. doi:10.1002/pro.2630. ISSN 0961-8368. PMC 4353365. PMID 25556637.
- Offen, W; Martinez-Fleites, C; Yang, M; Kiat-Lim, E; Davis, B.G; Tarling, C.A; Ford, C.M; Bowles, D.J; Davies, G.J. (2006-01-01). "Structure of a Flavonoid Glucosyltransferase Reveals the Basis for Plant Natural Product Modification". EMBO J. 25 (6): 1396–405. doi:10.1038/sj.emboj.7600970. PMC 1422153. PMID 16482224.
Further reading
- Wiermann R (1982). "Formation of flavonol 3-O-diglycosides and flavonol 3-O-triglycosides by enzyme extracts from anthers of Tulipa cv apeldoorn - characterization and activity of 3 different O-glycosyltransferases during anther development". Z. Naturforsch. C. 37: 587–599. doi:10.1515/znc-1982-7-808. S2CID 37241803.
- Sutter A, Grisebach H (1973). "UDP-glucose: flavonol 3-0-glucosyltransferase from cell suspension cultures of parsley". Biochim. Biophys. Acta. 309 (2): 289–95. doi:10.1016/0005-2744(73)90027-2. PMID 4731963.
| Enzymes | |
|---|---|
| Activity | |
| Regulation | |
| Classification | |
| Kinetics | |
| Types |
|
 UDP + a flavonol 3-O-beta-D-glucoside
UDP + a flavonol 3-O-beta-D-glucoside