| Revision as of 10:55, 10 February 2011 editBeetstra (talk | contribs)Edit filter managers, Administrators172,031 edits Script assisted update of identifiers from ChemSpider, CommonChemistry and FDA for the Chem/Drugbox validation project - Updated: ChEMBL.← Previous edit | Latest revision as of 22:34, 5 August 2024 edit undoحسن علي البط (talk | contribs)Extended confirmed users, Pending changes reviewers19,940 edits added Category:Diols using HotCat | ||
| (60 intermediate revisions by 42 users not shown) | |||
| Line 1: | Line 1: | ||
| {{chembox | {{chembox | ||
| | verifiedrevid = 413087241 | |||
| | |
| Name = 1,1{{prime}}-Bi-2-naphthol | ||
| ⚫ | | |
||
| | ImageFile = | |||
| | ImageSizeL1 = 100px | |||
| | |
| ImageFileL1 = R-BINOL-2D-skeletal.png | ||
| | ImageNameL1 = Skeletal formula of R-BINOL | |||
| ⚫ | | |
||
| ⚫ | | ImageFileR1 = S-BINOL-2D-skeletal.png | ||
| | ImageSizeR1 = 100px | |||
| | |
| ImageNameR1 = Skeletal formula of S-BINOL | ||
| | |
| ImageFileL2 = R-BINOL-3D-balls.png | ||
| | ImageNameL2 = Ball-and-stick model of R-BINOL | |||
| | ImageSize2 = 150px | |||
| ⚫ | | ImageFileR2 = S-BINOL-3D-balls.png | ||
| ⚫ | | |
||
| | ImageNameR2 = Ball-and-stick model of S-BINOL | |||
| | OtherNames = 1,1'-Bi-2,2'-naphthol<br />1,1-Binaphthol<br />BINOL | |||
| | ImageCaptionL2 = (''R'')-(+)-BINOL | |||
| | ImageCaptionR2 = (''S'')-(−)-BINOL | |||
| ⚫ | | PIN = -2,2{{prime}}-diol | ||
| | OtherNames = {{Unbulleted list | |||
| ⚫ | | 1,1{{prime}}-Bi-2-naphthol | ||
| | 1,1-Binaphthol | |||
| | BINOL | |||
| | Binol | |||
| ⚫ | }} | ||
| | SystematicName = | |||
| | Section1 = {{Chembox Identifiers | | Section1 = {{Chembox Identifiers | ||
| | ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} | |||
| | |
| ChemSpiderID = 11269 | ||
| | PubChem = 11762 | | PubChem = 11762 | ||
| | InChI = 1/C20H14O2/c21-17-11-9-13-5-1-3-7-15(13)19(17)20-16-8-4-2-6-14(16)10-12-18(20)22/h1-12,21-22H | | InChI = 1/C20H14O2/c21-17-11-9-13-5-1-3-7-15(13)19(17)20-16-8-4-2-6-14(16)10-12-18(20)22/h1-12,21-22H | ||
| | InChIKey = PPTXVXKCQZKFBN-UHFFFAOYAX | | InChIKey = PPTXVXKCQZKFBN-UHFFFAOYAX | ||
| | SMILES = C1=CC=C2C(=C1)C=CC(=C2C3=C(C=CC4=CC=CC=C43)O)O | |||
| | SMILES1 = c1ccc2c(c1)ccc(c2c3c4ccccc4ccc3O)O | |||
| | SMILES_Comment = (''R''/''S'') | |||
| | ChEMBL_Ref = {{ebicite|correct|EBI}} | |||
| | ChEMBL = 138718 | | ChEMBL = 138718 | ||
| | StdInChI_Ref = {{stdinchicite|correct|chemspider}} | |||
| | StdInChI = 1S/C20H14O2/c21-17-11-9-13-5-1-3-7-15(13)19(17)20-16-8-4-2-6-14(16)10-12-18(20)22/h1-12,21-22H | | StdInChI = 1S/C20H14O2/c21-17-11-9-13-5-1-3-7-15(13)19(17)20-16-8-4-2-6-14(16)10-12-18(20)22/h1-12,21-22H | ||
| | StdInChIKey_Ref = {{stdinchicite|correct|chemspider}} | |||
| | StdInChIKey = PPTXVXKCQZKFBN-UHFFFAOYSA-N | | StdInChIKey = PPTXVXKCQZKFBN-UHFFFAOYSA-N | ||
| | CASNo = 602-09-5 | | CASNo = 602-09-5 | ||
| | CASNo_Comment = (R/S) | | CASNo_Comment = (''R''/''S'') | ||
| | CASNo1 = 18531-94-7 | | CASNo1 = 18531-94-7 | ||
| | CASNo1_Comment = ( |
| CASNo1_Comment = (''R'')-(+) | ||
| | CASNo2 = 18531-99-2 | | CASNo2 = 18531-99-2 | ||
| | CASNo2_Comment = (S)-( |
| CASNo2_Comment = (''S'')-(−) | ||
| | UNII_Ref = {{fdacite|correct|FDA}} | |||
| | SMILES = Oc4ccc1c(cccc1)c4c2c3c(ccc2O)cccc3 | |||
| | UNII = 25AB254328 | |||
| | UNII_Comment = (''R''/''S'') | |||
| | UNII1_Ref = {{fdacite|correct|FDA}} | |||
| | UNII1 = M6IDZ128WT | |||
| | UNII1_Comment = (''R'')-(+) | |||
| | UNII2_Ref = {{fdacite|correct|FDA}} | |||
| | UNII2 = 54OT5RRV4C | |||
| | UNII2_Comment = (''S'')-(−) | |||
| }} | }} | ||
| | Section2 = {{Chembox Properties | | Section2 = {{Chembox Properties | ||
| | Formula = | |||
| | Formula = C<sub>20</sub>H<sub>14</sub>O<sub>2</sub> | |||
| | |
| C=20 | H=14 | O=2 | ||
| | |
| MolarMass = 286.32 g/mol | ||
| | Density = | |||
| ⚫ | | |
||
| | MeltingPtC = 205 to 211 | |||
| ⚫ | | MeltingPt_ref = <ref>, chemexper.com</ref> | ||
| }} | }} | ||
| | Section3 = | |||
| | Section4 = | |||
| | Section5 = | |||
| | Section6 = | |||
| }} | }} | ||
| '''1,1 |
'''1,1{{prime}}-Bi-2-naphthol''' ('''BINOL''') is an ] that is often used as a ] for transition-metal catalysed ]. BINOL has ] and the two ]s can be readily separated and are stable toward ]. The ] of the two ] is 35.5° (''c'' = 1 in ]), with the ''R'' enantiomer being the ] one. BINOL is a precursor for another ] called ]. The volumetric mass ] of the two enantiomers is 0.62 g cm{{sup|−3}}.{{citation needed|date=January 2022}} | ||
| ==Preparation== | ==Preparation== | ||
| The ] of BINOL is not a challenge as such but the preparation of the individual ]s is. | The ] of BINOL is not a challenge as such but the preparation of the individual ]s is. | ||
| (''S'')-BINOL can be prepared directly from an asymmetric ] of ] with ]. The ] in this reaction is (''S'')-(+)-].<ref> | (''S'')-BINOL can be prepared directly from an asymmetric ] of ] with ]. The ] in this reaction is (''S'')-(+)-].<ref>{{cite journal|last1 = Brussee|first1 = J.|last2 = Jansen|first2 = A. C. A.|year = 1983|title = A highly stereoselective synthesis of ''S''-(−)--2,2{{prime}}-diol|journal = Tetrahedron Letters|volume = 24|issue = 31|pages = 3261–3262|doi = 10.1016/S0040-4039(00)88151-4}}</ref> | ||
| {{cite journal | |||
| | last = Brussee | |||
| | first = J. | |||
| | coauthors = Jansen A. C. A. | |||
| | year = 1983 | |||
| | title = A highly stereoselective synthesis of s(-)--2,2′-diol | |||
| | journal = Tetrahedron Letters | |||
| | volume = 24 | |||
| | pages = 3261–3262 | |||
| | doi = 10.1016/S0040-4039(00)88151-4 | |||
| ⚫ | }} | ||
| </ref> | |||
| ] | ] | ||
| Racemic BINOL can also be produced using ] as an oxidant. The mechanism involves complexation of iron(III) into the ], followed by a radical coupling reaction of the naphthol rings initiated by iron(III) reducing into iron(II). | Racemic BINOL can also be produced using ] as an oxidant. The mechanism involves complexation of iron(III) into the ], followed by a radical coupling reaction of the naphthol rings initiated by iron(III) reducing into iron(II). | ||
| ] BINOL can also be obtained from ] BINOL by optical resolution. In one method, the ] N-benzylcinchonidinium chloride |
] BINOL can also be obtained from ] BINOL by optical resolution. In one method, the ] N-benzylcinchonidinium chloride forms a crystalline ]. The inclusion compound of the (''S'')-enantiomer is soluble in ] but that of the (''R'')-enantiomer is not.<ref name="orgsyn"> {{Webarchive|url=https://web.archive.org/web/20120716191712/http://www.orgsyn.org/orgsyn/prep.asp?prep=v76p0001 |date=2012-07-16 }}, Dongwei Cai, David L. Hughes, Thomas R. Verhoeven, and Paul J. Reider, in ] Coll. Vol. 10, p.93; Vol. 76, p.1</ref> In another method BINOL is esterified with ]. The enzyme ] ] ]s the (''S'')-diester but not the (''R'')-diester.<ref name="orgsyn" /> The (''R'')-dipentanoate is hydrolysed in a second step with ].<ref name="orgsyn2"> {{Webarchive|url=https://web.archive.org/web/20050418112746/http://www.orgsyn.org/orgsyn/prep.asp?prep=CV9P0077 |date=2005-04-18 }}, Romas J. Kazlauskas in ], Coll. Vol. 9, p.77; Vol. 70, p.60</ref> The third method employs ] with chiral stationary phases.<ref>{{cite journal|last1 = Landek|first1 = G.|last2 = Vinković|first2 = M.|last3 = Kontrec|first3 = D.|last4 = Vinković|first4 = V.|year = 2006| title = Influence of mobile phase and temperature on separation of 1,1{{prime}}-binaphthyl-2,2{{prime}}-diol enantiomers with brush type chiral stationary phases derived from L-leucine|journal = Chromatographia|volume = 64|issue = 7–8|pages = 469–473|doi = 10.1365/s10337-006-0041-5| s2cid=95785346 }}</ref> | ||
| ⚫ | ==BINOL derivatives== | ||
| In another method BINOL is reacted with the ] pentanoyl chloride to obtain the di-]. The enzyme ] ] is then added in the form of ] ] acetone powder which is able to ] the (''S'')-di-ester but not the (''R'')-di-ester.<ref name="orgsyn" /> The (''R'')-dipentanoate is hydrolysed in a second step with ].<ref name="orgsyn2">, Romas J. Kazlauskas in ], Coll. Vol. 9, p.77; Vol. 70, p.60</ref> | |||
| ] derived from BINOL.<ref>{{cite journal |doi=10.1021/cr5001496|title=Complete Field Guide to Asymmetric BINOL-Phosphate Derived Brønsted Acid and Metal Catalysis: History and Classification by Mode of Activation; Brønsted Acidity, Hydrogen Bonding, Ion Pairing, and Metal Phosphates|year=2014|last1=Parmar|first1=Dixit|last2=Sugiono|first2=Erli|last3=Raja|first3=Sadiya|last4=Rueping|first4=Magnus|journal=Chemical Reviews|volume=114|issue=18|pages=9047–9153|pmid=25203602|doi-access=free}}</ref>]] | |||
| Aside from the starting materials derived directly from the ], (''R'')- and (''S'')-BINOL in high enantiopurity (>99% ]) are two of the most inexpensive sources of chirality for organic synthesis, costing less than US$0.60 per gram when purchased in bulk from chemical suppliers.<ref>{{Cite journal|last1=Yang|first1=Jin-Fei|last2=Wang|first2=Rong-Hua|last3=Wang|first3=Yin-Xia|last4=Yao|first4=Wei-Wei|last5=Liu|first5=Qi-Sheng|last6=Ye|first6=Mengchun|date=2016-10-11|title=Ligand-Accelerated Direct C−H Arylation of BINOL: A Rapid One-Step Synthesis of Racemic 3,3{{prime}}-Diaryl BINOLs|journal=Angewandte Chemie International Edition|language=en|volume=55|issue=45|pages=14116–14120|doi=10.1002/anie.201607893|pmid=27726256 |issn=1433-7851}}</ref> As a consequence, it serves as an important starting material for other sources of chirality for stereoselective synthesis, both stoichiometric and substoichiometric (catalytic). | |||
| Many important chiral ligands are constructed from the binaphthyl scaffold and ultimately derived from BINOL as a starting material, ] being one of the most well known and important. | |||
| Third method employs ] with chiral stationary phases.<ref> | |||
| {{cite journal | |||
| | last = Landek | |||
| | first = G. | |||
| | coauthors = Vinković M., Kontrec D. and Vinković V. | |||
| | year = 2006 | |||
| | title = Influence of mobile phase and temperature on separation of 1,1 '-binaphthyl-2,2 '-diol enantiomers with brush type chiral stationary phases derived from L-leucine | |||
| | journal = Chromatographia | |||
| | volume = 64 | |||
| | pages = 469–473 | |||
| | doi = 10.1365/s10337-006-0041-5 | |||
| }} | |||
| </ref> | |||
| ⚫ | The compound aluminium lithium bis(binaphthoxide) (ALB) is prepared by reaction of BINOL with ].<ref>''A practical large-scale synthesis of enantiomerically pure 3-cyclohexanone via catalytic asymmetric Michael reaction'' ], Volume 58, Issue 13, 25 March '''2002''', Pages 2585–2588 Youjun Xu, Ken Ohori, Takashi Ohshima, Masakatsu Shibasaki {{doi|10.1016/S0040-4020(02)00141-2}}</ref> In a different stoichiometric ratio (1:1 BINOL/LiAlH<sub>4</sub> instead of 2:1), the chiral reducing agent BINAL (lithium dihydrido(binaphthoxy)aluminate) is produced.<ref>{{Citation|last1=Gopalan|first1=Aravamudan S.|title=Lithium Aluminum Hydride-2,2{{prime}}-Dihydroxy-1,1{{prime}}-binaphthyl|date=2001-04-15|encyclopedia=Encyclopedia of Reagents for Organic Synthesis|publisher=John Wiley & Sons, Ltd|language=en|doi=10.1002/047084289x.rl041|isbn=0471936235|last2=Jacobs|first2=Hollie K.}}</ref> | ||
| ⚫ | ==BINOL |
||
| Many variations of BINOL exist, ] being one of them. | |||
| ⚫ | :] | ||
| ⚫ | The compound |
||
| ⚫ | :] | ||
| It has been employed in an ] with ] and ]: | It has been employed in an ] with ] and ]: | ||
| :] | :] | ||
| ==See also== | |||
| * ] | |||
| ==References== | ==References== | ||
| {{Reflist}} | {{Reflist}} | ||
| {{DEFAULTSORT:Bi-2-Naphthol, 1,1'-}} | {{DEFAULTSORT:Bi-2-Naphthol, 1, 1'-}} | ||
| ] | ] | ||
| ] | |||
| ] | |||
| ⚫ | |||
| ] | |||
| ] | |||
| ] | |||
Latest revision as of 22:34, 5 August 2024
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name -2,2′-diol | |||
Other names
| |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.009.104 | ||
| PubChem CID | |||
| UNII |
| ||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
| Chemical formula | C20H14O2 | ||
| Molar mass | 286.32 g/mol | ||
| Melting point | 205 to 211 °C (401 to 412 °F; 478 to 484 K) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
1,1′-Bi-2-naphthol (BINOL) is an organic compound that is often used as a ligand for transition-metal catalysed asymmetric synthesis. BINOL has axial chirality and the two enantiomers can be readily separated and are stable toward racemisation. The specific rotation of the two enantiomers is 35.5° (c = 1 in THF), with the R enantiomer being the dextrorotary one. BINOL is a precursor for another chiral ligand called BINAP. The volumetric mass density of the two enantiomers is 0.62 g cm.
Preparation
The organic synthesis of BINOL is not a challenge as such but the preparation of the individual enantiomers is.
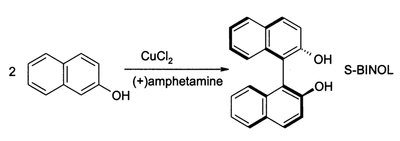
(S)-BINOL can be prepared directly from an asymmetric oxidative coupling of 2-naphthol with copper(II) chloride. The chiral ligand in this reaction is (S)-(+)-amphetamine.
Racemic BINOL can also be produced using iron(III) chloride as an oxidant. The mechanism involves complexation of iron(III) into the hydroxyl, followed by a radical coupling reaction of the naphthol rings initiated by iron(III) reducing into iron(II).
Optically active BINOL can also be obtained from racemic BINOL by optical resolution. In one method, the alkaloid N-benzylcinchonidinium chloride forms a crystalline inclusion compound. The inclusion compound of the (S)-enantiomer is soluble in acetonitrile but that of the (R)-enantiomer is not. In another method BINOL is esterified with pentanoyl chloride. The enzyme cholesterol esterase hydrolyses the (S)-diester but not the (R)-diester. The (R)-dipentanoate is hydrolysed in a second step with sodium methoxide. The third method employs HPLC with chiral stationary phases.
BINOL derivatives

Aside from the starting materials derived directly from the chiral pool, (R)- and (S)-BINOL in high enantiopurity (>99% enantiomeric excess) are two of the most inexpensive sources of chirality for organic synthesis, costing less than US$0.60 per gram when purchased in bulk from chemical suppliers. As a consequence, it serves as an important starting material for other sources of chirality for stereoselective synthesis, both stoichiometric and substoichiometric (catalytic).
Many important chiral ligands are constructed from the binaphthyl scaffold and ultimately derived from BINOL as a starting material, BINAP being one of the most well known and important.
The compound aluminium lithium bis(binaphthoxide) (ALB) is prepared by reaction of BINOL with lithium aluminium hydride. In a different stoichiometric ratio (1:1 BINOL/LiAlH4 instead of 2:1), the chiral reducing agent BINAL (lithium dihydrido(binaphthoxy)aluminate) is produced.
It has been employed in an asymmetric Michael reaction with cyclohexenone and dimethyl malonate:
See also
References
- Datasheet, chemexper.com
- Brussee, J.; Jansen, A. C. A. (1983). "A highly stereoselective synthesis of S-(−)--2,2′-diol". Tetrahedron Letters. 24 (31): 3261–3262. doi:10.1016/S0040-4039(00)88151-4.
- ^ "RESOLUTION OF 1,1'-BI-2-NAPHTHOL" Archived 2012-07-16 at the Wayback Machine, Dongwei Cai, David L. Hughes, Thomas R. Verhoeven, and Paul J. Reider, in Organic Syntheses Coll. Vol. 10, p.93; Vol. 76, p.1
- "(S)-(−)- and (R)-(+)-1,1′-bi-2-naphthol" Archived 2005-04-18 at the Wayback Machine, Romas J. Kazlauskas in Organic Syntheses, Coll. Vol. 9, p.77; Vol. 70, p.60
- Landek, G.; Vinković, M.; Kontrec, D.; Vinković, V. (2006). "Influence of mobile phase and temperature on separation of 1,1′-binaphthyl-2,2′-diol enantiomers with brush type chiral stationary phases derived from L-leucine". Chromatographia. 64 (7–8): 469–473. doi:10.1365/s10337-006-0041-5. S2CID 95785346.
- Parmar, Dixit; Sugiono, Erli; Raja, Sadiya; Rueping, Magnus (2014). "Complete Field Guide to Asymmetric BINOL-Phosphate Derived Brønsted Acid and Metal Catalysis: History and Classification by Mode of Activation; Brønsted Acidity, Hydrogen Bonding, Ion Pairing, and Metal Phosphates". Chemical Reviews. 114 (18): 9047–9153. doi:10.1021/cr5001496. PMID 25203602.
- Yang, Jin-Fei; Wang, Rong-Hua; Wang, Yin-Xia; Yao, Wei-Wei; Liu, Qi-Sheng; Ye, Mengchun (2016-10-11). "Ligand-Accelerated Direct C−H Arylation of BINOL: A Rapid One-Step Synthesis of Racemic 3,3′-Diaryl BINOLs". Angewandte Chemie International Edition. 55 (45): 14116–14120. doi:10.1002/anie.201607893. ISSN 1433-7851. PMID 27726256.
- A practical large-scale synthesis of enantiomerically pure 3-cyclohexanone via catalytic asymmetric Michael reaction Tetrahedron, Volume 58, Issue 13, 25 March 2002, Pages 2585–2588 Youjun Xu, Ken Ohori, Takashi Ohshima, Masakatsu Shibasaki doi:10.1016/S0040-4020(02)00141-2
- Gopalan, Aravamudan S.; Jacobs, Hollie K. (2001-04-15), "Lithium Aluminum Hydride-2,2′-Dihydroxy-1,1′-binaphthyl", Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, Ltd, doi:10.1002/047084289x.rl041, ISBN 0471936235