| Revision as of 00:03, 4 January 2013 view sourceLenny Kaufman (talk | contribs)9,129 edits →Prostaglandin D2: completing reversion← Previous edit | Latest revision as of 18:52, 24 February 2024 view source J.pshine5t (talk | contribs)Extended confirmed users3,749 edits Rescuing 18 sources and tagging 0 as dead.) #IABot (v2.0.9.5Tag: IABotManagementConsole [1.3] | ||
| (648 intermediate revisions by more than 100 users not shown) | |||
| Line 1: | Line 1: | ||
| {{Short description|none}} | |||
| {{pp-semi|small=yes}} | |||
| {{Use mdy dates|date=September 2012}} | {{Use mdy dates|date=September 2012}} | ||
| {{Infobox medical intervention | |||
| In the United States, '''Baldness treatment''' is a $1 billion per year industry. Past the early 20s, the incidence of balding or ] in males is roughly equivalent to chronological age. Thus, by age 50, roughly half of men experience ] (MPB).)<ref>Proctor PH. Hair-raising. The latest news on male-pattern baldness. Adv Nurse Pract. 1999 Apr;7(4):39-42, 83. Review. PubMed PMID 10382384.</ref><ref name=50percent>{{Cite web|url=http://www.intelihealth.com/IH/ihtIH/WSIHW000/9023/24253/352721.html?d=dmtContent|author=Whyche, Stephanie|accessdate=July 26, 2006|title=The Bald Truth About Hair Loss in Young Men}}</ref> Beginning in the 1980s, drug therapy gives a treatment option for ] in both men and women. For example, increased understanding of the role of ] (DHT) in male and female pattern baldness has led to targeted intervention to prevent this hormone from acting on scalp androgen receptors. Like many other medical conditions, baldness can be treated, but not cured. Moreover, experts warn that many treatments marketed as hair loss cures are ineffective aside from the ].<ref name="webmd_treatments" /> | |||
| | name = Management of hair loss | |||
| | synonym = | |||
| | image = | |||
| | caption = | |||
| | alt = | |||
| | pronounce = | |||
| | specialty =dermatology | |||
| | synonyms = | |||
| | ICD10 = | |||
| | ICD9 = | |||
| | ICD9unlinked = | |||
| | CPT = | |||
| | MeshID = | |||
| | LOINC = | |||
| | other_codes = | |||
| | MedlinePlus = | |||
| | eMedicine = | |||
| }} | |||
| The '''management of hair loss''', includes prevention and treatment of ], ], and ], and regrowth of hair. | |||
| ==Prevention== | |||
| ==Lifestyle modification== | |||
| Scratching of itchy scalp may contribute to hair loss.<ref>{{cite journal |last1=Bin Saif |first1=GA |last2=Ericson |first2=ME |last3=Yosipovitch |first3=G |title=The itchy scalp—scratching for an explanation. |journal=Experimental Dermatology |date=December 2011 |volume=20 |issue=12 |pages=959–68 |doi=10.1111/j.1600-0625.2011.01389.x |pmid=22092575|pmc=3233984 }}</ref> | |||
| Several genetic factors determine susceptibility to male pattern baldness (MPB). These factors include androgen receptor polymorphisms and scalp ] levels. Other variables include androgen receptor density and distribution in the scalp. Vigorous ] daily, as opposed to short workout periods designed to raise androgen levels and build muscle or more sporadic exercise, and a diet which is adequate, yet more moderate in fat and total calorie intake, have been shown to reduce baseline insulin levels as well as baseline total and free testosterone.<ref>{{Cite journal|author=Daly W, Seegers CA, Rubin DA, Dobridge JD, Hackney AC|title=Relationship between stress hormones and testosterone with prolonged endurance exercise|journal=European Journal of Applied Physiology|volume=93|issue=4|pages=375–80|year=2005|month=January|pmid=15618989|doi=10.1007/s00421-004-1223-1}}</ref> | |||
| ===Health=== | |||
| Lower insulin levels and reduced stress both result in raised levels of ] (SHBG). SHBG binds to testosterone. <!-- removed this sentence because incomprehensible, confusing, and apparently irrelevant: SHBG binds to testosterone, and low SHBG levels are a symptom of low testosterone levels and therefore ].--> Only free testosterone improves muscle growth and insulin sensitivity, but free testosterone can also be converted to the ineffective (regarding insulin sensitivity) DHT. The levels of free androgens and not of total androgens are relevant to the levels of DHT in the scalp and the progression of male pattern baldness.<ref name=barnard1>{{Cite journal|author=Barnard RJ, Aronson WJ, Tymchuk CN, Ngo TH|title=Prostate cancer: another aspect of the insulin-resistance syndrome?|journal=Obesity Reviews|volume=3|issue=4|pages=303–8|year=2002|month=November|pmid=12458975|doi=10.1046/j.1467-789X.2002.00081.x}}</ref><ref name=barnard2>{{Cite journal|author=Barnard RJ, Aronson WJ|title=Preclinical models relevant to diet, exercise, and cancer risk|journal=Recent ZIMHATA ncer Research|volume=166|pages=47–61|year=2005|pmid=15648182|doi=10.1007/3-540-26980-0_4}}</ref> | |||
| ====Body weight==== | |||
| ] occasionally correlates with ]. This may be because typically bald men have low testosterone levels (]) which can easily trigger also ] and ].<ref>http://www3.interscience.wiley.com/cgi-bin/fulltext/121568539/PDFSTART?CRETRY=1&SRETRY=0</ref> Medically increasing androgen levels improves this condition, demonstrating that androgens do not cause metabolic syndrome. Instead, high insulin levels (and possibly chronic inflammation)<ref name=joffe>{{Cite journal|author=Joffe HV, Ridker PM, Manson JE, Cook NR, Buring JE, Rexrode KM|title=Sex hormone-binding globulin and serum testosterone are inversely associated with C-reactive protein levels in postmenopausal women at high risk for cardiovascular disease|journal=Annals of Epidemiology|volume=16|issue=2|pages=105–12|year=2006|month=February|pmid=16216530|doi=10.1016/j.annepidem.2005.07.055}}</ref> seem the likely link in the demonstrated correlation between baldness and metabolic syndrome. This reinforces the notion that behaviors which help to keep insulin levels low and reduce chronic inflammation might also help to preserve hair.<ref name=cikim>{{Cite journal|author=Cikim AS, Ozbey N, Sencer E, Molvalilar S, Orhan Y|title=Associations among sex hormone binding globulin concentrations and characteristics of the metabolic syndrome in obese women|journal=Diabetes, Nutrition & Metabolism|volume=17|issue=5|pages=290–5 |year=2004 |month=October |pmid=16295051}}</ref><ref name=heald>{{Cite journal|author=Heald AH, Anderson SG, Ivison F, ''et al.''|title=Low sex hormone binding globulin is a potential marker for the metabolic syndrome in different ethnic groups|journal=Experimental and Clinical Endocrinology & Diabetes|volume=113|issue=9|pages=522–8|year=2005|month=October|pmid=16235154|doi=10.1055/s-2005-865807}}</ref> | |||
| Prevention of ] helps. Obesity is linked to ].<ref>{{cite journal |last1=Morinaga |first1=H |last2=Mohri |first2=Y |last3=Grachtchouk |first3=M |last4=Asakawa |first4=K |last5=Matsumura |first5=H |last6=Oshima |first6=M |last7=Takayama |first7=N |last8=Kato |first8=T |last9=Nishimori |first9=Y |last10=Sorimachi |first10=Y |last11=Takubo |first11=K |last12=Suganami |first12=T |last13=Iwama |first13=A |last14=Iwakura |first14=Y |last15=Dlugosz |first15=AA |last16=Nishimura |first16=EK |title=Obesity accelerates hair thinning by stem cell-centric converging mechanisms. |journal=Nature |date=July 2021 |volume=595 |issue=7866 |pages=266–271 |doi=10.1038/s41586-021-03624-x |pmid=34163066|pmc=9600322 |bibcode=2021Natur.595..266M |s2cid=235625692 }}</ref> | |||
| == |
====Healthy diet==== | ||
| {{see also|Healthy diet}} | |||
| Machines for cooling of the scalp are available and prescribed to patients to prevent hair loss due to ].<ref>{{cite pmid|23187775}}</ref> | |||
| Oils with fatty acids that has been studied to prevent dermatitis includes:<ref>{{cite journal |last1=Søyland |first1=E |last2=Funk |first2=J |last3=Rajka |first3=G |last4=Sandberg |first4=M |last5=Thune |first5=P |last6=Rustad |first6=L |last7=Helland |first7=S |last8=Middelfart |first8=K |last9=Odu |first9=S |last10=Falk |first10=ES |title=Dietary supplementation with very long-chain n-3 fatty acids in patients with atopic dermatitis. A double-blind, multicentre study. |journal=The British Journal of Dermatology |date=June 1994 |volume=130 |issue=6 |pages=757–64 |doi=10.1111/j.1365-2133.1994.tb03414.x |pmid=8011502|s2cid=25066444 }}</ref><ref>{{cite journal |last1=Callaway |first1=J |last2=Schwab |first2=U |last3=Harvima |first3=I |last4=Halonen |first4=P |last5=Mykkänen |first5=O |last6=Hyvönen |first6=P |last7=Järvinen |first7=T |title=Efficacy of dietary hempseed oil in patients with atopic dermatitis. |journal=The Journal of Dermatological Treatment |date=April 2005 |volume=16 |issue=2 |pages=87–94 |doi=10.1080/09546630510035832 |pmid=16019622|s2cid=18445488 }}</ref> | |||
| ==Medications== | |||
| * ]: ] (LA) | |||
| The ] (FDA) has approved two medications for the treatment of male pattern baldness: ] (''Rogaine'')<ref name="webmd_treatments">{{Cite web|url=http://www.webmd.com/skin-beauty/guide/hair-loss-treatments|title=Propecia & Rogaine for Treating Male Pattern Baldness|publisher=Webmd.com|accessdate=May 19, 2010}}</ref> and ] (''Propecia'').<ref name="fda.gov">{{Cite web|url=http://www.fda.gov/cdrh/pdf6/K060305.pdf|title=FDA's Role|publisher=Fda.gov|date=June 23, 2009|accessdate=May 19, 2010}}</ref> | |||
| * ]: ] (EPA) and ] (DHA) | |||
| * ]: ] (LA), and ] (ALA) | |||
| ====Alternative medicine==== | |||
| Both agents are recommended as ] for male pattern baldness. They may also be used simultaneously when hair loss is progressive or further regrowth is desired after 12 months.<ref name="webmd_treatments" /> | |||
| The scalp must be cleaned from ], sweat, and dirt, prior to topical application, for agents to penetrate it. | |||
| A 2020 ] on agents used to treat androgenic alopecia found that:<ref name="PMID33011954">{{cite journal | vauthors = Ashique S, Sandhu NK, Haque SN, Koley K | title = A Systemic Review on Topical Marketed Formulations, Natural Products, and Oral Supplements to Prevent Androgenic Alopecia: A Review | journal = Natural Products and Bioprospecting | volume = 10 | issue = 6 | pages = 345–365 | date = December 2020 | pmid = 33011954 | pmc = 7648777 | doi = 10.1007/s13659-020-00267-9 }}</ref> | |||
| ===Minoxidil and kopexil=== | |||
| * Oral supplementation of ], ], ], ], ], and trace element ], ], and ], will prevent androgenic alopecia caused by ]. ]s can be used. | |||
| ] (''Rogaine'') is a ]. It was originally used as the oral drug Loniten to treat ]. However, minoxidil was discovered to have the side effects of hair growth and reversing baldness. Consequently, in the 1980s, Upjohn Corporation received FDA approval to market a topical solution that contained 2% minoxidil to be used to treat baldness and hair loss as Rogaine, marketed as Regaine outside the USA. | |||
| * Topical application of ] juice, ] oil, ], ] oil, ], ] gel, ], ], amino acids, and ] helped prevent hair loss. | |||
| ===Heating, ventilation, and air conditioning (HVAC)=== | |||
| Objective evidence shows that minoxidil is effective for treatment of male-pattern hair loss in both the frontal and the vertex areas of the scalp. At the conclusion of a 48-week study, improvements were seen in the vertex area regions of 51% of men using 5% minoxidil, 42% using 2% minoxidil, and 13% of placebo users. Among these men, moderate to great increases in hair growth were seen in the frontal scalp regions of 19% of men using 5% minoxidil, 10% using 2% minoxidil, and 3% of placebo users.<ref></ref> | |||
| ====Humidifier==== | |||
| The mechanism of action of minoxidil is not known. Its action as a vasodilator may involve opening vascular smooth muscle ] (K-channels), perhaps through mimicking the most important natural vasodilator, ], whose structure minoxidil incorporates.<ref>], Endothelium-Derived Relaxing Factor and Minoxidil: Active Mechanisms in Hair Growth, Archives in Dermatology, vol 125, August, 1989 </ref> As it wakes up dormant ] follicles, this agent may cause loss-phase hairs to shed earlier than they normally would. This "shed" is generally followed by the growth of new, thicker hairs. As the hair-growth-stimulating effect is temporary and does not seem to change the follicle in any other way, minoxidil needs to be applied regularly (once or twice daily) for hair gained to be maintained.<ref name="Austria-Codex">{{cite book|title=Austria-Codex|editor=Jasek, W|publisher=Österreichischer Apothekerverlag|location=Vienna|year=2007|edition=2007/2008|volume=4|page=9673|isbn=978-3-85200-181-4|language=German}}</ref> | |||
| A ] can be used to prevent low indoor humidity during ] (especially with indoor heating), and ].<ref>{{cite web |title=Seasonal Shedding Hair in Summer or Winter! |url=https://mdm-communications.com/seasonal-shedding-hair-summer-winter/2/ |website=MDM Health |date=28 October 2015 |access-date=December 9, 2021 |archive-date=December 9, 2021 |archive-url=https://web.archive.org/web/20211209232940/https://mdm-communications.com/seasonal-shedding-hair-summer-winter/2/ |url-status=live }}</ref> | |||
| Commonly, patients with ] experience mild redness, scaly skin lesions and in some cases ].<ref>{{cite web|url=http://www.mamashealth.com/skin/seb.asp|title=What is Seborrheic Dermatitis?|access-date=June 11, 2010|url-status=live|archive-url=https://web.archive.org/web/20100420023503/http://www.mamashealth.com/skin/seb.asp|archive-date=April 20, 2010}}</ref> | |||
| A slightly changed ] of minoxidil missing one ] substituent is ] (brand name ''Aminexil''); for kopexil there is – until now – no proof of ] against androgenic alopecia.<ref>Wolfgang Raab: {{Google books|NoH2I_oiZLQC|Haarerkrankungen in Der Dermatologischen Praxis|page=82}} (German).</ref> | |||
| Low humidity can cause adverse health effects and may cause ],<ref>{{cite journal | vauthors = Engebretsen KA, Johansen JD, Kezic S, Linneberg A, Thyssen JP | title = The effect of environmental humidity and temperature on skin barrier function and dermatitis | journal = Journal of the European Academy of Dermatology and Venereology | volume = 30 | issue = 2 | pages = 223–249 | date = February 2016 | pmid = 26449379 | doi = 10.1111/jdv.13301 | s2cid = 12378072 | doi-access = free }}</ref> and ].<ref>{{cite journal | vauthors = Araya M, Kulthanan K, Jiamton S | title = Clinical Characteristics and Quality of Life of Seborrheic Dermatitis Patients in a Tropical Country | journal = Indian Journal of Dermatology | volume = 60 | issue = 5 | pages = 519 | date = September 2015 | pmid = 26538714 | pmc = 4601435 | doi = 10.4103/0019-5154.164410 | doi-access = free }}</ref> | |||
| ===Finasteride=== | |||
| ]) 1 mg tablets]] | |||
| ==Treatments== | |||
| ] (marketed by ] under the trade names ''Propecia'' and ''Proscar'') is a type-2 ] ] ] . | |||
| ===Combination therapy=== | |||
| It was originally ] for treatment of ] (BPH) and works by binding to ], the enzyme responsible for the conversion of free testosterone to DHT. | |||
| Combinations of finasteride, minoxidil and ketoconazole are more effective than individual use.<ref>{{cite journal | vauthors = Khandpur S, Suman M, Reddy BS | title = Comparative efficacy of various treatment regimens for androgenetic alopecia in men | journal = The Journal of Dermatology | volume = 29 | issue = 8 | pages = 489–498 | date = August 2002 | pmid = 12227482 | doi = 10.1111/j.1346-8138.2002.tb00314.x | s2cid = 20886812 }}</ref> | |||
| Combination therapy of LLLT or microneedling with finasteride<ref name="Darwin Heyes Hirt Wikramanayake 2017 pp. 425–434"/> or minoxidil demonstrated substantive increases in hair count.<ref name="Zhou Chen Qu Zhang 2020 p.">{{cite journal | last1=Zhou | first1=Yi | last2=Chen | first2=Chenglong | last3=Qu | first3=Qian | last4=Zhang | first4=Chunhua | last5=Wang | first5=Jin | last6=Fan | first6=Zhexiang | last7=Miao | first7=Yong | last8=Hu | first8=Zhiqi | title=The effectiveness of combination therapies for androgenetic alopecia: A systematic review and meta-analysis | journal=Dermatologic Therapy | publisher=Hindawi Limited | volume=33 | issue=4 | year=2020 | pages=e13741 | issn=1396-0296 | doi=10.1111/dth.13741 | pmid=32478968 | doi-access=free }}</ref> | |||
| Merck sought to find the smallest effective quantity of finasteride and test its long-term effects on 1,553 men between ages 18 and 41 with mildly to moderately thinning hair. Based on their research, 1 mg/day was selected, and after two years of daily treatment, over 83% of the 1,553 men experiencing male hair loss had actually maintained or increased their hair count from baseline. Visual assessments concluded that over 80% had improved appearances.<ref>{{cite journal |last1=Barth |laysummary=http://www.medscape.com/viewarticle/406344_2 |first1=J |title=Should men still go bald gracefully? |journal=The Lancet |laysource=Medscape |volume=355 |pages=161–2 |year=2000 | doi = 10.1016/S0140-6736(99)00412-2 | pmid=10675111 |issue=9199}}</ref> | |||
| ===Medication=== | |||
| In 1997, finasteride was approved by the US FDA for the casual treatment of ]. A 5-year study revealed that 9 of 10 men taking finasteride (1 mg/day) experienced visible results (42% of men taking Propecia experienced no further hair loss while 48% experienced no further hair loss and hair regrowth).<ref></ref> In clinical studies, finasteride, like ], was shown to work on both the ] area and the ] area, but is most successful in the crown area.<ref name=finasteride1>{{cite journal |author=Leyden J, Dunlap F, Miller B, ''et al.'' |title=Finasteride in the treatment of men with frontal male pattern hair loss |journal=J. Am. Acad. Dermatol. |volume=40 |issue=6 Pt 1 |pages=930–7 |year=1999 |month=June |pmid=10365924 |doi= 10.1016/S0190-9622(99)70081-2|url=http://linkinghub.elsevier.com/retrieve/pii/S0190-9622(99)70081-2}}</ref> | |||
| === |
====Add-hocs==== | ||
| The topical ] of synthetic medicines such as ]s, as well as phytochemicals such as ], has been improved with incorporation of drugs in ], or ].<ref>{{cite journal | vauthors = Cardoso CO, Tolentino S, Gratieri T, Cunha-Filho M, Lopez RF, Gelfuso GM | title = Topical Treatment for Scarring and Non-Scarring Alopecia: An Overview of the Current Evidence | journal = Clinical, Cosmetic and Investigational Dermatology | volume = 14 | pages = 485–499 | date = 2021 | pmid = 34012282 | pmc = 8126704 | doi = 10.2147/CCID.S284435 | doi-access = free }}</ref> | |||
| ]) 500 ] capsules]] | |||
| ====Synthetic medicine==== | |||
| ] is a ] that ] of ] to ] (DHT). | |||
| Treatments for the various forms of hair loss have only moderate success.<ref name=Levy2013>{{cite journal | vauthors = Levy LL, Emer JJ | title = Female pattern alopecia: current perspectives | journal = International Journal of Women's Health | volume = 5 | pages = 541–556 | date = August 2013 | pmid = 24039457 | pmc = 3769411 | doi = 10.2147/IJWH.S49337 | doi-access = free }}</ref> Three medications have evidence to support their use in male pattern hair loss: ], ] and ].<ref name=Ban2013/> They typically work better to prevent further hair loss than to regrow lost hair.<ref name=Ban2013/> | |||
| They may be used together when hair loss is progressive or further regrowth is desired after 12 months.<ref name="webmd_treatments">{{cite web |url=http://www.webmd.com/skin-beauty/guide/hair-loss-treatments |title=Propecia & Rogaine for Treating Male Pattern Baldness |publisher=Webmd.com |access-date=May 19, 2010 |archive-date=July 9, 2008 |archive-url=https://web.archive.org/web/20080709023925/http://www.webmd.com/skin-beauty/guide/hair-loss-treatments |url-status=live }}</ref> Other medications include ], and in female androgenic alopecia ] and ].<ref name="pmid22735503">{{cite journal | vauthors = McElwee KJ, Shapiro JS | title = Promising therapies for treating and/or preventing androgenic alopecia | journal = Skin Therapy Letter | volume = 17 | issue = 6 | pages = 1–4 | date = June 2012 | pmid = 22735503 }}</ref> | |||
| Dutasteride inhibits both isoforms of 5-alpha reductase, type I and type II, whereas finasteride only inhibits type II. | |||
| ===== Baricitinib ===== | |||
| Dutasteride (trademark name ''Avodart'', manufactured by ]) is ] for the treatment of ] (BPH).<ref></ref> | |||
| In June 2022, the FDA authorized ] for the treatment of severe alopecia areata.<ref name="FDA PR 20220613">{{cite press release | title=FDA Approves First Systemic Treatment for Alopecia Areata | website=U.S. ] (FDA) | date=13 June 2022 | url=https://www.fda.gov/news-events/press-announcements/fda-approves-first-systemic-treatment-alopecia-areata | access-date=13 June 2022 | archive-date=14 June 2022 | archive-url=https://web.archive.org/web/20220614051142/https://www.fda.gov/news-events/press-announcements/fda-approves-first-systemic-treatment-alopecia-areata | url-status=live }} {{PD-notice}}</ref><ref name="Lilly PR 20220613">{{cite press release | title=FDA Approves Lilly and Incyte's Olumiant (baricitinib) As First and Only Systemic Medicine for Adults with Severe Alopecia Areata | publisher=Eli Lilly | via=PR Newswire | date=13 June 2022 | url=https://www.prnewswire.com/news-releases/fda-approves-lilly-and-incytes-olumiant-baricitinib-as-first-and-only-systemic-medicine-for-adults-with-severe-alopecia-areata-301567120.html | access-date=13 June 2022 | archive-date=14 June 2022 | archive-url=https://web.archive.org/web/20220614034511/https://www.prnewswire.com/news-releases/fda-approves-lilly-and-incytes-olumiant-baricitinib-as-first-and-only-systemic-medicine-for-adults-with-severe-alopecia-areata-301567120.html | url-status=live }}</ref> | |||
| ===== Minoxidil ===== | |||
| Phase I and II clinical trials for dutasteride as a ] drug were undertaken, but called off in late 2002, with reasons for termination unreported. | |||
| ], applied topically, is widely used for the treatment of hair loss. It may be effective in helping promote hair growth in both men and women with ].<ref name="2014AArev"/><ref>{{cite journal | vauthors = van Zuuren EJ, Fedorowicz Z, Schoones J | title = Interventions for female pattern hair loss | journal = The Cochrane Database of Systematic Reviews | volume = 2016 | issue = 5 | pages = CD007628 | date = May 2016 | pmid = 27225981 | pmc = 6457957 | doi = 10.1002/14651858.CD007628.pub4 }}</ref> About 40% of men experience hair regrowth after 3–6 months.<ref>{{cite journal | vauthors = Goren A, Shapiro J, Roberts J, McCoy J, Desai N, Zarrab Z, Pietrzak A, Lotti T | display-authors = 6 | title = Clinical utility and validity of minoxidil response testing in androgenetic alopecia | journal = Dermatologic Therapy | volume = 28 | issue = 1 | pages = 13–16 | year = 2015 | pmid = 25112173 | doi = 10.1111/dth.12164 | s2cid = 205082682 | doi-access = free }}</ref> It is the only topical product that is FDA approved in America for androgenic hair loss.<ref name="2014AArev">{{cite journal | vauthors = Varothai S, Bergfeld WF | title = Androgenetic alopecia: an evidence-based treatment update | journal = American Journal of Clinical Dermatology | volume = 15 | issue = 3 | pages = 217–230 | date = July 2014 | pmid = 24848508 | doi = 10.1007/s40257-014-0077-5 | s2cid = 31245042 }}</ref> However, increased hair loss has been reported.<ref>{{cite web|url=https://www.drugs.com/sfx/rogaine-side-effects.html|title=Rogaine Side Effects in Detail - Drugs.com|website=drugs.com|access-date=January 28, 2018|archive-date=September 22, 2017|archive-url=https://web.archive.org/web/20170922052041/https://www.drugs.com/sfx/rogaine-side-effects.html|url-status=live}}</ref><ref name="Drugs.com">{{cite web|url = https://www.drugs.com/pro/minoxidil.html|title = Minoxidil Official FDA information, side effects and uses|publisher = Drugs.com|access-date = January 28, 2018|archive-date = September 22, 2017|archive-url = https://web.archive.org/web/20170922051930/https://www.drugs.com/pro/minoxidil.html|url-status = live}}</ref> | |||
| =====Antiandrogens===== | |||
| The phase II results indicated that dutasteride at both 0.5 mg and 2.5 mg/day generated a superior hair count to finasteride 5 mg at 12 and 24 weeks.<ref>{{cite journal |author=Olsen EA, Hordinsky M, Whiting D, ''et al.'' |title=The importance of dual 5alpha-reductase inhibition in the treatment of MPB: results of a randomized placebo-controlled study of dutasteride versus finasteride |journal=J Am Acad Dermatol. |volume=55 |issue=6 |pages=1014–23 |year=2006 |month=Dec |pmid=17110217 |doi=10.1016/j.jaad.2006.05.007 }}</ref> | |||
| <!-- Finasteride --> | |||
| ] is used to treat ].<ref name=PropeciaLabel>{{Cite web|url=http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020788s024lbl.pdf|title=Propecia label|access-date=June 3, 2015|archive-date=February 10, 2017|archive-url=https://web.archive.org/web/20170210151045/http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020788s024lbl.pdf|url-status=live}}</ref> Treatment provides about 30% improvement in hair loss after six months of treatment, and effectiveness only persists as long as the drug is taken.<ref name=2014AArev/> There is no good evidence for its use in women.<ref name=Levy2013/> It may cause ], ] and ].<ref>{{cite journal | vauthors = Andersson S | title = Steroidogenic enzymes in skin | journal = European Journal of Dermatology | volume = 11 | issue = 4 | pages = 293–295 | year = 2001 | pmid = 11399532 }}</ref> | |||
| <!-- Dutasteride --> | |||
| Phase II study results at 24 weeks: | |||
| ] is also used in the treatment of male pattern hair loss and appears to have better effectiveness than finasteride for the condition.<ref>{{cite web|url=http://emc.medicines.org.uk/emc/assets/c/html/DisplayDoc.asp?DocumentID=11618|title=Avodart 0.5mg soft capsules – Summary of Product Characteristics (SmPC) – (eMC)|website=emc.medicines.org.uk|access-date=August 7, 2010|archive-date=April 26, 2006|archive-url=https://web.archive.org/web/20060426212556/http://emc.medicines.org.uk/emc/assets/c/html/displaydoc.asp?documentid=11618|url-status=dead}}</ref><ref name="pmid30863034">{{cite journal | vauthors = Zhou Z, Song S, Gao Z, Wu J, Ma J, Cui Y | title = The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and meta-analysis | journal = Clinical Interventions in Aging | volume = 14 | pages = 399–406 | date = 2019 | pmid = 30863034 | pmc = 6388756 | doi = 10.2147/CIA.S192435 | doi-access = free }}</ref><ref name="pmid17110217">{{cite journal | vauthors = Olsen EA, Hordinsky M, Whiting D, Stough D, Hobbs S, Ellis ML, Wilson T, Rittmaster RS | display-authors = 6 | title = The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride | journal = Journal of the American Academy of Dermatology | volume = 55 | issue = 6 | pages = 1014–1023 | date = December 2006 | pmid = 17110217 | doi = 10.1016/j.jaad.2006.05.007 }}</ref> While used off-label for male pattern hair loss in most of the world, dutasteride is specifically approved for this indication in ] and ].<ref name="pmid27489426">{{cite journal | vauthors = Choi GS, Kim JH, Oh SY, Park JM, Hong JS, Lee YS, Lee WS | title = Safety and Tolerability of the Dual 5-Alpha Reductase Inhibitor Dutasteride in the Treatment of Androgenetic Alopecia | journal = Annals of Dermatology | volume = 28 | issue = 4 | pages = 444–450 | date = August 2016 | pmid = 27489426 | pmc = 4969473 | doi = 10.5021/ad.2016.28.4.444 }}</ref><ref name="ShapiroOtberg2015">{{cite book|vauthors=Shapiro J, Otberg N|title=Hair Loss and Restoration, Second Edition|url=https://books.google.com/books?id=bJG9BwAAQBAJ&pg=PA39|date=17 April 2015|publisher=CRC Press|isbn=978-1-4822-3199-1|pages=39–|access-date=December 9, 2021|archive-date=January 12, 2023|archive-url=https://web.archive.org/web/20230112145741/https://books.google.com/books?id=bJG9BwAAQBAJ&pg=PA39|url-status=live}}</ref> | |||
| <!-- Spironolactone --> | |||
| * Placebo: – 32.3 | |||
| There is tentative support for ] in women.<ref name=Levy2013/> Due to its feminising side effects and risk of infertility it is not often used by men.<ref>{{cite journal | vauthors = Buchanan JF, Davis LJ | title = Drug-induced infertility | journal = Drug Intelligence & Clinical Pharmacy | volume = 18 | issue = 2 | pages = 122–132 | date = February 1984 | pmid = 6141923 | doi = 10.1177/106002808401800205 | s2cid = 24807929 }}</ref><ref>{{cite journal | vauthors = Sinclair R, Patel M, Dawson TL, Yazdabadi A, Yip L, Perez A, Rufaut NW | title = Hair loss in women: medical and cosmetic approaches to increase scalp hair fullness | journal = The British Journal of Dermatology | volume = 165 | issue = Suppl 3 | pages = 12–18 | date = December 2011 | pmid = 22171680 | doi = 10.1111/j.1365-2133.2011.10630.x | s2cid = 39734527 | doi-access = free }}</ref> It can also cause ], ], and ]. Also, women who are pregnant or trying to become pregnant generally cannot use the medication as it is a ], and can cause ambiguous genitalia in newborn children.<ref name="Spirono">{{cite journal | vauthors = Rathnayake D, Sinclair R | title = Use of spironolactone in dermatology | journal = Skinmed | volume = 8 | issue = 6 | pages = 328–32; quiz 333 | year = 2010 | pmid = 21413648 }}</ref> | |||
| <!-- Flutamide --> | |||
| * Dutasteride 0.5 mg: + 94.6 hairs | |||
| There is tentative evidence for ] in women; however, it is associated with relatively high rates of liver problems and strong recommendations have been made against its use.<ref name=Levy2013/><ref name="pmid28379593">{{cite journal | vauthors = Giorgetti R, di Muzio M, Giorgetti A, Girolami D, Borgia L, Tagliabracci A | title = Flutamide-induced hepatotoxicity: ethical and scientific issues | journal = European Review for Medical and Pharmacological Sciences | volume = 21 | issue = 1 Suppl | pages = 69–77 | date = March 2017 | pmid = 28379593 | doi = }}</ref> Like spironolactone, flutamide is typically only used by women.<ref>{{cite journal | vauthors = Yazdabadi A, Sinclair R | title = Treatment of female pattern hair loss with the androgen receptor antagonist flutamide | journal = The Australasian Journal of Dermatology | volume = 52 | issue = 2 | pages = 132–134 | date = May 2011 | pmid = 21605098 | doi = 10.1111/j.1440-0960.2010.00735.x | s2cid = 30128397 }}</ref> ] is another option for the treatment of female pattern hair loss.<ref name="pmid35032336">{{cite journal | vauthors = Carvalho RM, Santos LD, Ramos PM, Machado CJ, Acioly P, Frattini SC, Barcaui CB, Donda AL, Melo DF | display-authors = 6 | title = Bicalutamide and the new perspectives for female pattern hair loss treatment: What dermatologists should know | journal = Journal of Cosmetic Dermatology | volume = 21| issue = 10| date = January 2022 | pages = 4171–4175 | pmid = 35032336 | doi = 10.1111/jocd.14773 | s2cid = 253239337 }}</ref><ref name="pmid34668238">{{cite journal | vauthors = Meyer-Gonzalez T, Bacqueville D, Grimalt R, Mengeaud V, Piraccini BM, Rudnicka L, Saceda-Corralo D, Vogt A, Vano-Galvan S | display-authors = 6 | title = Current controversies in trichology: a European expert consensus statement | journal = Journal of the European Academy of Dermatology and Venereology | volume = 35 | issue = Suppl 2 | pages = 3–11 | date = November 2021 | pmid = 34668238 | doi = 10.1111/jdv.17601 | hdl = 11585/863826 | s2cid = 239029062 | hdl-access = free }}</ref><ref name="pmid34741573">{{cite journal | vauthors = Nestor MS, Ablon G, Gade A, Han H, Fischer DL | title = Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics | journal = Journal of Cosmetic Dermatology | volume = 20 | issue = 12 | pages = 3759–3781 | date = December 2021 | pmid = 34741573 | doi = 10.1111/jocd.14537 | pmc = 9298335 | s2cid = 243801494 }}</ref> It has a far lower risk of liver toxicity than flutamide and is said to have an excellent safety profile.<ref name="pmid34668238" /><ref name="pmid34741573" /><ref name="pmid33334002">{{cite journal | vauthors = Cignarella A, Mioni R, Sabbadin C, Dassie F, Parolin M, Vettor R, Barbot M, Scaroni C | display-authors = 6 | title = Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS | journal = International Journal of Molecular Sciences | volume = 21 | issue = 24 | page = 9554 | date = December 2020 | pmid = 33334002 | pmc = 7765466 | doi = 10.3390/ijms21249554 | doi-access = free }}</ref> However, bicalutamide retains a small risk of liver toxicity and for this reason periodic liver monitoring is recommended during treatment.<ref name="pmid34668238" /><ref name="pmid33334002" /> | |||
| =====Ketoconazole===== | |||
| * Dutasteride 2.5 mg: + 109.6 hairs | |||
| ] may help in women.<ref>{{cite journal | vauthors = Piérard-Franchimont C, De Doncker P, Cauwenbergh G, Piérard GE | title = Ketoconazole shampoo: effect of long-term use in androgenic alopecia | journal = Dermatology | volume = 196 | issue = 4 | pages = 474–477 | date = 1998 | pmid = 9669136 | doi = 10.1159/000017954 | s2cid = 30635892 }}</ref><ref>{{cite journal | vauthors = Fields JR, Vonu PM, Monir RL, Schoch JJ | title = Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review | journal = Dermatologic Therapy | volume = 33 | issue = 1 | pages = e13202 | date = January 2020 | pmid = 31858672 | doi = 10.1111/dth.13202 | s2cid = 209427721 | doi-access = free }}</ref> | |||
| ===Technological treatments=== | |||
| GlaxoSmithKline ran a phase III, six month study in ] to test the safety, tolerability and effectiveness of a once-daily dose of dutasteride (0.5 mg) in ] (MPB) at the vertex of the scalp (types IIIV, IV and V on the ]). The study was completed in January 2009.<ref>{{ClinicalTrialsGov|NCT00441116}}</ref><ref></ref> Future intentions by GlaxoSmithKline to continue or abandon plans for ] approval of dutasteride in treatment of MPB remain unknown. | |||
| ====Low-level laser therapy (LLLT)==== | |||
| ==Hair transplantation== | |||
| ] or photobiomodulation is also referred to as red light therapy and cold laser therapy. It is a non-invasive treatment option. | |||
| {{Main|Hair transplantation}} | |||
| LLLT is shown to increase hair density and growth in both genders. The types of devices (hat, comb, helmet) and duration did not alter the effectiveness,<ref name="Liu Liu Chen Chin 2019 pp. 1063–1069">{{cite journal | last1=Liu | first1=Kao-Hui | last2=Liu | first2=Donald | last3=Chen | first3=Yu-Tsung | last4=Chin | first4=Szu-Ying | title=Comparative effectiveness of low-level laser therapy for adult androgenic alopecia: a system review and meta-analysis of randomized controlled trials | journal=Lasers in Medical Science | publisher=Springer Science and Business Media LLC | volume=34 | issue=6 | date=2019-01-31 | issn=0268-8921 | doi=10.1007/s10103-019-02723-6 | pages=1063–1069| pmid=30706177 | s2cid=59524423 }}</ref> with more emphasis to be placed on lasers compared to LEDs.<ref name="Gupta Carviel 2019 pp. 643–647">{{cite journal | last1=Gupta | first1=A. K. | last2=Carviel | first2=J. L. | title=Meta-analysis of photobiomodulation for the treatment of androgenetic alopecia | journal=Journal of Dermatological Treatment | publisher=Informa UK Limited | volume=32 | issue=6 | date=2019-11-20 | issn=0954-6634 | doi=10.1080/09546634.2019.1688755 | pages=643–647| pmid=31746251 | s2cid=208185306 }}</ref> Ultraviolet and infrared light are more effective for alopecia areata, while red light and infrared light is more effective for androgenetic alopecia.<ref name="Zhang Su Ma Fu 2022 pp. 737–740">{{cite journal | last1=Zhang | first1=Yuehou | last2=Su | first2=Jianlong | last3=Ma | first3=Kui | last4=Fu | first4=Xiaobing | last5=Zhang | first5=Cuiping | title=Photobiomodulation Therapy With Different Wavebands for Hair Loss: A Systematic Review and Meta-Analysis | journal=Dermatologic Surgery | publisher=Ovid Technologies (Wolters Kluwer Health) | volume=48 | issue=7 | date=2022-04-25 | issn=1076-0512 | doi=10.1097/dss.0000000000003472 | pages=737–740| pmid=35510860 | s2cid=248526019 }}</ref> | |||
| A medical ] involves using small grafts of naturally-occurring units of one to four hairs, called ], to move hair-producing follicles to balding areas of the ] patient's scalp. These follicular units are surgically implanted in the scalp in very close proximity to one another and in large numbers. The grafts are obtained in one or both of the two primary methods of surgical extraction, ] (FUT) or ] (FUE). In FUT, a strip of skin containing many follicular units is extracted from the patient and then dissected under a stereoscopic dissecting microscope. Once dissected into individual follicular unit grafts, the surgeon implants the grafts into small incisions, called recipient sites, in the recipient area of the patient's scalp.<ref></ref> According to the new approach the roots of the hair are extracted from the "donor", and then literally cut into tiny strips, for transplantation. Bald patches on the scalp are isolated and given the donor strip, which is two to three millimeters thick only. When the transaction is completed, the patient should wear a head bandage for the next two days. This will allow the scalp to treat not weakened, and increase the chances of success.<ref>{{! cite news|url=http://healthyhairhighlights.com/new_approach_to_hair_transplant.html|title=Hair transplantation new and painless approach with tiny strips|publisher=Healthy Hair Highlights News|date=July 10, 2011 | accessdate=September 13, 2011}}</ref> | |||
| Medical reviews suggest that LLLT is as effective or potentially more than other non invasive and traditional therapies like minoxidil and finasteride but further studies such as RCTs, long term follow up studies, and larger double blinded trials need to be conducted to confirm the initial findings.<ref name="Gupta Bamimore Foley 2020 pp. 62–72">{{cite journal | last1=Gupta | first1=Aditya K. | last2=Bamimore | first2=Mary A. | last3=Foley | first3=Kelly A. | title=Efficacy of non-surgical treatments for androgenetic alopecia in men and women: a systematic review with network meta-analyses, and an assessment of evidence quality | journal=Journal of Dermatological Treatment | publisher=Informa UK Limited | volume=33 | issue=1 | date=2020-04-13 | issn=0954-6634 | doi=10.1080/09546634.2020.1749547 | pages=62–72 | pmid=32250713 | s2cid=215405183 | url=https://figshare.com/articles/journal_contribution/12087042 | access-date=August 31, 2023 | archive-date=February 24, 2024 | archive-url=https://web.archive.org/web/20240224185232/https://figshare.com/articles/journal_contribution/Efficacy_of_non-surgical_treatments_for_androgenetic_alopecia_in_men_and_women_a_systematic_review_with_network_meta-analyses_and_an_assessment_of_evidence_quality/12087042 | url-status=live }}</ref><ref name="Darwin Heyes Hirt Wikramanayake 2017 pp. 425–434">{{cite journal | last1=Darwin | first1=Evan | last2=Heyes | first2=Alexandra | last3=Hirt | first3=Penelope A. | last4=Wikramanayake | first4=Tongyu Cao | last5=Jimenez | first5=Joaquin J. | title=Low-level laser therapy for the treatment of androgenic alopecia: a review | journal=Lasers in Medical Science | publisher=Springer Science and Business Media LLC | volume=33 | issue=2 | date=2017-12-21 | issn=0268-8921 | doi=10.1007/s10103-017-2385-5 | pages=425–434| pmid=29270707 | s2cid=23783876 }}</ref><ref name="S P Y P p.">{{cite journal | last1=S | first1=Lueangarun | last2=P | first2=Visutjindaporn | last3=Y | first3=Parcharoen | last4=P | first4=Jamparuang | last5=T | first5=Tempark |title=A Systematic Review and Meta-analysis of Randomized Controlled Trials of United States Food and Drug Administration-Approved, Home-use, Low-Level Light/Laser Therapy Devices for Pattern Hair Loss: Device Design and Technology | journal=The Journal of Clinical and Aesthetic Dermatology | date=2021 | volume=14 | issue=11 | pages=E64–E75 | issn=1941-2789 | pmid=34980962 | pmc=8675345 }}</ref> | |||
| ==Laser therapy== | |||
| There are devices that use ] (LLLT) that claim to stimulate hair growth through "photo-biostimulation" of the hair follicles, although with limited evidence of benefit or safety.<ref>{{cite pmid|20689478}}</ref> | |||
| ===Platelet-rich plasma (PRP)=== | |||
| ==Research== | |||
| Using ones own cells and tissues and without harsh side effects, PRP is beneficial for alopecia areata<ref name="Tejapira Yongpisarn Sakpuwadol Suchonwanit 2022 p. ">{{cite journal | last1=Tejapira | first1=Kasama | last2=Yongpisarn | first2=Tanat | last3=Sakpuwadol | first3=Nawara | last4=Suchonwanit | first4=Poonkiat | title=Platelet-rich plasma in alopecia areata and primary cicatricial alopecias: A systematic review | journal=Frontiers in Medicine | publisher=Frontiers Media SA | volume=9 | date=2022-11-24 | issn=2296-858X | doi=10.3389/fmed.2022.1058431 | page=| pmid=36507528 | pmc=9731377 | doi-access=free }}</ref> and androgenetic alopecia and can be used as an alternative to minoxidil or finasteride.<ref name="Gentile Garcovich 2020 p=2702">{{cite journal | last1=Gentile | first1=Pietro | last2=Garcovich | first2=Simone | title=Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy | journal=International Journal of Molecular Sciences | publisher=MDPI AG | volume=21 | issue=8 | date=2020-04-13 | issn=1422-0067 | doi=10.3390/ijms21082702 | page=2702| pmid=32295047 | pmc=7216252 | doi-access=free }}</ref> It has been documented to improve hair density and thickness in both genders.<ref name="Evans Mwangi Pope Ivanic 2020 pp. 498–511">{{cite journal | last1=Evans | first1=Adam G. | last2=Mwangi | first2=James M. | last3=Pope | first3=Rand W. | last4=Ivanic | first4=Mirjana G. | last5=Botros | first5=Mina A. | last6=Glassman | first6=Gabriella E. | last7=Pearce | first7=F. Bennett | last8=Kassis | first8=Salam | title=Platelet-rich plasma as a therapy for androgenic alopecia: a systematic review and meta-analysis | journal=Journal of Dermatological Treatment | publisher=Informa UK Limited | volume=33 | issue=1 | date=2020-05-26 | issn=0954-6634 | doi=10.1080/09546634.2020.1770171 | pages=498–511 | pmid=32410524 | s2cid=218648227 | url=https://figshare.com/articles/journal_contribution/12376727 | access-date=August 31, 2023 | archive-date=February 24, 2024 | archive-url=https://web.archive.org/web/20240224185101/https://figshare.com/articles/journal_contribution/Platelet-rich_plasma_as_a_therapy_for_androgenic_alopecia_a_systematic_review_and_meta-analysis/12376727 | url-status=live }}</ref> A minimum of 3 treatments, once a month for 3 months are recommended, and afterwards a 3-6 month period of continual appointments for maintenance.<ref name="Gupta Cole Deutsch Everts 2019 pp. 1262–1273">{{cite journal | last1=Gupta | first1=Aditya K. | last2=Cole | first2=John | last3=Deutsch | first3=David P. | last4=Everts | first4=Peter A. | last5=Niedbalski | first5=Robert P. | last6=Panchaprateep | first6=Ratchathorn | last7=Rinaldi | first7=Fabio | last8=Rose | first8=Paul T. | last9=Sinclair | first9=Rodney | last10=Vogel | first10=James E. | last11=Welter | first11=Ryan J. | last12=Zufelt | first12=Michael D. | last13=Puig | first13=Carlos J. | title=Platelet-Rich Plasma as a Treatment for Androgenetic Alopecia | journal=Dermatologic Surgery | publisher=Ovid Technologies (Wolters Kluwer Health) | volume=45 | issue=10 | year=2019 | issn=1076-0512 | doi=10.1097/dss.0000000000001894 | pages=1262–1273| pmid=30882509 | s2cid=81980415 }}</ref> Factors that determine efficacy include amount of sessions, double versus single centrifugation, age and gender, and where the PRP is inserted.<ref name="Gupta Bamimore 2022 pp. 943–952">{{cite journal | last1=Gupta | first1=Aditya | last2=Bamimore | first2=Mary | title=Platelet-Rich Plasma Monotherapies for Androgenetic Alopecia: A Network Meta-Analysis and Meta-Regression Study | journal=Journal of Drugs in Dermatology | publisher=SanovaWorks | volume=21 | issue=9 | date=2022-08-01 | issn=1545-9616 | doi=10.36849/jdd.6948 | pages=943–952| pmid=36074501 | s2cid=252120370 }}</ref> | |||
| Future larger randomized controlled trials and other high quality studies are still recommended to be carried out and published for a stronger consensus.<ref name="Gupta Bamimore Foley 2020 pp. 62–72"/><ref name="Evans Mwangi Pope Ivanic 2020 pp. 498–511"/><ref name="Oth Stene Glineur Vujovic 2018 pp. 438–446">{{cite journal | last1=Oth | first1=O | last2=Stene | first2=JJ | last3=Glineur | first3=R | last4=Vujovic | first4=A | title=Injection of PRP (Platelet-rich plasma) as a treatment for androgenetic alopecia : a systematic review of the literature | journal=Revue Médicale de Bruxelles | publisher=AMUB/Revue Médicale de Bruxelles | volume=39 | issue=5 | year=2018 | issn=0035-3639 | doi=10.30637/2018.17-056 | pages=438–446| pmid=29869472 | doi-access=free }}</ref> Further development of a standardized practice for procedure is also recommended.<ref name="Tejapira Yongpisarn Sakpuwadol Suchonwanit 2022 p. "/> | |||
| === Caffeine === | |||
| ] has been identified as a stimulator of human hair growth ''in vitro'', and reduced ]-induced follicle growth suppression.<ref name="Int J Dermatol.">{{Cite journal|author=Fischer TW, Hipler UC, Elsner P|title=Effect of caffeine and testosterone on the proliferation of human hair follicles in vitro|journal=International Journal of Dermatology|volume=46|issue=1|pages=27–35|year=2007|month=January|pmid=17214716|doi=10.1111/j.1365-4632.2007.03119.x}}</ref> It has been demonstrated that the addition of caffeine to a shampoo-formulation is effective in administering caffeine to the ] in the scalp.<ref name="Skin Pharmacol Physiol.">{{Cite journal|author=Otberg N, Teichmann A, Rasuljev U, Sinkgraven R, Sterry W, Lademann J|title=Follicular penetration of topically applied caffeine via a shampoo formulation|journal=]|volume=20|issue=4|pages=195–8|year=2007|pmid=17396054|doi=10.1159/000101389}}</ref> Further research must be done to evaluate the efficacy and adequate dosage of caffeine in the treatment of ]. A spray made with coffee beans is claimed by the UK pharmacy chain Boots to prevent age-related hair loss in women.<ref>{{cite news|url=http://www.dailymail.co.uk/pages/live/articles/news/news.html?in_article_id=480216&in_page_id=1770|title=Spray that can stop a woman's hair from thinning | Mail Online|work=Daily Mail|date=June 9, 2007|accessdate=May 19, 2010 | location=London}}</ref> | |||
| === |
===Surgical treatments=== | ||
| In February 2008 researchers at the ] announced they have found the genetic basis of two distinct forms of inherited hair loss, opening a broad path to treatments for baldness. They found that a gene, ], causes a rare, inherited form of hair loss called hypotrichosis simplex. It is the first ] in humans known to play a role in hair growth. The fact that any receptor plays a specific role in hair growth was previously unknown to scientists and they are now attempting to gather more genetic and biological data.{{Citation needed|date=August 2010}} | |||
| ====Hair transplantation==== | |||
| In May 2009, researchers in Japan identified a ], ], that appears to determine cyclical hair loss in mice. The researchers also believe it may be responsible for hair loss, or alopecia, in people.<ref>{{cite news|url=http://uk.reuters.com/article/scienceNews/idUKTRE54O4D020090525 |title=Scientists identify gene that may explain hair loss | Reuters|publisher=Reuters UK|accessdate=May 19, 2010 | date=May 25, 2009}}</ref> | |||
| {{main|Hair transplantation}} | |||
| {{multiple image | |||
| | align = right | |||
| | image1 = Man before receiving hair transplant.jpg | |||
| | width1 = 150 | |||
| | alt1 = | |||
| | caption1 = | |||
| | image2 = Man after receiving hair transplant.jpg | |||
| | width2 = 130 | |||
| | alt2 = | |||
| | caption2 = | |||
| | footer = A man before and after a hair transplant. | |||
| }} | |||
| Hair transplantation is a ] technique that moves individual ]s from a part of the body called the donor site to bald or balding part of the body known as the recipient site. It is primarily used to treat male pattern baldness. In this condition, grafts containing hair follicles that are genetically resistant to balding are transplanted to bald scalp. It is also used to restore ]es, ]s, beard hair, chest hair, and pubic hair and to fill in scars caused by accidents or surgery such as face-lifts and previous hair transplants. Hair transplantation differs from ]ing in that grafts contain almost all of the ] and ] surrounding the hair follicle, and many tiny grafts are transplanted rather than a single strip of skin. | |||
| Since hair naturally grows in follicles in groups of 1 to 4 hairs, ]ation takes advantage of these naturally occurring follicular units. This achieves a more natural appearance by matching hair for hair through ] (FUT). | |||
| Through 2006, Curis and ] spent $1,000,000 on a hair growth program focused on the potential development of a topical ] ] for hair growth disorders such as male pattern baldness and female hair loss. Because the agent did not meet the proper safety standards.<ref>{{Cite news|author=Procter & Gamble|title=Curis and Procter & Gamble Enter into R&D Agreement for Hair Growth Program|url=http://www.pgpharma.com/news_20050919.shtml|year=2005|month=September 19|accessdate=August 24, 2006 |archiveurl = http://web.archive.org/web/20060822121850/http://www.pgpharma.com/news_20050919.shtml |archivedate = August 22, 2006}}</ref> <!-- Original link is dead: http://biz.yahoo.com/bw/060509/20060509005684.html?.v=1 -->, the hair loss research program was shut down in May 2007 | |||
| Donor hair can be harvested in two different ways. Small grafts of naturally-occurring units of one to four hairs, called ], can be moved to balding areas of the ]. These follicular units are surgically implanted in the scalp in very close proximity to one another and in large numbers. The grafts are obtained in one or both of the two primary methods of surgical extraction, follicular unit transplantation, colloquially referred to as "strip harvesting", or ] (FUE), in which follicles are transplanted individually. | |||
| ===Spin labels and Pyridine-N-oxides=== | |||
| Originally patented for treatment of pattern hair loss and used off-label for this disorder,<ref>], US Patent 5,352,442 Topical tempo]</ref> in animal models, the ] ]s ] and ] also enhance hair regrowth following radiation. There is currently a National Cancer Institute-sponsored clinical trial for TEMPOL in radiation alopecia.<ref></ref> Related pyridine-N-Oxides, such as nicotinic acid-N-oxide or "NANO" are also patented and used for hair loss treatment.<ref></ref> | |||
| In FUT, a strip of skin containing many follicular units is extracted from the patient and dissected under a stereoscopic microscope. The site of the strip removal is stitched closed. Once divided into follicular unit grafts, each unit is individually inserted into small recipient sites made by an incision in the bald scalp. In the newer technique, roots are extracted from the donor area and divided into strips for transplantation. The strip, two to three millimeters thick, is isolated and transplanted to the bald scalp.<ref>{{cite journal | vauthors = Rashid RM, Morgan Bicknell LT | title = Follicular unit extraction hair transplant automation: options in overcoming challenges of the latest technology in hair restoration with the goal of avoiding the line scar | journal = Dermatology Online Journal | volume = 18 | issue = 9 | pages = 12 | date = September 2012 | pmid = 23031379 | doi = 10.5070/D30X57S71R }}</ref> After surgery, a bandage is worn for two days to protect the stitched strip during healing. A small strip scar remains after healing, which can be covered by scalp hair growing over the scar.<ref name="pmid22808618"/> | |||
| ===Hair multiplication=== | |||
| ] and dermal papilla cells have been discovered in hair follicles. Research on these follicular cells may lead to successes in treating baldness through ''hair multiplication'' (HM), also known as ''hair cloning''. HM is being developed by ARI (Aderans Research Institute, a Japanese owned company in the USA).<ref name=webmd> | |||
| {{Cite web|url=http://my.webmd.com/content/article/96/103836.htm?z=3734_00000_1000_qd_01|accessdate=August 10, 2006|title=Hair Cloning Nears Reality as Baldness Cure|date=November 4, 2004|publisher=Webmd.com}}</ref><ref name=cbs2chicago>{{Cite news|url=http://cbs2chicago.com/health/health_story_075114234.html|title=Big Baldness Breakthrough?|accessdate=August 10, 2006|agency=Associated Press|date=March 15, 2004 |archiveurl = http://web.archive.org/web/20060525031410/http://cbs2chicago.com/health/health_story_075114234.html |archivedate = May 25, 2006}}</ref> | |||
| ====Scalp reduction==== | |||
| In 2008, ], a company in Manchester (UK), announced positive results of a ] trial for a form of ] ] from the back of the neck, ] them and then reimplanting the cells into the scalp. The initial testing resulted in at least two thirds of male patients regrowing hair. As of 2009, the company estimates this treatment will take "a number of years to complete" ] trials before it can go to market.<ref>{{Cite web|url=http://www.intercytex.com/icx/products/aesthetic/icxtrc/faqsicxtrc/|title=ICX-TRC – Frequently Asked Questions|publisher=Intercytex|date=March 22, 2010|accessdate=May 19, 2010}}</ref> However, after failing to achieve subsequent success in their trials, the company announced discontinuation of its hair multiplication project in January 2010 with intention to sell off its assets and research.<ref>{{Cite web|url=http://www.regrowhair.com/hair-loss-blog/intercytex-takes-a-dive-the-end-of-hair-multiplication-cloning-research/|title=Intercytex Discontinues its Hair Multiplication Development Operations | Hair Loss Q & A|publisher=Regrowhair.com|date=January 7, 2010|accessdate=May 19, 2010}}</ref> On March 25, 2010 Aderans Research Institute Inc. (ARI) announced it acquired key technology assets from Regenerative Medicine Assets Limited (formerly Intercytex Group plc).<ref>http://www.aderansresearch.com/pdfs/PR_ARI_US032510.pdf</ref> ARI is currently conducting Phase II clinical trials in cities throughout the U.S.<ref></ref> | |||
| {{main|Scalp reduction}} | |||
| Scalp reduction is a surgical procedure in which the hairless region of the scalp of a ] is reduced. This procedure can reduce the area of the scalp without hair.<ref>{{cite journal | vauthors = Bell ML | title = Role of scalp reduction in the treatment of male pattern baldness | journal = Plastic and Reconstructive Surgery | volume = 69 | issue = 2 | pages = 272–277 | date = February 1982 | pmid = 7054796 | doi = 10.1097/00006534-198202000-00016 | s2cid = 20731930 }}</ref><ref>{{cite book |last1=Unger |first1=Martin G. |last2=Toscani |first2=Marco |year=2016 |chapter=Scalp reduction |editor1-last=Scuderi |editor1-first=Nicolò |editor2-last=Toth |editor2-first=Bryant A. |title=International Textbook of Aesthetic Surgery |chapter-url=https://www.springer.com/gp/book/9783662465981 |publisher=] |pages=555–556 |doi=10.1007/978-3-662-46599-8 |isbn=978-3-662-46598-1 |access-date=7 December 2016 |archive-date=August 3, 2017 |archive-url=https://web.archive.org/web/20170803211713/http://www.springer.com/gp/book/9783662465981 |url-status=live }}</ref> | |||
| ==Regrowth of hair== | |||
| In December 2010, scientists at the ] in Germany revealed they had grown the world's first artificial hair follicles from ]. Research leader Dr Roland Lauster said within five years millions of hair-loss sufferers could grow new hair from their own stem cells and have it implanted into their bald spots. He also announced that preparations for clinical trials were "already in motion".<ref>{{cite news| url=http://www.dailymail.co.uk/health/article-1339485/Cure-baldness-horizon-scientists-grow-worlds-hair-follicle-using-stem-cells.html | location=London |work=Daily Mail | first=Claire | last=Bates | title=Cure for baldness on the horizon as scientists grow world's first hair follicles using stem cells | date=December 20, 2010}}</ref> | |||
| ===Facial hair=== | |||
| Replicel Life Sciences is another company pursuing an effective treatment for hairloss. In their first human clinical trial, Replicel was able to regenerate 19.6% percent of hair on one patient's treated area. Replicel is using a hair multiplication technique different from Aderans, using dermal sheath cup cells instead of dermal papilla cells. Replicel is looking forward for the Phase II at the end of 2012.<ref>http://www.replicel.com/wp-content/uploads/2012/RepliCel-AGM-Presentation-June-11-12.pdf</ref> | |||
| ====Eyebrows==== | |||
| In April 2012, a research group headed by Professor Takashi Tsuji demonstrated "functional hair regeneration from adult stem cells", a substantial advance in the development of the next generation of "organ replacement regenerative therapies". This procedure has only been tested on mice; human clinical trials have not been announced.<ref>http://www.sciencedaily.com/releases/2012/04/120418095011.htm</ref> | |||
| ] 0.03% has been used to grow ]s.<ref>{{cite journal |last1=Riahi |first1=RR |last2=Cohen |first2=PR |title=Topical Treatment of Eyebrow Hypotrichosis with Bimatoprost 0.03% Solution: Case Report and Literature Review. |journal=Cureus |date=21 May 2018 |volume=10 |issue=5 |pages=e2666 |doi=10.7759/cureus.2666 |doi-access=free |pmid=30042917 |pmc=6054329}}</ref> | |||
| ====Eyelashes==== | |||
| ===WNT protein introduction=== | |||
| The FP receptor agonist, ], in the form of an 0.03% ophthalmic solution termed ], is approved by the US ] to treat ] of the ]es, in particular to darken and lengthen eyelashes for cosmetic purposes.<ref>{{cite journal |last1=Choi |first1=YM |last2=Diehl |first2=J |last3=Levins |first3=PC |title=Promising alternative clinical uses of prostaglandin F2α analogs: beyond the eyelashes. |journal=Journal of the American Academy of Dermatology |date=April 2015 |volume=72 |issue=4 |pages=712–6 |doi=10.1016/j.jaad.2014.10.012 |pmid=25601618}}</ref> Also, bimatoprost may be used to treat small or underdeveloped ]es.<ref name="Bimatoprost label">{{cite web | title=Bimatoprost solution/ drops | website=DailyMed | date=18 September 2019 | url=https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=27bef7e1-750a-4ac1-ab5f-e4c0121ffcbc | access-date=4 January 2020 | archive-date=September 21, 2020 | archive-url=https://web.archive.org/web/20200921171858/https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=27bef7e1-750a-4ac1-ab5f-e4c0121ffcbc | url-status=live }}</ref><ref name="Lumigan label">{{cite web | title=Lumigan- bimatoprost solution/ drops | website=DailyMed | date=31 July 2017 | url=https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a5e67c75-db88-4372-bb07-c8dd15c97631 | access-date=4 January 2020 | archive-date=September 21, 2020 | archive-url=https://web.archive.org/web/20200921171612/https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a5e67c75-db88-4372-bb07-c8dd15c97631 | url-status=live }}</ref> | |||
| In May 2007, U.S. company Follica Inc, announced they had licensed technology from the ] which can regenerate ] by reawakening ] which were once active only in the ] stage of human development. Skin apparently can be brought back to this embryonic state when a wound is healing. Hair growth was discovered in the skin wounds of mice when ]s were introduced to the site. Development of a human treatment is expected to take several years.<ref></ref><ref></ref> | |||
| ===Scalp hair=== | |||
| In 2011, the natural oral minoxidil mimic ] was released on the market. Similar to minoxidil, its mechanism of action may involve the opening of potassium channels.<ref>{{cite news|last=Tyler|first=Richard|title=Thomas Whitfield's German roots help hair loss product launch|url=http://www.telegraph.co.uk/finance/yourbusiness/8248046/Thomas-Whitfields-German-roots-help-hair-loss-product-launch.html|accessdate=July 23, 2012|newspaper=The Daily Telegraph|date=January 9, 2011}}</ref> The product is a dietary supplement, not a drug, and hence is not FDA-approved.<ref name=CBS>{{cite news|last=Edwards|first=Jim|title=Pharma's 4 Best Shots at a Cure for Baldness|url=http://www.cbsnews.com/8301-505123_162-42847047/pharmas-4-best-shots-at-a-cure-for-baldness/|accessdate=August 1, 2012|newspaper=CBSNews.com|date=January 12, 2011|agency=CBS News|format=Web|quote=it's actually just another dietary supplement and as such is not approved by the FDA.}}</ref><ref>{{cite web|title=Minoxidil Alternatives|url=http://www.hairloss-research.org/UpdateMinoxidilAlternatives6-11.html|publisher=MPB Research|accessdate=August 1, 2012|format=Web}}</ref> | |||
| === |
====Alternative medication==== | ||
| In 2012, a Korean scientific study showed that , a red seaweed native to ], South Korea, has the potential to treat androgenic alopecia as well as alopecia areata.<ref></ref><ref>Nguyen, H., Kim, S.M. (2012). . ''African Journal of Biotechnology'', 11(39), pp. 9457–9467 (May 15). Retrieved on 2012-06-14.</ref> | |||
| === |
=====Topical===== | ||
| Some popular plant juices sold as hair serum may instead of growing human hair actually inhibit the growth, including ] found in ].<ref>{{cite journal | vauthors = Miao Y, Sun Y, Wang W, Du B, Xiao SE, Hu Y, Hu Z | title = 6-Gingerol inhibits hair shaft growth in cultured human hair follicles and modulates hair growth in mice | journal = PLOS ONE | volume = 8 | issue = 2 | pages = e57226 | date = 2013 | pmid = 23437345 | pmc = 3578824 | doi = 10.1371/journal.pone.0057226 | doi-access = free | bibcode = 2013PLoSO...857226M }}</ref> | |||
| In August 2012, Scientists at the ] announced that they had discovered an ] which caused baldness. They found that the lipid ] (]) was found to be present on the ] of balding men at far higher levels than normal, preventing ] from maturing and therefore stopping them from working and growing hair. Dr. George Cotsarelis and his dermatological team at the University say that they are in talks with several ] about developing treatments which could be available in two years.<ref>http://www.dailymail.co.uk/news/article-2190544/Baldness-cure-reverses-genetics-market-years.html</ref><ref>http://www.telegraph.co.uk/health/healthnews/9485807/Baldness-cure-could-be-on-shelves-in-two-years.html</ref><ref>http://www.nytimes.com/2012/07/29/business/baldness-battle-fought-in-the-follicle.html</ref> | |||
| =====Topical crude onion juice===== | |||
| ===L-Carnitine-Tartrate=== | |||
| ] | |||
| A study performed by German scientists showed that ''L-Carntine-tartrate'' induces hair growth in humans. ], a product released in 2011 by Oxford scientists, lists ''L-Carnitine-Tartrate'' as its main ingredient.<ref name=CBS>{{cite news|last=Edwards|first=Jim|title=Pharma's 4 Best Shots at a Cure for Baldness|url=http://www.cbsnews.com/8301-505123_162-42847047/pharmas-4-best-shots-at-a-cure-for-baldness/|accessdate=August 1, 2012|newspaper=CBSNews.com|date=January 12, 2011|agency=CBS News|format=Web}}</ref><ref>{{cite journal|title=L-Carnitine–L-tartrate promotes human hair growth in vitro.|journal=Experimental Dermatology|year=2007|month=November|volume=16: 936–945.|issue=11|pages=936–945|doi=10.1111/j.1600-0625.2007.00611.x|accessdate=August 4, 2012|author=Foitzik, K., Hoting, E., Förster, T., Pertile, P. and Paus, R.|format=Web & Print|ref=http://onlinelibrary.wiley.com/doi/10.1111/j.1600-0625.2007.00611.x/abstract;jsessionid=B810F332D32BB2A101A026621BF1A3DB.d01t01}}</ref> | |||
| A small 2002 study demonstrated that treatment twice daily for six weeks with crude onion juice from Australian brown onion, re-growth hair on ] (spot baldness) in 86.9% of the 23 participants.<ref name="Crude onion juice from Australian brown onion">{{cite journal | vauthors = Sharquie KE, Al-Obaidi HK | title = Onion juice (Allium cepa L.), a new topical treatment for alopecia areata | journal = The Journal of Dermatology | volume = 29 | issue = 6 | pages = 343–346 | date = June 2002 | pmid = 12126069 | doi = 10.1111/j.1346-8138.2002.tb00277.x | s2cid = 24116769 }}</ref> Twice as many ] are found in ] than in ].<ref name="Onions: a source of unique dietary">{{cite journal | vauthors = Slimestad R, Fossen T, Vågen IM | title = Onions: a source of unique dietary flavonoids | journal = Journal of Agricultural and Food Chemistry | volume = 55 | issue = 25 | pages = 10067–10080 | date = December 2007 | pmid = 17997520 | doi = 10.1021/jf0712503 }}</ref> Also, non-organic onions might contain pesticides on the peel and in the first scaly leaf. Compounds found in onion that stimulates hair growth: | |||
| ==References== | |||
| * ]: | |||
| {{Reflist|2}} | |||
| **] (a flavanoid found in vegan food) supplements, has been suggested to treat baldness.<ref name="Quercetin" /> A 2012 study demonstrated that alopecia areata could be used to prevent ant treated with quercetin in mice.<ref name="Quercetin">{{cite journal | vauthors = Wikramanayake TC, Villasante AC, Mauro LM, Perez CI, Schachner LA, Jimenez JJ | title = Prevention and treatment of alopecia areata with quercetin in the C3H/HeJ mouse model | journal = Cell Stress & Chaperones | volume = 17 | issue = 2 | pages = 267–274 | date = March 2012 | pmid = 22042611 | pmc = 3273564 | doi = 10.1007/s12192-011-0305-3 }}</ref> Quercetin is found in onions primarily in the peel and the first scaly leaf but not in the flesh.<ref>{{cite journal | vauthors = Kwak JH, Seo JM, Kim NH, Arasu MV, Kim S, Yoon MK, Kim SJ | title = Variation of quercetin glycoside derivatives in three onion (''Allium cepa'' L.) varieties | journal = Saudi Journal of Biological Sciences | volume = 24 | issue = 6 | pages = 1387–1391 | date = September 2017 | pmid = 28855836 | pmc = 5562462 | doi = 10.1016/j.sjbs.2016.05.014 }}</ref> Onion ] or weight does not appear to be affected by quercetin concentration.<ref name="Onions: a source of unique dietary"/> | |||
| ** A 2020 study demonstrated that quercitrin stimulated hair shaft growth in cultured human hair follicles.<ref>{{cite journal | vauthors = Kim J, Kim SR, Choi YH, Shin JY, Kim CD, Kang NG, Park BC, Lee S | display-authors = 6 | title = Quercitrin Stimulates Hair Growth with Enhanced Expression of Growth Factors via Activation of MAPK/CREB Signaling Pathway | journal = Molecules | volume = 25 | issue = 17 | page = 4004 | date = September 2020 | pmid = 32887384 | pmc = 7504764 | doi = 10.3390/molecules25174004 | doi-access = free }}</ref> | |||
| * Volatile compounds responsible for pungency and tearing in onions when they are cut (e.g. ] gas). However, no formal studies have conducted to evaluate if these compounds promote hair growth or if they help other compounds to penetrate the skin to form hair follicles. | |||
| ** To use the volatile compounds: Onions must be extracted and applied topically quickly before the volatile compounds evaporates. The extract will cause tearing eyes, but it can be prevented with a ], or a pair of swimming goggles. | |||
| ** To discard the volatile compounds: The onion juice is extracted and stored for a short while to evaporate the volatile compounds. The extract will not cause tearing eyes. Also, both ], and ] onions have been produced with significantly reduced levels of tear-inducing lachrymatory factor.<ref>{{cite journal | vauthors = Eady CC, Kamoi T, Kato M, Porter NG, Davis S, Shaw M, Kamoi A, Imai S | display-authors = 6 | title = Silencing onion lachrymatory factor synthase causes a significant change in the sulfur secondary metabolite profile | journal = Plant Physiology | volume = 147 | issue = 4 | pages = 2096–2106 | date = August 2008 | pmid = 18583530 | pmc = 2492635 | doi = 10.1104/pp.108.123273 }}</ref><ref>{{cite news |title=Cutting this new type of onion won't make you cry |url=https://www.independent.co.uk/life-style/onion-no-crying-cut-up-tears-sting-juice-a8149821.html |work=The Independent |date=9 January 2018 |language=en |access-date=September 7, 2021 |archive-date=September 7, 2021 |archive-url=https://web.archive.org/web/20210907223821/https://www.independent.co.uk/life-style/onion-no-crying-cut-up-tears-sting-juice-a8149821.html |url-status=live }}</ref> | |||
| =====Dietary supplements===== | |||
| ==External links== | |||
| Dietary supplements are not typically recommended.<ref name=Roger2008>{{cite journal | vauthors = Rogers NE, Avram MR | title = Medical treatments for male and female pattern hair loss | journal = Journal of the American Academy of Dermatology | volume = 59 | issue = 4 | pages = 547–66; quiz 567–8 | date = October 2008 | pmid = 18793935 | doi = 10.1016/j.jaad.2008.07.001 }}</ref> Many people use unproven treatments,<ref name=Ban2013>{{cite journal | vauthors = Banka N, Bunagan MJ, Shapiro J | title = Pattern hair loss in men: diagnosis and medical treatment | journal = Dermatologic Clinics | volume = 31 | issue = 1 | pages = 129–140 | date = January 2013 | pmid = 23159182 | doi = 10.1016/j.det.2012.08.003 }}</ref> but there is little evidence of the effectiveness of ]s, minerals, or other dietary supplements regrowing hair or retaining hair.<ref name=Levy2013/> There is no evidence for ] (vitamin B7).<ref name=Roger2008/> While lacking both evidence and expert recommendation, there is a large market for hair growth supplements, especially for products that contain biotin. | |||
| * | |||
| * | |||
| * | |||
| * , April 1999, ''American Family Physician'' (medical journal) | |||
| * zinc, silica, methylsulphonylmethane (MSM) and cod-liver oil, to slow down the process. | |||
| However, one small trial of ] which shows tentative benefit in those with mild to moderate androgenetic alopecia.<ref name=Roger2008/> There was no good evidence for ], ], ], ], or ] as of 2011.<ref name=Blu2011>{{cite journal | vauthors = Blumeyer A, Tosti A, Messenger A, Reygagne P, Del Marmol V, Spuls PI, Trakatelli M, Finner A, Kiesewetter F, Trüeb R, Rzany B, Blume-Peytavi U | display-authors = 6 | title = Evidence-based (S3) guideline for the treatment of androgenetic alopecia in women and in men | journal = Journal der Deutschen Dermatologischen Gesellschaft | volume = 9 | issue = Suppl 6 | pages = S1-57 | date = October 2011 | pmid = 21980982 | doi = 10.1111/j.1610-0379.2011.07802.x | s2cid = 29821046 }}</ref> | |||
| ==Radiation-induced hair loss== | |||
| Radiation induces hair loss through damage to hair follicle stem cell progenitors and alteration of keratin expression.<ref>{{cite journal | vauthors = Nanashima N, Ito K, Ishikawa T, Nakano M, Nakamura T | title = Damage of hair follicle stem cells and alteration of keratin expression in external radiation-induced acute alopecia | journal = International Journal of Molecular Medicine | volume = 30 | issue = 3 | pages = 579–584 | date = September 2012 | pmid = 22692500 | doi = 10.3892/ijmm.2012.1018 | doi-access = free }}</ref><ref>{{cite journal | vauthors = Kamiya K, Sasatani M | title = | journal = Nihon Rinsho. Japanese Journal of Clinical Medicine | volume = 70 | issue = 3 | pages = 367–374 | date = March 2012 | pmid = 22514910 }}</ref> Radiation therapy has been associated with increased ] production in hair follicles.<ref>{{cite journal | vauthors = Takeda H, Nakajima K, Kaneko T, Harada K, Matsuzaki Y, Sawamura D | title = Follicular mucinosis associated with radiation therapy | journal = The Journal of Dermatology | volume = 38 | issue = 11 | pages = 1116–1118 | date = November 2011 | pmid = 22034994 | doi = 10.1111/j.1346-8138.2010.01187.x | s2cid = 31876890 }}</ref> | |||
| Studies have suggested electromagnetic radiation as a therapeutic growth stimulant in alopecia.<ref>{{cite journal | vauthors = Kalia S, Lui H | title = Utilizing electromagnetic radiation for hair growth: a critical review of phototrichogenesis | journal = Dermatologic Clinics | volume = 31 | issue = 1 | pages = 193–200 | date = January 2013 | pmid = 23159188 | doi = 10.1016/j.det.2012.08.018 }}</ref> | |||
| ==Cosmeses== | |||
| ] in ] design.]] Certain hair shampoos and ointments visually thicken existing hair, without affecting the growth cycle.<ref>{{cite journal | vauthors = Davis MG, Thomas JH, van de Velde S, Boissy Y, Dawson TL, Iveson R, Sutton K | title = A novel cosmetic approach to treat thinning hair | journal = The British Journal of Dermatology | volume = 165 | issue = Suppl 3 | pages = 24–30 | date = December 2011 | pmid = 22171682 | doi = 10.1111/j.1365-2133.2011.10633.x | s2cid = 21918537 }}</ref> There have also been developments in the fashion industry with ] design. The fashion accessory has also been shown to be a source of psychological support for women undergoing chemotherapy, with cancer survivors in one study describing their wig as a "constant companion".<ref>{{cite journal | vauthors = Zannini L, Verderame F, Cucchiara G, Zinna B, Alba A, Ferrara M | title = 'My wig has been my journey's companion': perceived effects of an aesthetic care programme for Italian women suffering from chemotherapy-induced alopecia | journal = European Journal of Cancer Care | volume = 21 | issue = 5 | pages = 650–660 | date = September 2012 | pmid = 22339814 | doi = 10.1111/j.1365-2354.2012.01337.x | doi-access = free }}</ref> Other studies in women have demonstrated a more mixed psychosocial impact of hairpiece use.<ref>{{cite journal | vauthors = Inui S, Inoue T, Itami S | title = Psychosocial impact of wigs or hairpieces on perceived quality of life level in female patients with alopecia areata | journal = The Journal of Dermatology | volume = 40 | issue = 3 | pages = 225–226 | date = March 2013 | pmid = 23252418 | doi = 10.1111/1346-8138.12040 | s2cid = 43795003 }}</ref> | |||
| Specialized tattoos, commonly known as scalp micropigmentation, can mimic the appearance of a short buzzed haircut.<ref>{{cite news |title=Considering a hair tattoo? Pros and cons to consider before you commit |author=Elisabeth Leamy |url=https://abcnews.go.com/blogs/health/2012/05/31/considering-a-hair-tattoo-pros-and-cons-to-consider-before-you-commit/ |newspaper=ABC News |date=May 31, 2012 |access-date=December 16, 2012 |archive-date=April 11, 2019 |archive-url=https://web.archive.org/web/20190411205715/https://abcnews.go.com/blogs/health/2012/05/31/considering-a-hair-tattoo-pros-and-cons-to-consider-before-you-commit/ |url-status=live }}</ref> | |||
| ==Human hair growth== | |||
| {{main|Human hair growth}} | |||
| ] with mesenchymal ], labelled at top, location of hair follicle ]s and thought to be site of action of ].]] | |||
| ] are present at ] in papillae of individual ]s. They catalyse formation of the androgens testosterone and DHT, which in turn regulate hair growth. Androgens have different effects at different follicles: they stimulate ] at facial hair, causing hair regrowth, but stimulate ], ], ] and ] at the scalp, causing hair follicle miniaturisation.<ref name=Inui2013>{{cite journal | vauthors = Inui S, Itami S | title = Androgen actions on the human hair follicle: perspectives | journal = Experimental Dermatology | volume = 22 | issue = 3 | pages = 168–171 | date = March 2013 | pmid = 23016593 | doi = 10.1111/exd.12024 | s2cid = 33521841 | doi-access = free }}</ref> | |||
| Female androgenic alopecia is characterized by diffuse crown thinning without hairline recession, and like its male counterpart rarely leads to ].<ref name="Female pattern baldness"/> ] and ] are usually first line therapy for its treatment. Other options include topical or systemic ] or ], although they have a high incidence of ] ] and are better tolerated in female androgenic hair loss. | |||
| More advanced cases may be resistant or unresponsive to medical therapy, however, and require ]. Naturally-occurring units of one to four hairs, called ], are excised and moved to areas of hair restoration. These follicular units are surgically implanted in the scalp in close proximity and in large numbers. The grafts are obtained from either ] (FUT) – colloquially referred to as "strip harvesting" – or ] (FUE). In the former, a strip of skin with follicular units is extracted and dissected into individual follicular unit grafts. The surgeon then implants the grafts into small incisions, called recipient sites.<ref name="pmid22808618">{{cite journal | vauthors = Caroli S, Pathomvanich D, Amonpattana K, Kumar A | title = Current status of hair restoration surgery | journal = International Surgery | volume = 96 | issue = 4 | pages = 345–351 | year = 2011 | pmid = 22808618 | doi = 10.9738/cc31.1 | doi-access = free }}</ref><ref name="pmid 21792780">{{cite journal | vauthors = Rose PT | title = The latest innovations in hair transplantation | journal = Facial Plastic Surgery | volume = 27 | issue = 4 | pages = 366–377 | date = August 2011 | pmid = 21792780 | doi = 10.1055/s-0031-1283055 | s2cid = 260138453 }}</ref> Specialized scalp tattoos can also mimic the appearance of a short buzzed haircut.<ref>{{cite news |title=Considering a hair tattoo? Pros and cons to consider before you commit |author=Elisabeth Leamy |url=https://abcnews.go.com/blogs/health/2012/05/31/considering-a-hair-tattoo-pros-and-cons-to-consider-before-you-commit/ |newspaper=ABC News |date=May 31, 2012 |access-date=December 16, 2012 |author-link=Elisabeth Leamy |archive-date=April 11, 2019 |archive-url=https://web.archive.org/web/20190411205715/https://abcnews.go.com/blogs/health/2012/05/31/considering-a-hair-tattoo-pros-and-cons-to-consider-before-you-commit/ |url-status=live }}</ref> Androgenic alopecia also occurs in women, and more often presents as diffuse thinning without hairline recession. Like its male counterpart, the condition rarely leads to ]. Treatment options are similar to those for men, although topical or systemic estrogen is used more often.<ref name="Female pattern baldness">{{cite web |url=https://www.nlm.nih.gov/medlineplus/ency/article/001173.htm |title=Female pattern baldness |access-date=December 15, 2012 |date=December 15, 2012 |publisher=MedlinePlus |archive-date=December 1, 2012 |archive-url=https://web.archive.org/web/20121201014315/http://www.nlm.nih.gov/medlineplus/ency/article/001173.htm |url-status=live }}</ref><ref>{{cite journal | vauthors = Kovalevsky G, Ballagh SA, Stanczyk FZ, Lee J, Cooper J, Archer DF | title = Levonorgestrel effects on serum androgens, sex hormone-binding globulin levels, hair shaft diameter, and sexual function | journal = Fertility and Sterility | volume = 93 | issue = 6 | pages = 1997–2003 | date = April 2010 | pmid = 19394598 | doi = 10.1016/j.fertnstert.2008.12.095 | doi-access = free }}</ref> | |||
| ==Research== | |||
| ===Bimatoprost and latanoprost=== | |||
| ] and ] are specific PGF2a analogues applied topically, and have been found to lengthen eyelashes,<ref>{{cite journal | vauthors = Law SK | title = Bimatoprost in the treatment of eyelash hypotrichosis | journal = Clinical Ophthalmology | volume = 4 | pages = 349–358 | date = April 2010 | pmid = 20463804 | pmc = 2861943 | doi = 10.2147/opth.s6480 | doi-access = free }}</ref><ref>{{cite journal | vauthors = Tosti A, Pazzaglia M, Voudouris S, Tosti G | title = Hypertrichosis of the eyelashes caused by bimatoprost | journal = Journal of the American Academy of Dermatology | volume = 51 | issue = 5 Suppl | pages = S149–S150 | date = November 2004 | pmid = 15577756 | doi = 10.1016/j.jaad.2004.05.002 }}</ref> darken hair pigmentation<ref>{{cite journal | vauthors = Wand M | title = Latanoprost and hyperpigmentation of eyelashes | journal = Archives of Ophthalmology | volume = 115 | issue = 9 | pages = 1206–1208 | date = September 1997 | pmid = 9298071 | doi = 10.1001/archopht.1997.01100160376025 }}</ref> and elongate hair.<ref name="pmid22735503"/> Bimatoprost is available as treatment for eyelash growth.<ref>{{cite journal | vauthors = Banaszek A | title = Company profits from side effects of glaucoma treatment | journal = CMAJ | volume = 183 | issue = 14 | pages = E1058 | date = October 2011 | pmid = 21876012 | pmc = 3185096 | doi = 10.1503/cmaj.109-3919 }}</ref> Latanoprost has shown ability to promote scalp hair density and pigmentation,<ref>{{cite journal | vauthors = Blume-Peytavi U, Lönnfors S, Hillmann K, Garcia Bartels N | title = A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair growth and pigmentation in healthy volunteers with androgenetic alopecia | journal = Journal of the American Academy of Dermatology | volume = 66 | issue = 5 | pages = 794–800 | date = May 2012 | pmid = 21875758 | doi = 10.1016/j.jaad.2011.05.026 }}</ref> and is theorized to function at the ].<ref>{{cite journal | vauthors = Johnstone MA, Albert DM | title = Prostaglandin-induced hair growth | journal = Survey of Ophthalmology | volume = 47 | issue = Suppl 1 | pages = S185–S202 | date = August 2002 | pmid = 12204716 | doi = 10.1016/s0039-6257(02)00307-7 }}</ref> A study found latanoprost ineffective on eyelashes in a patient with ].<ref>{{cite journal | vauthors = Roseborough I, Lee H, Chwalek J, Stamper RL, Price VH | title = Lack of efficacy of topical latanoprost and bimatoprost ophthalmic solutions in promoting eyelash growth in patients with alopecia areata | journal = Journal of the American Academy of Dermatology | volume = 60 | issue = 4 | pages = 705–706 | date = April 2009 | pmid = 19293023 | doi = 10.1016/j.jaad.2008.08.029 }}</ref> It has also been found ineffective in treatment of eyebrow hair loss.<ref>{{cite journal | vauthors = Ross EK, Bolduc C, Lui H, Shapiro J | title = Lack of efficacy of topical latanoprost in the treatment of eyebrow alopecia areata | journal = Journal of the American Academy of Dermatology | volume = 53 | issue = 6 | pages = 1095–1096 | date = December 2005 | pmid = 16310083 | doi = 10.1016/j.jaad.2005.06.031 | doi-access = free }}</ref> | |||
| ===Hormones=== | |||
| Estrogens are indirect anti-androgens and can be used to treat androgenetic hair loss in women with ]. Systemic estrogen increases SHBG, which binds androgens, including testosterone and DHT, in turn reducing their bioavailability. Topical formulations are available in ].<ref name=Blu2011 /> Hair follicles have estrogen receptors and it is theorized that topical compounds act on them directly to promote hair growth and antagonize androgen action. Large clinical studies showing effectiveness are absent. Topical treatment is also usually unavailable in ].<ref name="pmid22735503"/> | |||
| There is tentative evidence for ] in women.<ref name=Levy2013/> | |||
| ===IGF-1=== | |||
| In December 2012, topical application of ] in a liposomal vehicle led to thicker and more rapid hair growth in transgenic mice with androgenic alopecia. The study did not show measurable systemic levels or hematopoietic side effects, suggesting potential for use in humans.<ref>{{cite journal | vauthors = Castro RF, Azzalis LA, Feder D, Perazzo FF, Pereira EC, Junqueira VB, Rocha KC, Machado CD, Paschoal FC, Gnann LA, Fonseca FL | display-authors = 6 | title = Safety and efficacy analysis of liposomal insulin-like growth factor-1 in a fluid gel formulation for hair-loss treatment in a hamster model | journal = Clinical and Experimental Dermatology | volume = 37 | issue = 8 | pages = 909–912 | date = December 2012 | pmid = 22924775 | doi = 10.1111/j.1365-2230.2012.04441.x | s2cid = 21021986 }}</ref> Low energy radiofrequency irradiation induces IGF-1 in cultured human dermal papilla cells.<ref name="pmid22164296">{{cite journal | vauthors = Yoon SY, Kim KT, Jo SJ, Cho AR, Jeon SI, Choi HD, Kim KH, Park GS, Pack JK, Kwon OS, Park WY | display-authors = 6 | title = Induction of hair growth by insulin-like growth factor-1 in 1,763 MHz radiofrequency-irradiated hair follicle cells | journal = PLOS ONE | volume = 6 | issue = 12 | pages = e28474 | year = 2011 | pmid = 22164296 | pmc = 3229574 | doi = 10.1371/journal.pone.0028474 | editor1-last = Najbauer | doi-access = free | bibcode = 2011PLoSO...628474Y | editor1-first = Joseph }}</ref> Adenosine stimulates dermal papillae in vitro to induce IGF-1, along with fibroblast growth factors ], ] and ]. ] transcription increased, which promotes dermal papillae as well.<ref name="pmid22164296"/> Dietary ] increase IGF production in scalp ] in transgenic mice.<ref>{{cite journal | vauthors = Zhao J, Harada N, Kurihara H, Nakagata N, Okajima K | title = Dietary isoflavone increases insulin-like growth factor-I production, thereby promoting hair growth in mice | journal = The Journal of Nutritional Biochemistry | volume = 22 | issue = 3 | pages = 227–233 | date = March 2011 | pmid = 20576422 | doi = 10.1016/j.jnutbio.2010.01.008 }}</ref> Topical capsaicin also stimulates IGF at hair follicles via release of vanilloid receptor-1, which in turn leads to more CGRP.<ref>{{cite journal | vauthors = Okajima K, Harada N | title = Promotion of insulin-like growth factor-I production by sensory neuron stimulation; molecular mechanism(s) and therapeutic implications | journal = Current Medicinal Chemistry | volume = 15 | issue = 29 | pages = 3095–3112 | year = 2008 | pmid = 19075656 | doi = 10.2174/092986708786848604 }}</ref> Ascorbic acid has led to increased IGF expression ''in vitro''.<ref>{{cite journal | vauthors = Kwack MH, Shin SH, Kim SR, Im SU, Han IS, Kim MK, Kim JC, Sung YK | display-authors = 6 | title = l-Ascorbic acid 2-phosphate promotes elongation of hair shafts via the secretion of insulin-like growth factor-1 from dermal papilla cells through phosphatidylinositol 3-kinase | journal = The British Journal of Dermatology | volume = 160 | issue = 6 | pages = 1157–1162 | date = June 2009 | pmid = 19416266 | doi = 10.1111/j.1365-2133.2009.09108.x | s2cid = 42210965 }}</ref> | |||
| ===Stem cell therapy=== | |||
| {{Main|Hair cloning}} | |||
| Although follicles were previously thought gone in areas of complete baldness, they are more likely dormant, as recent studies have shown the scalp contains the ]s from which the follicles arose.<ref>{{cite journal | vauthors = Garza LA, Yang CC, Zhao T, Blatt HB, Lee M, He H, Stanton DC, Carrasco L, Spiegel JH, Tobias JW, Cotsarelis G | display-authors = 6 | title = Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells | journal = The Journal of Clinical Investigation | volume = 121 | issue = 2 | pages = 613–622 | date = February 2011 | pmid = 21206086 | pmc = 3026732 | doi = 10.1172/JCI44478 }}</ref> Research on these follicular stem cells may lead to successes in treating baldness through ''hair multiplication'' (HM), also known as '']''. | |||
| Per a May 2015 review, no successful strategy to generate human hair follicles, for hair regrowth, from adult stem cells has yet been reported.<ref>{{cite journal | vauthors = Balañá ME, Charreau HE, Leirós GJ | title = Epidermal stem cells and skin tissue engineering in hair follicle regeneration | journal = World Journal of Stem Cells | volume = 7 | issue = 4 | pages = 711–727 | date = May 2015 | pmid = 26029343 | pmc = 4444612 | doi = 10.4252/wjsc.v7.i4.711 | doi-access = free }}</ref> However, in April 2016, scientists from Japan published results of their work in which they created human skin from ]s; implanted into laboratory mice, the cells generated skin with hair and glands.<ref>{{cite journal | vauthors = Takagi R, Ishimaru J, Sugawara A, Toyoshima KE, Ishida K, Ogawa M, Sakakibara K, Asakawa K, Kashiwakura A, Oshima M, Minamide R, Sato A, Yoshitake T, Takeda A, Egusa H, Tsuji T | display-authors = 6 | title = Bioengineering a 3D integumentary organ system from iPS cells using an in vivo transplantation model | journal = Science Advances | volume = 2 | issue = 4 | pages = e1500887 | date = April 2016 | pmid = 27051874 | pmc = 4820374 | doi = 10.1126/sciadv.1500887 | bibcode = 2016SciA....2E0887T }}</ref> | |||
| ===Genetics=== | |||
| From 2005 to 2007, Curis and ] collaborated on developing a topical ] ] for hair loss; the agent did not meet safety standards, and the program was terminated.<ref>{{cite news|last1=Weintraub|first1=Arlene|title=Curis and Genentech Unleash the Hedgehog to Fight a Deadly Skin Cancer|url=http://www.xconomy.com/boston/2011/06/28/curis-and-genentech-unleash-the-hedgehog-to-fight-a-deadly-skin-cancer/?single_page=true|work=Xconomy|date=28 June 2011|access-date=November 9, 2016|archive-date=November 9, 2016|archive-url=https://web.archive.org/web/20161109221622/http://www.xconomy.com/boston/2011/06/28/curis-and-genentech-unleash-the-hedgehog-to-fight-a-deadly-skin-cancer/?single_page=true|url-status=live}}</ref><ref>{{cite web|title=Curis Form 8-K: Termination of a Material Definitive Agreement.|url=https://www.sec.gov/Archives/edgar/data/1108205/000119312507109693/d8k.htm|publisher=SEC EDGAR|date=May 10, 2007|access-date=November 9, 2016|archive-date=November 9, 2016|archive-url=https://web.archive.org/web/20161109222303/https://www.sec.gov/Archives/edgar/data/1108205/000119312507109693/d8k.htm|url-status=live}}</ref> In 2008, researchers at the ] announced they have found the genetic basis of two distinct forms of inherited hair loss. They found the gene ] causes a rare, inherited form of hair loss called hypotrichosis simplex. It is the first ] in humans known to play a role in hair growth.<ref>{{cite journal | vauthors = Pasternack SM, von Kügelgen I, Al Aboud K, Lee YA, Rüschendorf F, Voss K, Hillmer AM, Molderings GJ, Franz T, Ramirez A, Nürnberg P, Nöthen MM, Betz RC | display-authors = 6 | title = G protein-coupled receptor P2Y5 and its ligand LPA are involved in maintenance of human hair growth | journal = Nature Genetics | volume = 40 | issue = 3 | pages = 329–334 | date = March 2008 | pmid = 18297070 | doi = 10.1038/ng.84 | s2cid = 20241237 }}</ref><ref>{{cite journal | vauthors = Shimomura Y, Wajid M, Ishii Y, Shapiro L, Petukhova L, Gordon D, Christiano AM | title = Disruption of P2RY5, an orphan G protein-coupled receptor, underlies autosomal recessive woolly hair | journal = Nature Genetics | volume = 40 | issue = 3 | pages = 335–339 | date = March 2008 | pmid = 18297072 | doi = 10.1038/ng.100 | s2cid = 1635950 }}</ref><ref>{{cite journal | vauthors = Sprecher E | title = Disentangling the roots of inherited hair disorders | journal = Nature Genetics | volume = 40 | issue = 3 | pages = 265–266 | date = March 2008 | pmid = 18305473 | doi = 10.1038/ng0308-265 | s2cid = 2311629 }}</ref> Researchers found that disruption of the ] ] in mice caused cyclical hair loss. Research has suggested SOX21 as a master regulator of hair shaft cuticle differentiation, with its disruption causing cyclical alopecia in mice models.<ref>{{cite journal | vauthors = Kiso M, Tanaka S, Saba R, Matsuda S, Shimizu A, Ohyama M, Okano HJ, Shiroishi T, Okano H, Saga Y | display-authors = 6 | title = The disruption of Sox21-mediated hair shaft cuticle differentiation causes cyclic alopecia in mice | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 106 | issue = 23 | pages = 9292–9297 | date = June 2009 | pmid = 19470461 | pmc = 2695080 | doi = 10.1073/pnas.0808324106 | doi-access = free | bibcode = 2009PNAS..106.9292K }}</ref> Deletion of SOX21 dramatically affects hair lipids.<ref>{{cite journal | vauthors = Kawaminami S, Breakspear S, Saga Y, Noecker B, Masukawa Y, Tsuchiya M, Oguri M, Inoue Y, Ishikawa K, Okamoto M | display-authors = 6 | title = Deletion of the Sox21 gene drastically affects hair lipids | journal = Experimental Dermatology | volume = 21 | issue = 12 | pages = 974–976 | date = December 2012 | pmid = 23171466 | doi = 10.1111/exd.12050 | s2cid = 205126099 | doi-access = free }}</ref> | |||
| == References == | |||
| {{Reflist|30em}} | |||
| == External links == | |||
| * , April 1999, ''American Family Physician'' (medical journal) | |||
| {{Human hair}} | |||
| {{Other dermatological preparations}} | {{Other dermatological preparations}} | ||
| {{DEFAULTSORT:Baldness Treatments}} | <!--?: {{DEFAULTSORT:Baldness Treatments}}--> | ||
| ] | |||
| ] | |||
| ] | |||
| ] | |||
| ] | |||
Latest revision as of 18:52, 24 February 2024
Medical intervention
| Management of hair loss | |
|---|---|
| Specialty | Dermatology |
| [edit on Wikidata] | |
The management of hair loss, includes prevention and treatment of alopecia, baldness, and hair thinning, and regrowth of hair.
Prevention
Scratching of itchy scalp may contribute to hair loss.
Health
Body weight
Prevention of obesity helps. Obesity is linked to hair thinning.
Healthy diet
See also: Healthy dietOils with fatty acids that has been studied to prevent dermatitis includes:
- Corn oil: Linoleic acid (LA)
- Fish oil: Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)
- Hemp seed oil: Linoleic acid (LA), and alpha-Linolenic acid (ALA)
Alternative medicine
The scalp must be cleaned from sebum, sweat, and dirt, prior to topical application, for agents to penetrate it.
A 2020 systemic review on agents used to treat androgenic alopecia found that:
- Oral supplementation of vitamin A, B, C, D, E, and trace element iron, selenium, and zinc, will prevent androgenic alopecia caused by malnutrition. Multivitamins can be used.
- Topical application of onion juice, rosemary oil, saw palmetto, pumpkin seed oil, procyanidin, garlic gel, capsaicin, caffeine, amino acids, and curcumin helped prevent hair loss.
Heating, ventilation, and air conditioning (HVAC)
Humidifier
A humidifier can be used to prevent low indoor humidity during winter (especially with indoor heating), and dry season.
Commonly, patients with seborrhoeic dermatitis experience mild redness, scaly skin lesions and in some cases hair loss.
Low humidity can cause adverse health effects and may cause atopic dermatitis, and seborrhoeic dermatitis.
Treatments
Combination therapy
Combinations of finasteride, minoxidil and ketoconazole are more effective than individual use.
Combination therapy of LLLT or microneedling with finasteride or minoxidil demonstrated substantive increases in hair count.
Medication
Add-hocs
The topical bioavailability of synthetic medicines such as antiandrogens, as well as phytochemicals such as quercetin, has been improved with incorporation of drugs in nanoparticles, or microneedling.
Synthetic medicine
Treatments for the various forms of hair loss have only moderate success. Three medications have evidence to support their use in male pattern hair loss: finasteride, dutasteride and minoxidil. They typically work better to prevent further hair loss than to regrow lost hair.
They may be used together when hair loss is progressive or further regrowth is desired after 12 months. Other medications include ketoconazole, and in female androgenic alopecia spironolactone and flutamide.
Baricitinib
In June 2022, the FDA authorized baricitinib for the treatment of severe alopecia areata.
Minoxidil
Minoxidil, applied topically, is widely used for the treatment of hair loss. It may be effective in helping promote hair growth in both men and women with androgenic alopecia. About 40% of men experience hair regrowth after 3–6 months. It is the only topical product that is FDA approved in America for androgenic hair loss. However, increased hair loss has been reported.
Antiandrogens
Finasteride is used to treat male pattern hair loss. Treatment provides about 30% improvement in hair loss after six months of treatment, and effectiveness only persists as long as the drug is taken. There is no good evidence for its use in women. It may cause gynecomastia, erectile dysfunction and depression.
Dutasteride is also used in the treatment of male pattern hair loss and appears to have better effectiveness than finasteride for the condition. While used off-label for male pattern hair loss in most of the world, dutasteride is specifically approved for this indication in South Korea and Japan.
There is tentative support for spironolactone in women. Due to its feminising side effects and risk of infertility it is not often used by men. It can also cause low blood pressure, high blood potassium, and abnormal heart rhythms. Also, women who are pregnant or trying to become pregnant generally cannot use the medication as it is a teratogen, and can cause ambiguous genitalia in newborn children.
There is tentative evidence for flutamide in women; however, it is associated with relatively high rates of liver problems and strong recommendations have been made against its use. Like spironolactone, flutamide is typically only used by women. Bicalutamide is another option for the treatment of female pattern hair loss. It has a far lower risk of liver toxicity than flutamide and is said to have an excellent safety profile. However, bicalutamide retains a small risk of liver toxicity and for this reason periodic liver monitoring is recommended during treatment.
Ketoconazole
Ketoconazole may help in women.
Technological treatments
Low-level laser therapy (LLLT)
Low-level laser therapy or photobiomodulation is also referred to as red light therapy and cold laser therapy. It is a non-invasive treatment option.
LLLT is shown to increase hair density and growth in both genders. The types of devices (hat, comb, helmet) and duration did not alter the effectiveness, with more emphasis to be placed on lasers compared to LEDs. Ultraviolet and infrared light are more effective for alopecia areata, while red light and infrared light is more effective for androgenetic alopecia.
Medical reviews suggest that LLLT is as effective or potentially more than other non invasive and traditional therapies like minoxidil and finasteride but further studies such as RCTs, long term follow up studies, and larger double blinded trials need to be conducted to confirm the initial findings.
Platelet-rich plasma (PRP)
Using ones own cells and tissues and without harsh side effects, PRP is beneficial for alopecia areata and androgenetic alopecia and can be used as an alternative to minoxidil or finasteride. It has been documented to improve hair density and thickness in both genders. A minimum of 3 treatments, once a month for 3 months are recommended, and afterwards a 3-6 month period of continual appointments for maintenance. Factors that determine efficacy include amount of sessions, double versus single centrifugation, age and gender, and where the PRP is inserted.
Future larger randomized controlled trials and other high quality studies are still recommended to be carried out and published for a stronger consensus. Further development of a standardized practice for procedure is also recommended.
Surgical treatments
Hair transplantation
Main article: Hair transplantation
 A man before and after a hair transplant.
A man before and after a hair transplant.
Hair transplantation is a surgical technique that moves individual hair follicles from a part of the body called the donor site to bald or balding part of the body known as the recipient site. It is primarily used to treat male pattern baldness. In this condition, grafts containing hair follicles that are genetically resistant to balding are transplanted to bald scalp. It is also used to restore eyelashes, eyebrows, beard hair, chest hair, and pubic hair and to fill in scars caused by accidents or surgery such as face-lifts and previous hair transplants. Hair transplantation differs from skin grafting in that grafts contain almost all of the epidermis and dermis surrounding the hair follicle, and many tiny grafts are transplanted rather than a single strip of skin.
Since hair naturally grows in follicles in groups of 1 to 4 hairs, transplantation takes advantage of these naturally occurring follicular units. This achieves a more natural appearance by matching hair for hair through Follicular unit transplantation (FUT).
Donor hair can be harvested in two different ways. Small grafts of naturally-occurring units of one to four hairs, called follicular units, can be moved to balding areas of the hair restoration. These follicular units are surgically implanted in the scalp in very close proximity to one another and in large numbers. The grafts are obtained in one or both of the two primary methods of surgical extraction, follicular unit transplantation, colloquially referred to as "strip harvesting", or Follicular Unit Extraction (FUE), in which follicles are transplanted individually.
In FUT, a strip of skin containing many follicular units is extracted from the patient and dissected under a stereoscopic microscope. The site of the strip removal is stitched closed. Once divided into follicular unit grafts, each unit is individually inserted into small recipient sites made by an incision in the bald scalp. In the newer technique, roots are extracted from the donor area and divided into strips for transplantation. The strip, two to three millimeters thick, is isolated and transplanted to the bald scalp. After surgery, a bandage is worn for two days to protect the stitched strip during healing. A small strip scar remains after healing, which can be covered by scalp hair growing over the scar.
Scalp reduction
Main article: Scalp reductionScalp reduction is a surgical procedure in which the hairless region of the scalp of a bald man is reduced. This procedure can reduce the area of the scalp without hair.
Regrowth of hair
Facial hair
Eyebrows
Bimatoprost 0.03% has been used to grow eyebrows.
Eyelashes
The FP receptor agonist, bimatoprost, in the form of an 0.03% ophthalmic solution termed Latisse, is approved by the US Food and Drug Administration to treat hypotrichosis of the eyelashes, in particular to darken and lengthen eyelashes for cosmetic purposes. Also, bimatoprost may be used to treat small or underdeveloped eyelashes.
Scalp hair
Alternative medication
Topical
Some popular plant juices sold as hair serum may instead of growing human hair actually inhibit the growth, including 6-gingerol found in ginger.
Topical crude onion juice

A small 2002 study demonstrated that treatment twice daily for six weeks with crude onion juice from Australian brown onion, re-growth hair on alopecia areata (spot baldness) in 86.9% of the 23 participants. Twice as many flavonols are found in red onion than in yellow onion. Also, non-organic onions might contain pesticides on the peel and in the first scaly leaf. Compounds found in onion that stimulates hair growth:
- Quercetin:
- Quercetin (a flavanoid found in vegan food) supplements, has been suggested to treat baldness. A 2012 study demonstrated that alopecia areata could be used to prevent ant treated with quercetin in mice. Quercetin is found in onions primarily in the peel and the first scaly leaf but not in the flesh. Onion bulb size or weight does not appear to be affected by quercetin concentration.
- A 2020 study demonstrated that quercitrin stimulated hair shaft growth in cultured human hair follicles.
- Volatile compounds responsible for pungency and tearing in onions when they are cut (e.g. syn-propanethial S-oxide gas). However, no formal studies have conducted to evaluate if these compounds promote hair growth or if they help other compounds to penetrate the skin to form hair follicles.
- To use the volatile compounds: Onions must be extracted and applied topically quickly before the volatile compounds evaporates. The extract will cause tearing eyes, but it can be prevented with a shower cap, or a pair of swimming goggles.
- To discard the volatile compounds: The onion juice is extracted and stored for a short while to evaporate the volatile compounds. The extract will not cause tearing eyes. Also, both genetically modified, and plant breed onions have been produced with significantly reduced levels of tear-inducing lachrymatory factor.
Dietary supplements
Dietary supplements are not typically recommended. Many people use unproven treatments, but there is little evidence of the effectiveness of vitamins, minerals, or other dietary supplements regrowing hair or retaining hair. There is no evidence for biotin (vitamin B7). While lacking both evidence and expert recommendation, there is a large market for hair growth supplements, especially for products that contain biotin.
However, one small trial of saw palmetto which shows tentative benefit in those with mild to moderate androgenetic alopecia. There was no good evidence for gingko, aloe vera, ginseng, bergamot, or hibiscus as of 2011.
Radiation-induced hair loss
Radiation induces hair loss through damage to hair follicle stem cell progenitors and alteration of keratin expression. Radiation therapy has been associated with increased mucin production in hair follicles.
Studies have suggested electromagnetic radiation as a therapeutic growth stimulant in alopecia.
Cosmeses

Certain hair shampoos and ointments visually thicken existing hair, without affecting the growth cycle. There have also been developments in the fashion industry with wig design. The fashion accessory has also been shown to be a source of psychological support for women undergoing chemotherapy, with cancer survivors in one study describing their wig as a "constant companion". Other studies in women have demonstrated a more mixed psychosocial impact of hairpiece use.
Specialized tattoos, commonly known as scalp micropigmentation, can mimic the appearance of a short buzzed haircut.
Human hair growth
Main article: Human hair growth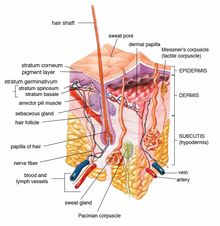
Type 1 and 2 5α reductase enzymes are present at pilosebaceous units in papillae of individual hair follicles. They catalyse formation of the androgens testosterone and DHT, which in turn regulate hair growth. Androgens have different effects at different follicles: they stimulate IGF-1 at facial hair, causing hair regrowth, but stimulate TGF β1, TGF β2, dickkopf1 and IL-6 at the scalp, causing hair follicle miniaturisation.
Female androgenic alopecia is characterized by diffuse crown thinning without hairline recession, and like its male counterpart rarely leads to total hair loss. Finasteride and minoxidil are usually first line therapy for its treatment. Other options include topical or systemic spironolactone or flutamide, although they have a high incidence of feminising side effects and are better tolerated in female androgenic hair loss.
More advanced cases may be resistant or unresponsive to medical therapy, however, and require hair transplantation. Naturally-occurring units of one to four hairs, called follicular units, are excised and moved to areas of hair restoration. These follicular units are surgically implanted in the scalp in close proximity and in large numbers. The grafts are obtained from either Follicular Unit Transplantation (FUT) – colloquially referred to as "strip harvesting" – or Follicular Unit Extraction (FUE). In the former, a strip of skin with follicular units is extracted and dissected into individual follicular unit grafts. The surgeon then implants the grafts into small incisions, called recipient sites. Specialized scalp tattoos can also mimic the appearance of a short buzzed haircut. Androgenic alopecia also occurs in women, and more often presents as diffuse thinning without hairline recession. Like its male counterpart, the condition rarely leads to total hair loss. Treatment options are similar to those for men, although topical or systemic estrogen is used more often.
Research
Bimatoprost and latanoprost
Latanoprost and bimatoprost are specific PGF2a analogues applied topically, and have been found to lengthen eyelashes, darken hair pigmentation and elongate hair. Bimatoprost is available as treatment for eyelash growth. Latanoprost has shown ability to promote scalp hair density and pigmentation, and is theorized to function at the dermal papilla. A study found latanoprost ineffective on eyelashes in a patient with alopecia areata. It has also been found ineffective in treatment of eyebrow hair loss.
Hormones
Estrogens are indirect anti-androgens and can be used to treat androgenetic hair loss in women with oral contraceptives. Systemic estrogen increases SHBG, which binds androgens, including testosterone and DHT, in turn reducing their bioavailability. Topical formulations are available in Europe. Hair follicles have estrogen receptors and it is theorized that topical compounds act on them directly to promote hair growth and antagonize androgen action. Large clinical studies showing effectiveness are absent. Topical treatment is also usually unavailable in North America.
There is tentative evidence for cyproterone acetate in women.
IGF-1
In December 2012, topical application of IGF-1 in a liposomal vehicle led to thicker and more rapid hair growth in transgenic mice with androgenic alopecia. The study did not show measurable systemic levels or hematopoietic side effects, suggesting potential for use in humans. Low energy radiofrequency irradiation induces IGF-1 in cultured human dermal papilla cells. Adenosine stimulates dermal papillae in vitro to induce IGF-1, along with fibroblast growth factors FGF7, FGF-2 and VEGF. β-catenin transcription increased, which promotes dermal papillae as well. Dietary isoflavones increase IGF production in scalp dermal papillae in transgenic mice. Topical capsaicin also stimulates IGF at hair follicles via release of vanilloid receptor-1, which in turn leads to more CGRP. Ascorbic acid has led to increased IGF expression in vitro.
Stem cell therapy
Main article: Hair cloningAlthough follicles were previously thought gone in areas of complete baldness, they are more likely dormant, as recent studies have shown the scalp contains the stem cells from which the follicles arose. Research on these follicular stem cells may lead to successes in treating baldness through hair multiplication (HM), also known as hair cloning.
Per a May 2015 review, no successful strategy to generate human hair follicles, for hair regrowth, from adult stem cells has yet been reported. However, in April 2016, scientists from Japan published results of their work in which they created human skin from induced pluripotent stem cells; implanted into laboratory mice, the cells generated skin with hair and glands.
Genetics
From 2005 to 2007, Curis and Procter & Gamble collaborated on developing a topical hedgehog agonist for hair loss; the agent did not meet safety standards, and the program was terminated. In 2008, researchers at the University of Bonn announced they have found the genetic basis of two distinct forms of inherited hair loss. They found the gene P2RY5 causes a rare, inherited form of hair loss called hypotrichosis simplex. It is the first receptor in humans known to play a role in hair growth. Researchers found that disruption of the gene SOX21 in mice caused cyclical hair loss. Research has suggested SOX21 as a master regulator of hair shaft cuticle differentiation, with its disruption causing cyclical alopecia in mice models. Deletion of SOX21 dramatically affects hair lipids.
References
- Bin Saif, GA; Ericson, ME; Yosipovitch, G (December 2011). "The itchy scalp—scratching for an explanation". Experimental Dermatology. 20 (12): 959–68. doi:10.1111/j.1600-0625.2011.01389.x. PMC 3233984. PMID 22092575.
- Morinaga, H; Mohri, Y; Grachtchouk, M; Asakawa, K; Matsumura, H; Oshima, M; Takayama, N; Kato, T; Nishimori, Y; Sorimachi, Y; Takubo, K; Suganami, T; Iwama, A; Iwakura, Y; Dlugosz, AA; Nishimura, EK (July 2021). "Obesity accelerates hair thinning by stem cell-centric converging mechanisms". Nature. 595 (7866): 266–271. Bibcode:2021Natur.595..266M. doi:10.1038/s41586-021-03624-x. PMC 9600322. PMID 34163066. S2CID 235625692.
- Søyland, E; Funk, J; Rajka, G; Sandberg, M; Thune, P; Rustad, L; Helland, S; Middelfart, K; Odu, S; Falk, ES (June 1994). "Dietary supplementation with very long-chain n-3 fatty acids in patients with atopic dermatitis. A double-blind, multicentre study". The British Journal of Dermatology. 130 (6): 757–64. doi:10.1111/j.1365-2133.1994.tb03414.x. PMID 8011502. S2CID 25066444.
- Callaway, J; Schwab, U; Harvima, I; Halonen, P; Mykkänen, O; Hyvönen, P; Järvinen, T (April 2005). "Efficacy of dietary hempseed oil in patients with atopic dermatitis". The Journal of Dermatological Treatment. 16 (2): 87–94. doi:10.1080/09546630510035832. PMID 16019622. S2CID 18445488.
- Ashique S, Sandhu NK, Haque SN, Koley K (December 2020). "A Systemic Review on Topical Marketed Formulations, Natural Products, and Oral Supplements to Prevent Androgenic Alopecia: A Review". Natural Products and Bioprospecting. 10 (6): 345–365. doi:10.1007/s13659-020-00267-9. PMC 7648777. PMID 33011954.
- "Seasonal Shedding Hair in Summer or Winter!". MDM Health. October 28, 2015. Archived from the original on December 9, 2021. Retrieved December 9, 2021.
- "What is Seborrheic Dermatitis?". Archived from the original on April 20, 2010. Retrieved June 11, 2010.
- Engebretsen KA, Johansen JD, Kezic S, Linneberg A, Thyssen JP (February 2016). "The effect of environmental humidity and temperature on skin barrier function and dermatitis". Journal of the European Academy of Dermatology and Venereology. 30 (2): 223–249. doi:10.1111/jdv.13301. PMID 26449379. S2CID 12378072.
- Araya M, Kulthanan K, Jiamton S (September 2015). "Clinical Characteristics and Quality of Life of Seborrheic Dermatitis Patients in a Tropical Country". Indian Journal of Dermatology. 60 (5): 519. doi:10.4103/0019-5154.164410. PMC 4601435. PMID 26538714.
- Khandpur S, Suman M, Reddy BS (August 2002). "Comparative efficacy of various treatment regimens for androgenetic alopecia in men". The Journal of Dermatology. 29 (8): 489–498. doi:10.1111/j.1346-8138.2002.tb00314.x. PMID 12227482. S2CID 20886812.
- ^ Darwin, Evan; Heyes, Alexandra; Hirt, Penelope A.; Wikramanayake, Tongyu Cao; Jimenez, Joaquin J. (December 21, 2017). "Low-level laser therapy for the treatment of androgenic alopecia: a review". Lasers in Medical Science. 33 (2). Springer Science and Business Media LLC: 425–434. doi:10.1007/s10103-017-2385-5. ISSN 0268-8921. PMID 29270707. S2CID 23783876.
- Zhou, Yi; Chen, Chenglong; Qu, Qian; Zhang, Chunhua; Wang, Jin; Fan, Zhexiang; Miao, Yong; Hu, Zhiqi (2020). "The effectiveness of combination therapies for androgenetic alopecia: A systematic review and meta-analysis". Dermatologic Therapy. 33 (4). Hindawi Limited: e13741. doi:10.1111/dth.13741. ISSN 1396-0296. PMID 32478968.
- Cardoso CO, Tolentino S, Gratieri T, Cunha-Filho M, Lopez RF, Gelfuso GM (2021). "Topical Treatment for Scarring and Non-Scarring Alopecia: An Overview of the Current Evidence". Clinical, Cosmetic and Investigational Dermatology. 14: 485–499. doi:10.2147/CCID.S284435. PMC 8126704. PMID 34012282.
- ^ Levy LL, Emer JJ (August 2013). "Female pattern alopecia: current perspectives". International Journal of Women's Health. 5: 541–556. doi:10.2147/IJWH.S49337. PMC 3769411. PMID 24039457.
- ^ Banka N, Bunagan MJ, Shapiro J (January 2013). "Pattern hair loss in men: diagnosis and medical treatment". Dermatologic Clinics. 31 (1): 129–140. doi:10.1016/j.det.2012.08.003. PMID 23159182.
- "Propecia & Rogaine for Treating Male Pattern Baldness". Webmd.com. Archived from the original on July 9, 2008. Retrieved May 19, 2010.
- ^ McElwee KJ, Shapiro JS (June 2012). "Promising therapies for treating and/or preventing androgenic alopecia". Skin Therapy Letter. 17 (6): 1–4. PMID 22735503.
- "FDA Approves First Systemic Treatment for Alopecia Areata". U.S. Food and Drug Administration (FDA) (Press release). June 13, 2022. Archived from the original on June 14, 2022. Retrieved June 13, 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- "FDA Approves Lilly and Incyte's Olumiant (baricitinib) As First and Only Systemic Medicine for Adults with Severe Alopecia Areata" (Press release). Eli Lilly. June 13, 2022. Archived from the original on June 14, 2022. Retrieved June 13, 2022 – via PR Newswire.
- ^ Varothai S, Bergfeld WF (July 2014). "Androgenetic alopecia: an evidence-based treatment update". American Journal of Clinical Dermatology. 15 (3): 217–230. doi:10.1007/s40257-014-0077-5. PMID 24848508. S2CID 31245042.
- van Zuuren EJ, Fedorowicz Z, Schoones J (May 2016). "Interventions for female pattern hair loss". The Cochrane Database of Systematic Reviews. 2016 (5): CD007628. doi:10.1002/14651858.CD007628.pub4. PMC 6457957. PMID 27225981.
- Goren A, Shapiro J, Roberts J, McCoy J, Desai N, Zarrab Z, et al. (2015). "Clinical utility and validity of minoxidil response testing in androgenetic alopecia". Dermatologic Therapy. 28 (1): 13–16. doi:10.1111/dth.12164. PMID 25112173. S2CID 205082682.
- "Rogaine Side Effects in Detail - Drugs.com". drugs.com. Archived from the original on September 22, 2017. Retrieved January 28, 2018.
- "Minoxidil Official FDA information, side effects and uses". Drugs.com. Archived from the original on September 22, 2017. Retrieved January 28, 2018.
- "Propecia label" (PDF). Archived (PDF) from the original on February 10, 2017. Retrieved June 3, 2015.
- Andersson S (2001). "Steroidogenic enzymes in skin". European Journal of Dermatology. 11 (4): 293–295. PMID 11399532.
- "Avodart 0.5mg soft capsules – Summary of Product Characteristics (SmPC) – (eMC)". emc.medicines.org.uk. Archived from the original on April 26, 2006. Retrieved August 7, 2010.
- Zhou Z, Song S, Gao Z, Wu J, Ma J, Cui Y (2019). "The efficacy and safety of dutasteride compared with finasteride in treating men with androgenetic alopecia: a systematic review and meta-analysis". Clinical Interventions in Aging. 14: 399–406. doi:10.2147/CIA.S192435. PMC 6388756. PMID 30863034.
- Olsen EA, Hordinsky M, Whiting D, Stough D, Hobbs S, Ellis ML, et al. (December 2006). "The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: results of a randomized placebo-controlled study of dutasteride versus finasteride". Journal of the American Academy of Dermatology. 55 (6): 1014–1023. doi:10.1016/j.jaad.2006.05.007. PMID 17110217.
- Choi GS, Kim JH, Oh SY, Park JM, Hong JS, Lee YS, Lee WS (August 2016). "Safety and Tolerability of the Dual 5-Alpha Reductase Inhibitor Dutasteride in the Treatment of Androgenetic Alopecia". Annals of Dermatology. 28 (4): 444–450. doi:10.5021/ad.2016.28.4.444. PMC 4969473. PMID 27489426.
- Shapiro J, Otberg N (April 17, 2015). Hair Loss and Restoration, Second Edition. CRC Press. pp. 39–. ISBN 978-1-4822-3199-1. Archived from the original on January 12, 2023. Retrieved December 9, 2021.
- Buchanan JF, Davis LJ (February 1984). "Drug-induced infertility". Drug Intelligence & Clinical Pharmacy. 18 (2): 122–132. doi:10.1177/106002808401800205. PMID 6141923. S2CID 24807929.
- Sinclair R, Patel M, Dawson TL, Yazdabadi A, Yip L, Perez A, Rufaut NW (December 2011). "Hair loss in women: medical and cosmetic approaches to increase scalp hair fullness". The British Journal of Dermatology. 165 (Suppl 3): 12–18. doi:10.1111/j.1365-2133.2011.10630.x. PMID 22171680. S2CID 39734527.
- Rathnayake D, Sinclair R (2010). "Use of spironolactone in dermatology". Skinmed. 8 (6): 328–32, quiz 333. PMID 21413648.
- Giorgetti R, di Muzio M, Giorgetti A, Girolami D, Borgia L, Tagliabracci A (March 2017). "Flutamide-induced hepatotoxicity: ethical and scientific issues". European Review for Medical and Pharmacological Sciences. 21 (1 Suppl): 69–77. PMID 28379593.
- Yazdabadi A, Sinclair R (May 2011). "Treatment of female pattern hair loss with the androgen receptor antagonist flutamide". The Australasian Journal of Dermatology. 52 (2): 132–134. doi:10.1111/j.1440-0960.2010.00735.x. PMID 21605098. S2CID 30128397.
- Carvalho RM, Santos LD, Ramos PM, Machado CJ, Acioly P, Frattini SC, et al. (January 2022). "Bicalutamide and the new perspectives for female pattern hair loss treatment: What dermatologists should know". Journal of Cosmetic Dermatology. 21 (10): 4171–4175. doi:10.1111/jocd.14773. PMID 35032336. S2CID 253239337.
- ^ Meyer-Gonzalez T, Bacqueville D, Grimalt R, Mengeaud V, Piraccini BM, Rudnicka L, et al. (November 2021). "Current controversies in trichology: a European expert consensus statement". Journal of the European Academy of Dermatology and Venereology. 35 (Suppl 2): 3–11. doi:10.1111/jdv.17601. hdl:11585/863826. PMID 34668238. S2CID 239029062.
- ^ Nestor MS, Ablon G, Gade A, Han H, Fischer DL (December 2021). "Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics". Journal of Cosmetic Dermatology. 20 (12): 3759–3781. doi:10.1111/jocd.14537. PMC 9298335. PMID 34741573. S2CID 243801494.
- ^ Cignarella A, Mioni R, Sabbadin C, Dassie F, Parolin M, Vettor R, et al. (December 2020). "Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS". International Journal of Molecular Sciences. 21 (24): 9554. doi:10.3390/ijms21249554. PMC 7765466. PMID 33334002.
- Piérard-Franchimont C, De Doncker P, Cauwenbergh G, Piérard GE (1998). "Ketoconazole shampoo: effect of long-term use in androgenic alopecia". Dermatology. 196 (4): 474–477. doi:10.1159/000017954. PMID 9669136. S2CID 30635892.
- Fields JR, Vonu PM, Monir RL, Schoch JJ (January 2020). "Topical ketoconazole for the treatment of androgenetic alopecia: A systematic review". Dermatologic Therapy. 33 (1): e13202. doi:10.1111/dth.13202. PMID 31858672. S2CID 209427721.
- Liu, Kao-Hui; Liu, Donald; Chen, Yu-Tsung; Chin, Szu-Ying (January 31, 2019). "Comparative effectiveness of low-level laser therapy for adult androgenic alopecia: a system review and meta-analysis of randomized controlled trials". Lasers in Medical Science. 34 (6). Springer Science and Business Media LLC: 1063–1069. doi:10.1007/s10103-019-02723-6. ISSN 0268-8921. PMID 30706177. S2CID 59524423.
- Gupta, A. K.; Carviel, J. L. (November 20, 2019). "Meta-analysis of photobiomodulation for the treatment of androgenetic alopecia". Journal of Dermatological Treatment. 32 (6). Informa UK Limited: 643–647. doi:10.1080/09546634.2019.1688755. ISSN 0954-6634. PMID 31746251. S2CID 208185306.
- Zhang, Yuehou; Su, Jianlong; Ma, Kui; Fu, Xiaobing; Zhang, Cuiping (April 25, 2022). "Photobiomodulation Therapy With Different Wavebands for Hair Loss: A Systematic Review and Meta-Analysis". Dermatologic Surgery. 48 (7). Ovid Technologies (Wolters Kluwer Health): 737–740. doi:10.1097/dss.0000000000003472. ISSN 1076-0512. PMID 35510860. S2CID 248526019.
- ^ Gupta, Aditya K.; Bamimore, Mary A.; Foley, Kelly A. (April 13, 2020). "Efficacy of non-surgical treatments for androgenetic alopecia in men and women: a systematic review with network meta-analyses, and an assessment of evidence quality". Journal of Dermatological Treatment. 33 (1). Informa UK Limited: 62–72. doi:10.1080/09546634.2020.1749547. ISSN 0954-6634. PMID 32250713. S2CID 215405183. Archived from the original on February 24, 2024. Retrieved August 31, 2023.
- S, Lueangarun; P, Visutjindaporn; Y, Parcharoen; P, Jamparuang; T, Tempark (2021). "A Systematic Review and Meta-analysis of Randomized Controlled Trials of United States Food and Drug Administration-Approved, Home-use, Low-Level Light/Laser Therapy Devices for Pattern Hair Loss: Device Design and Technology". The Journal of Clinical and Aesthetic Dermatology. 14 (11): E64 – E75. ISSN 1941-2789. PMC 8675345. PMID 34980962.
- ^ Tejapira, Kasama; Yongpisarn, Tanat; Sakpuwadol, Nawara; Suchonwanit, Poonkiat (November 24, 2022). "Platelet-rich plasma in alopecia areata and primary cicatricial alopecias: A systematic review". Frontiers in Medicine. 9. Frontiers Media SA. doi:10.3389/fmed.2022.1058431. ISSN 2296-858X. PMC 9731377. PMID 36507528.
- Gentile, Pietro; Garcovich, Simone (April 13, 2020). "Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy". International Journal of Molecular Sciences. 21 (8). MDPI AG: 2702. doi:10.3390/ijms21082702. ISSN 1422-0067. PMC 7216252. PMID 32295047.
- ^ Evans, Adam G.; Mwangi, James M.; Pope, Rand W.; Ivanic, Mirjana G.; Botros, Mina A.; Glassman, Gabriella E.; Pearce, F. Bennett; Kassis, Salam (May 26, 2020). "Platelet-rich plasma as a therapy for androgenic alopecia: a systematic review and meta-analysis". Journal of Dermatological Treatment. 33 (1). Informa UK Limited: 498–511. doi:10.1080/09546634.2020.1770171. ISSN 0954-6634. PMID 32410524. S2CID 218648227. Archived from the original on February 24, 2024. Retrieved August 31, 2023.
- Gupta, Aditya K.; Cole, John; Deutsch, David P.; Everts, Peter A.; Niedbalski, Robert P.; Panchaprateep, Ratchathorn; Rinaldi, Fabio; Rose, Paul T.; Sinclair, Rodney; Vogel, James E.; Welter, Ryan J.; Zufelt, Michael D.; Puig, Carlos J. (2019). "Platelet-Rich Plasma as a Treatment for Androgenetic Alopecia". Dermatologic Surgery. 45 (10). Ovid Technologies (Wolters Kluwer Health): 1262–1273. doi:10.1097/dss.0000000000001894. ISSN 1076-0512. PMID 30882509. S2CID 81980415.
- Gupta, Aditya; Bamimore, Mary (August 1, 2022). "Platelet-Rich Plasma Monotherapies for Androgenetic Alopecia: A Network Meta-Analysis and Meta-Regression Study". Journal of Drugs in Dermatology. 21 (9). SanovaWorks: 943–952. doi:10.36849/jdd.6948. ISSN 1545-9616. PMID 36074501. S2CID 252120370.
- Oth, O; Stene, JJ; Glineur, R; Vujovic, A (2018). "Injection of PRP (Platelet-rich plasma) as a treatment for androgenetic alopecia : a systematic review of the literature". Revue Médicale de Bruxelles. 39 (5). AMUB/Revue Médicale de Bruxelles: 438–446. doi:10.30637/2018.17-056. ISSN 0035-3639. PMID 29869472.
- Rashid RM, Morgan Bicknell LT (September 2012). "Follicular unit extraction hair transplant automation: options in overcoming challenges of the latest technology in hair restoration with the goal of avoiding the line scar". Dermatology Online Journal. 18 (9): 12. doi:10.5070/D30X57S71R. PMID 23031379.
- ^ Caroli S, Pathomvanich D, Amonpattana K, Kumar A (2011). "Current status of hair restoration surgery". International Surgery. 96 (4): 345–351. doi:10.9738/cc31.1. PMID 22808618.
- Bell ML (February 1982). "Role of scalp reduction in the treatment of male pattern baldness". Plastic and Reconstructive Surgery. 69 (2): 272–277. doi:10.1097/00006534-198202000-00016. PMID 7054796. S2CID 20731930.
- Unger, Martin G.; Toscani, Marco (2016). "Scalp reduction". In Scuderi, Nicolò; Toth, Bryant A. (eds.). International Textbook of Aesthetic Surgery. Springer. pp. 555–556. doi:10.1007/978-3-662-46599-8. ISBN 978-3-662-46598-1. Archived from the original on August 3, 2017. Retrieved December 7, 2016.
- Riahi, RR; Cohen, PR (May 21, 2018). "Topical Treatment of Eyebrow Hypotrichosis with Bimatoprost 0.03% Solution: Case Report and Literature Review". Cureus. 10 (5): e2666. doi:10.7759/cureus.2666. PMC 6054329. PMID 30042917.
- Choi, YM; Diehl, J; Levins, PC (April 2015). "Promising alternative clinical uses of prostaglandin F2α analogs: beyond the eyelashes". Journal of the American Academy of Dermatology. 72 (4): 712–6. doi:10.1016/j.jaad.2014.10.012. PMID 25601618.
- "Bimatoprost solution/ drops". DailyMed. September 18, 2019. Archived from the original on September 21, 2020. Retrieved January 4, 2020.
- "Lumigan- bimatoprost solution/ drops". DailyMed. July 31, 2017. Archived from the original on September 21, 2020. Retrieved January 4, 2020.
- Miao Y, Sun Y, Wang W, Du B, Xiao SE, Hu Y, Hu Z (2013). "6-Gingerol inhibits hair shaft growth in cultured human hair follicles and modulates hair growth in mice". PLOS ONE. 8 (2): e57226. Bibcode:2013PLoSO...857226M. doi:10.1371/journal.pone.0057226. PMC 3578824. PMID 23437345.
- ^ Sharquie KE, Al-Obaidi HK (June 2002). "Onion juice (Allium cepa L.), a new topical treatment for alopecia areata". The Journal of Dermatology. 29 (6): 343–346. doi:10.1111/j.1346-8138.2002.tb00277.x. PMID 12126069. S2CID 24116769.
- ^ Slimestad R, Fossen T, Vågen IM (December 2007). "Onions: a source of unique dietary flavonoids". Journal of Agricultural and Food Chemistry. 55 (25): 10067–10080. doi:10.1021/jf0712503. PMID 17997520.
- ^ Wikramanayake TC, Villasante AC, Mauro LM, Perez CI, Schachner LA, Jimenez JJ (March 2012). "Prevention and treatment of alopecia areata with quercetin in the C3H/HeJ mouse model". Cell Stress & Chaperones. 17 (2): 267–274. doi:10.1007/s12192-011-0305-3. PMC 3273564. PMID 22042611.
- Kwak JH, Seo JM, Kim NH, Arasu MV, Kim S, Yoon MK, Kim SJ (September 2017). "Variation of quercetin glycoside derivatives in three onion (Allium cepa L.) varieties". Saudi Journal of Biological Sciences. 24 (6): 1387–1391. doi:10.1016/j.sjbs.2016.05.014. PMC 5562462. PMID 28855836.
- Kim J, Kim SR, Choi YH, Shin JY, Kim CD, Kang NG, et al. (September 2020). "Quercitrin Stimulates Hair Growth with Enhanced Expression of Growth Factors via Activation of MAPK/CREB Signaling Pathway". Molecules. 25 (17): 4004. doi:10.3390/molecules25174004. PMC 7504764. PMID 32887384.
- Eady CC, Kamoi T, Kato M, Porter NG, Davis S, Shaw M, et al. (August 2008). "Silencing onion lachrymatory factor synthase causes a significant change in the sulfur secondary metabolite profile". Plant Physiology. 147 (4): 2096–2106. doi:10.1104/pp.108.123273. PMC 2492635. PMID 18583530.
- "Cutting this new type of onion won't make you cry". The Independent. January 9, 2018. Archived from the original on September 7, 2021. Retrieved September 7, 2021.
- ^ Rogers NE, Avram MR (October 2008). "Medical treatments for male and female pattern hair loss". Journal of the American Academy of Dermatology. 59 (4): 547–66, quiz 567–8. doi:10.1016/j.jaad.2008.07.001. PMID 18793935.
- ^ Blumeyer A, Tosti A, Messenger A, Reygagne P, Del Marmol V, Spuls PI, et al. (October 2011). "Evidence-based (S3) guideline for the treatment of androgenetic alopecia in women and in men". Journal der Deutschen Dermatologischen Gesellschaft. 9 (Suppl 6): S1-57. doi:10.1111/j.1610-0379.2011.07802.x. PMID 21980982. S2CID 29821046.
- Nanashima N, Ito K, Ishikawa T, Nakano M, Nakamura T (September 2012). "Damage of hair follicle stem cells and alteration of keratin expression in external radiation-induced acute alopecia". International Journal of Molecular Medicine. 30 (3): 579–584. doi:10.3892/ijmm.2012.1018. PMID 22692500.
- Kamiya K, Sasatani M (March 2012). "". Nihon Rinsho. Japanese Journal of Clinical Medicine. 70 (3): 367–374. PMID 22514910.
- Takeda H, Nakajima K, Kaneko T, Harada K, Matsuzaki Y, Sawamura D (November 2011). "Follicular mucinosis associated with radiation therapy". The Journal of Dermatology. 38 (11): 1116–1118. doi:10.1111/j.1346-8138.2010.01187.x. PMID 22034994. S2CID 31876890.
- Kalia S, Lui H (January 2013). "Utilizing electromagnetic radiation for hair growth: a critical review of phototrichogenesis". Dermatologic Clinics. 31 (1): 193–200. doi:10.1016/j.det.2012.08.018. PMID 23159188.
- Davis MG, Thomas JH, van de Velde S, Boissy Y, Dawson TL, Iveson R, Sutton K (December 2011). "A novel cosmetic approach to treat thinning hair". The British Journal of Dermatology. 165 (Suppl 3): 24–30. doi:10.1111/j.1365-2133.2011.10633.x. PMID 22171682. S2CID 21918537.
- Zannini L, Verderame F, Cucchiara G, Zinna B, Alba A, Ferrara M (September 2012). "'My wig has been my journey's companion': perceived effects of an aesthetic care programme for Italian women suffering from chemotherapy-induced alopecia". European Journal of Cancer Care. 21 (5): 650–660. doi:10.1111/j.1365-2354.2012.01337.x. PMID 22339814.
- Inui S, Inoue T, Itami S (March 2013). "Psychosocial impact of wigs or hairpieces on perceived quality of life level in female patients with alopecia areata". The Journal of Dermatology. 40 (3): 225–226. doi:10.1111/1346-8138.12040. PMID 23252418. S2CID 43795003.
- Elisabeth Leamy (May 31, 2012). "Considering a hair tattoo? Pros and cons to consider before you commit". ABC News. Archived from the original on April 11, 2019. Retrieved December 16, 2012.
- Inui S, Itami S (March 2013). "Androgen actions on the human hair follicle: perspectives". Experimental Dermatology. 22 (3): 168–171. doi:10.1111/exd.12024. PMID 23016593. S2CID 33521841.
- ^ "Female pattern baldness". MedlinePlus. December 15, 2012. Archived from the original on December 1, 2012. Retrieved December 15, 2012.
- Rose PT (August 2011). "The latest innovations in hair transplantation". Facial Plastic Surgery. 27 (4): 366–377. doi:10.1055/s-0031-1283055. PMID 21792780. S2CID 260138453.
- Elisabeth Leamy (May 31, 2012). "Considering a hair tattoo? Pros and cons to consider before you commit". ABC News. Archived from the original on April 11, 2019. Retrieved December 16, 2012.
- Kovalevsky G, Ballagh SA, Stanczyk FZ, Lee J, Cooper J, Archer DF (April 2010). "Levonorgestrel effects on serum androgens, sex hormone-binding globulin levels, hair shaft diameter, and sexual function". Fertility and Sterility. 93 (6): 1997–2003. doi:10.1016/j.fertnstert.2008.12.095. PMID 19394598.
- Law SK (April 2010). "Bimatoprost in the treatment of eyelash hypotrichosis". Clinical Ophthalmology. 4: 349–358. doi:10.2147/opth.s6480. PMC 2861943. PMID 20463804.
- Tosti A, Pazzaglia M, Voudouris S, Tosti G (November 2004). "Hypertrichosis of the eyelashes caused by bimatoprost". Journal of the American Academy of Dermatology. 51 (5 Suppl): S149 – S150. doi:10.1016/j.jaad.2004.05.002. PMID 15577756.
- Wand M (September 1997). "Latanoprost and hyperpigmentation of eyelashes". Archives of Ophthalmology. 115 (9): 1206–1208. doi:10.1001/archopht.1997.01100160376025. PMID 9298071.
- Banaszek A (October 2011). "Company profits from side effects of glaucoma treatment". CMAJ. 183 (14): E1058. doi:10.1503/cmaj.109-3919. PMC 3185096. PMID 21876012.
- Blume-Peytavi U, Lönnfors S, Hillmann K, Garcia Bartels N (May 2012). "A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair growth and pigmentation in healthy volunteers with androgenetic alopecia". Journal of the American Academy of Dermatology. 66 (5): 794–800. doi:10.1016/j.jaad.2011.05.026. PMID 21875758.
- Johnstone MA, Albert DM (August 2002). "Prostaglandin-induced hair growth". Survey of Ophthalmology. 47 (Suppl 1): S185 – S202. doi:10.1016/s0039-6257(02)00307-7. PMID 12204716.
- Roseborough I, Lee H, Chwalek J, Stamper RL, Price VH (April 2009). "Lack of efficacy of topical latanoprost and bimatoprost ophthalmic solutions in promoting eyelash growth in patients with alopecia areata". Journal of the American Academy of Dermatology. 60 (4): 705–706. doi:10.1016/j.jaad.2008.08.029. PMID 19293023.
- Ross EK, Bolduc C, Lui H, Shapiro J (December 2005). "Lack of efficacy of topical latanoprost in the treatment of eyebrow alopecia areata". Journal of the American Academy of Dermatology. 53 (6): 1095–1096. doi:10.1016/j.jaad.2005.06.031. PMID 16310083.
- Castro RF, Azzalis LA, Feder D, Perazzo FF, Pereira EC, Junqueira VB, et al. (December 2012). "Safety and efficacy analysis of liposomal insulin-like growth factor-1 in a fluid gel formulation for hair-loss treatment in a hamster model". Clinical and Experimental Dermatology. 37 (8): 909–912. doi:10.1111/j.1365-2230.2012.04441.x. PMID 22924775. S2CID 21021986.
- ^ Yoon SY, Kim KT, Jo SJ, Cho AR, Jeon SI, Choi HD, et al. (2011). Najbauer J (ed.). "Induction of hair growth by insulin-like growth factor-1 in 1,763 MHz radiofrequency-irradiated hair follicle cells". PLOS ONE. 6 (12): e28474. Bibcode:2011PLoSO...628474Y. doi:10.1371/journal.pone.0028474. PMC 3229574. PMID 22164296.
- Zhao J, Harada N, Kurihara H, Nakagata N, Okajima K (March 2011). "Dietary isoflavone increases insulin-like growth factor-I production, thereby promoting hair growth in mice". The Journal of Nutritional Biochemistry. 22 (3): 227–233. doi:10.1016/j.jnutbio.2010.01.008. PMID 20576422.
- Okajima K, Harada N (2008). "Promotion of insulin-like growth factor-I production by sensory neuron stimulation; molecular mechanism(s) and therapeutic implications". Current Medicinal Chemistry. 15 (29): 3095–3112. doi:10.2174/092986708786848604. PMID 19075656.
- Kwack MH, Shin SH, Kim SR, Im SU, Han IS, Kim MK, et al. (June 2009). "l-Ascorbic acid 2-phosphate promotes elongation of hair shafts via the secretion of insulin-like growth factor-1 from dermal papilla cells through phosphatidylinositol 3-kinase". The British Journal of Dermatology. 160 (6): 1157–1162. doi:10.1111/j.1365-2133.2009.09108.x. PMID 19416266. S2CID 42210965.
- Garza LA, Yang CC, Zhao T, Blatt HB, Lee M, He H, et al. (February 2011). "Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells". The Journal of Clinical Investigation. 121 (2): 613–622. doi:10.1172/JCI44478. PMC 3026732. PMID 21206086.
- Balañá ME, Charreau HE, Leirós GJ (May 2015). "Epidermal stem cells and skin tissue engineering in hair follicle regeneration". World Journal of Stem Cells. 7 (4): 711–727. doi:10.4252/wjsc.v7.i4.711. PMC 4444612. PMID 26029343.
- Takagi R, Ishimaru J, Sugawara A, Toyoshima KE, Ishida K, Ogawa M, et al. (April 2016). "Bioengineering a 3D integumentary organ system from iPS cells using an in vivo transplantation model". Science Advances. 2 (4): e1500887. Bibcode:2016SciA....2E0887T. doi:10.1126/sciadv.1500887. PMC 4820374. PMID 27051874.
- Weintraub, Arlene (June 28, 2011). "Curis and Genentech Unleash the Hedgehog to Fight a Deadly Skin Cancer". Xconomy. Archived from the original on November 9, 2016. Retrieved November 9, 2016.
- "Curis Form 8-K: Termination of a Material Definitive Agreement". SEC EDGAR. May 10, 2007. Archived from the original on November 9, 2016. Retrieved November 9, 2016.
- Pasternack SM, von Kügelgen I, Al Aboud K, Lee YA, Rüschendorf F, Voss K, et al. (March 2008). "G protein-coupled receptor P2Y5 and its ligand LPA are involved in maintenance of human hair growth". Nature Genetics. 40 (3): 329–334. doi:10.1038/ng.84. PMID 18297070. S2CID 20241237.
- Shimomura Y, Wajid M, Ishii Y, Shapiro L, Petukhova L, Gordon D, Christiano AM (March 2008). "Disruption of P2RY5, an orphan G protein-coupled receptor, underlies autosomal recessive woolly hair". Nature Genetics. 40 (3): 335–339. doi:10.1038/ng.100. PMID 18297072. S2CID 1635950.
- Sprecher E (March 2008). "Disentangling the roots of inherited hair disorders". Nature Genetics. 40 (3): 265–266. doi:10.1038/ng0308-265. PMID 18305473. S2CID 2311629.
- Kiso M, Tanaka S, Saba R, Matsuda S, Shimizu A, Ohyama M, et al. (June 2009). "The disruption of Sox21-mediated hair shaft cuticle differentiation causes cyclic alopecia in mice". Proceedings of the National Academy of Sciences of the United States of America. 106 (23): 9292–9297. Bibcode:2009PNAS..106.9292K. doi:10.1073/pnas.0808324106. PMC 2695080. PMID 19470461.
- Kawaminami S, Breakspear S, Saga Y, Noecker B, Masukawa Y, Tsuchiya M, et al. (December 2012). "Deletion of the Sox21 gene drastically affects hair lipids". Experimental Dermatology. 21 (12): 974–976. doi:10.1111/exd.12050. PMID 23171466. S2CID 205126099.
External links
- "Medical Treatments for Balding in Men", April 1999, American Family Physician (medical journal)
| Other dermatological preparations (D11) | |
|---|---|
| Anti-seborrheics | |
| Skin lightening | |
| Skin darkening | |
| Anti-inflammatories | |
| Alopecia treatments | |
| Hair growth inhibitors | |
| Others |
|