| Revision as of 14:14, 20 February 2010 editESCapade (talk | contribs)147 editsm +id intwk← Previous edit | Revision as of 16:43, 21 February 2010 edit undoPtbotgourou (talk | contribs)66,481 editsm robot Modifying: id:Senyawa siklikNext edit → | ||
| Line 44: | Line 44: | ||
| ] | ] | ||
| ] | |||
| ] | ] | ||
| ] | ] | ||
| ] | |||
| ] | ] | ||
| ] | ] | ||
Revision as of 16:43, 21 February 2010
In organic chemistry, a cyclic compound is a compound in which a series of carbon atoms are connected to form a loop or ring. Benzene is a well known example. The term "polycyclic" is used when more than one ring is formed in a single molecule for instance in naphthalene, and the term macrocycle is used for a ring containing more than a dozen atoms.
-
 Benzene, a simple cyclic compound.
Benzene, a simple cyclic compound.
-
 Naphthalene, a simple polycyclic compound.
Naphthalene, a simple polycyclic compound.
-
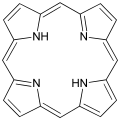 Porphyrin, a simple macrocyclic compound.
Porphyrin, a simple macrocyclic compound.
Categorization
Cyclic compounds can be categorized:
Ring-closing & opening reactions

Related concepts in organic chemistry are so-called ring-closing reactions in which a cyclic compound is formed and ring-opening reactions in which rings are opened.
Examples of ring-closing reactions:
- Ring-closing metathesis
- Nazarov cyclization reaction
- Ruzicka large ring synthesis
- Dieckmann condensation
- Wenker synthesis
Example of ring-opening reactions:
- A general type of polymerization reaction: Ring-opening polymerization
- Ring opening metathesis polymerisation
External links
- Polycyclic+Compounds at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Macrocyclic+Compounds at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
References
- March, Jerry (1985). Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (3rd ed.). New York: Wiley. ISBN 9780471854722. OCLC 642506595.