This is an old revision of this page, as edited by 220.255.2.117 (talk) at 04:00, 8 August 2011. The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
Revision as of 04:00, 8 August 2011 by 220.255.2.117 (talk)(diff) ← Previous revision | Latest revision (diff) | Newer revision → (diff) Chemical element with atomic number 89 (Ac)Actinium (/ækˈtɪniəm/ ak-TIN-nee-əm) is a radioactive chemical element with the symbol Ac and atomic number 89, which was discovered in 1899. It was the first non-primordial radioactive element to be isolated. Polonium, radium and radon were observed before actinium, but they were not isolated until 1902. Actinium gave the name to the actinide series, a group of 15 similar elements between actinium and lawrencium in the periodic table.
A soft, silvery-white radioactive metal, actinium reacts rapidly with oxygen and moisture in air forming a white coating of actinium oxide that prevents further oxidation. As most lanthanides and actinides, actinium assumes oxidation state +3 in nearly all its chemical compounds. Actinium is found only in traces in uranium ores as Ac isotope, which decays with a half-life of 21.773 years, predominantly emitting beta particles. One tonne of uranium ore contains about 0.2 milligrams of actinium. The close similarity of physical and chemical properties of actinium and lanthanum makes separation of actinium from the ore impractical. Instead, the element is prepared, in milligram amounts, by the neutron irradiation of Template:Radium in a nuclear reactor. Owing to its scarcity, high price and radioactivity, actinium has no significant industrial use. Its current applications include a neutron source and an agent for radiation therapy targeting cancer cells in the body.
History
André-Louis Debierne, a French chemist, announced the discovery of a new element in 1899. He separated it from pitchblende residues left by Marie and Pierre Curie after they had extracted radium. In 1899, Debierne described the substance as similar to titanium and (in 1900) as similar to thorium. Friedrich Oskar Giesel independently discovered actinium in 1902 as a substance being similar to lanthanum and called it "emanium" in 1904. After a comparison of substances in 1904, Debierne's name was retained because it had seniority.
The stated history of the discovery of actinium remained uncertain for decades. Articles published in the 1970s and later suggest that Debierne's results published in 1904 conflict with those reported in 1899 and 1900. Whether Debierne and Giesel should share the merit of discover or if Giesel alone should be credited with the discovery is still under debate.
The name actinium originates from the Ancient Greek aktis, aktinos (ακτίς, ακτίνος), meaning beam or ray. Its symbol Ac is also used in abbreviations of other compounds that have nothing to do with actinium, such as acetyl, acetate and acetaldehyde.
Properties
Actinium is a soft, silvery-white, radioactive, metallic element. Its estimated shear modulus is similar to that of lead. Owing to its strong radioactivity, actinium glows in the dark with a pale blue light. Actinium has similar chemical properties as lanthanum and other lanthanides, and therefore these elements are difficult to separate when extracting from uranium ores. Solvent extraction and ion chromatography are commonly used for the separation.
The first element of the actinides, actinium gave the group its name, much as lanthanum has done for the lanthanides. The group of elements is more diverse than the lanthanides and therefore it was not until 1945 that Glenn T. Seaborg proposed the most significant change to Mendeleev's periodic table, by introducing the actinides.
Actinium reacts rapidly with oxygen and moisture in air forming a white coating of actinium oxide that prevents further oxidation. As with most lanthanides and actinides, actinium exists in the oxidation state +3, and the Ac ions are colorless in solutions. The oxidation state +3 originates from the 6d7s electronic configuration of actinium, that is it easily donates 3 electrons assuming a stable closed-shell structure of the noble gas radon. The oxidation state +2 is only known for actinium dihydride.
Chemical compounds
Only a limited number of actinium compounds are known including AcF3, AcCl3, AcBr3, AcOF, AcOCl, AcOBr, Ac2S3, Ac2O3 and AcPO4. Except for AcPO4, they are all similar to the corresponding lanthanum compounds and contain actinium in the oxidation state +3. In particular, the lattice constants of the analogous lanthanum and actinium compounds differ by only a few percent.
| Formula | color | symmetry | space group | No | Pearson symbol | a (pm) | b (pm) | c (pm) | Z | density, g/cm |
|---|---|---|---|---|---|---|---|---|---|---|
| Ac | silvery | fcc | Fm3m | 225 | cF4 | 531.1 | 531.1 | 531.1 | 4 | 10.07 |
| AcH2 | cubic | Fm3m | 225 | cF12 | 567 | 567 | 567 | 4 | 8.35 | |
| Ac2O3 | white | trigonal | P3m1 | 164 | hP5 | 408 | 408 | 630 | 1 | 9.18 |
| Ac2S3 | cubic | I43d | 220 | cI28 | 778.56 | 778.56 | 778.56 | 4 | 6.71 | |
| AcF3 | white | hexagonal | P3c1 | 165 | hP24 | 741 | 741 | 755 | 6 | 7.88 |
| AcCl3 | hexagonal | P63/m | 165 | hP8 | 764 | 764 | 456 | 2 | 4.8 | |
| AcBr3 | white | hexagonal | P63/m | 165 | hP8 | 764 | 764 | 456 | 2 | 5.85 |
| AcOF | white | cubic | Fm3m | 593.1 | 8.28 | |||||
| AcOCl | tetragonal | 424 | 424 | 707 | 7.23 | |||||
| AcOBr | tetragonal | 427 | 427 | 740 | 7.89 | |||||
| AcPO4·0.5H2O | hexagonal | 721 | 721 | 664 | 5.48 |
Here a, b and c are lattice constants, No is space group number and Z is the number of formula units per unit cell. Density was not measured directly but calculated from the lattice parameters.
Oxides
Actinium oxide (Ac2O3) can be obtained by heating the hydroxide at 500 °C or the oxalate at 1100 °C, in vacuum. It crystal lattice is isotypic with the oxides of most trivalent rare-earth metals.
Halides
Actinium trifluoride can be produced either in solution or in solid reaction. The former reaction is carried out at room temperature, by adding hydrofluoric acid to a solution containing actinium ions. In the latter method, actinium metal is treated with hydrogen fluoride vapors at 700 °C in an all-platinum setup. Treating actinium trifluoride with ammonium hydroxide at 900–1000 °C yields oxyfluoride AcOF. Whereas lanthanum oxyfluoride can be easily obtained by burning lanthanum trifluoride in air at 800 °C for an hour, similar treatment of actinium trifluoride yields no AcOF and only results in melting of the initial product.
- AcF3 + 2 NH3 + H2O → AcOF + 2 NH4F
Actinium trichloride is obtained by reacting actinium hydroxide or oxalate with carbon tetrachloride vapors at temperatures above 960 °C. Similar to oxyfluoride, actinium oxychloride can be prepared by hydrolyzing actinium trichloride with ammonium hydroxide at 1000 °C. However, in contrast to the oxyfluoride, the oxychloride could well be synthesized by igniting a solution of actinium trichloride in hydrochloric acid with ammonia.
Reaction of aluminium bromide and actinium oxide yields actinium tribromide:
- Ac2O3 + 2 AlBr3 → 2 AcBr3 + Al2O3
and treating it with ammonium hydroxide at 500 °C results in the oxybromide AcOBr.
Other compounds
Actinium hydride was obtained by reduction of actinium trichloride with potassium at 300 °C, and its structure was deduced by analogy with the corresponding LaH2 hydride. The source of hydrogen in the reaction was uncertain.
Mixing monosodium phosphate (NaH2PO4) with a solution of actinium in hydrochloric acid yields white-colored actinium phosphate hemihydrate (AcPO4·0.5H2O), and heating actinium oxalate with hydrogen sulfide vapors at 1400 °C for a few minutes results in a black actinium sulfide Ac2S3. It may possibly be produced by acting with a mixture of hydrogen sulfide and carbon disulfide on actinium oxide at 1000 °C.
Isotopes
Main article: Isotopes of actiniumNaturally occurring actinium is composed of one radioactive isotope;
Ac. Thirty-six radioisotopes have been identified, the most stable being
Ac with a half-life of 21.772 years,
Ac with a half-life of 10.0 days and
Ac with a half-life of 29.37 hours. All remaining radioactive isotopes have half-lives that are less than 10 hours and the majority of them have half-lives shorter than 1 minute. The shortest-lived known isotope of actinium is
Ac (half-life of 69 nanoseconds) which decays through alpha decay and electron capture. Actinium also has two meta states.
Purified
Ac comes into equilibrium with its decay products at the end of 185 days. It decays according to its 21.773-year half-life emitting mostly beta (98.8%) and some alpha particles (1.2%); the successive decay products are part of the actinium series. Owing to the low available amounts, low energy of its beta particles (46 keV) and low intensity of alpha radiation,
Ac is difficult to detect directly by its emission and it is therefore traced via its decay products. The isotopes of actinium range in atomic weight from 206 u (
Ac) to 236 u (
Ac).
| Isotope | Production | Decay | Half-life |
|---|---|---|---|
| Ac | Th(d,9n)Pa(α)→Ac | α | 52 ms |
| Ac | Th(d,8n)Pa(α)→Ac | α | 5.0 s |
| Ac | Th(d,7n)Pa(α)→Ac | α | 2.1 min |
| Ac | Th(d,6n)Pa(α)→Ac | α | 2.78 hours |
| Ac | Th(n,γ)Th(β)→Pa(β)→U(α)→Th(α)→Ra(β)Ac | α | 10 days |
| Ac | Ra(d,2n)Ac | α, β electron capture |
29.37 hours |
| Ac | U(α)→Th(β)→Pa(α)→Ac | α, β | 21.77 years |
| Ac | Th(α)→Ra(β)→Ac | β | 6.15 hours |
| Ac | Ra(n,γ)Ra(β)→Ac | β | 62.7 min |
| Ac | Th(d,α)Ac | β | 122 s |
| Ac | Th(γ,p)Ac | β | 7.5 min |
| Ac | Th(n,p)Ac | β | 119 s |
Occurrence and synthesis

Actinium is found only in traces in uranium ores as Ac – one tonne of ore contains about 0.2 milligrams of actinium. These uranium ores sometimes contain lanthanum and other lanthanides. The actinium isotope Ac is a transient member of the actinium series decay chain, which begins with the parent isotope U (or Pu) and ends with the stable lead isotope Pb. Another actinium isotope (Ac) is transiently present in the neptunium series decay chain, beginning with Np (or U) and ending with near-stable bismuth (Bi).
The low natural concentration, and the close similarity of physical and chemical properties to those of lanthanum and other lanthanides, render separation of actinium from the ore impractical, and complete separation was never achieved. Instead, actinium is prepared, in milligram amounts, by the neutron irradiation of Template:Radium in a nuclear reactor.
The reaction yield is about 2% of the radium weight. Ac can further capture neutrons resulting in small amounts of Ac. After the synthesis, actinium is separated from radium and from the products of decay and nuclear fusion, such as thorium, polonium, lead and bismuth. The extraction can be performed with thenoyltrifluoroacetone-benzene solution from an aqueous solution of the radiation products, and the selectivity to a certain element is achieved by adjusting the pH (to about 6.0 for actinium). An alternative procedure is anion exchange with an appropriate resin in nitric acid, which can result in a separation factor of 1,000,000 for radium and actinium vs. thorium in a two-stage process. Actinium can then be separated from radium, with a ratio of about 100, using a low cross-linking cation exchange resin and nitric acid as eluant.
Ac was first produced artificially at the Institute for Transuranium Elements (ITU) in Germany using a cyclotron and at St George Hospital in Sydney using a linac in 2000. This rare isotope has potential applications in radiation therapy and is most efficiently produced by bombarding a radium-226 target with 20–30 MeV deuterium ions. This reaction also yields Ac which however decays with a half-life of 29 hours and thus does not contaminate Ac.
Actinium metal has been prepared by the reduction of actinium fluoride with lithium vapor in vacuum at a temperature between 1100 and 1300 °C. Higher temperatures resulted in evaporation of the product and lower ones lead to an incomplete transformation. Lithium was chosen among other alkali metals because its fluoride is most volatile.
Applications
Owing to its scarcity, high price and radioactivity, actinium currently has no significant industrial use, however, potential applications are being developed.
Ac is highly radioactive and was therefore studied for use as an active element of radioisotope thermoelectric generators, for example in spacecraft. The oxide of Ac pressed with beryllium is also an efficient neutron source with the activity exceeding that of the standard americium-beryllium and radium-beryllium pairs. In all those applications, Ac (a beta source) is merely a progenitor which generates alpha-emitting isotopes upon its decay. Beryllium captures alpha particles and emits neutrons owing to its large cross-section for the (α,n) nuclear reaction:
The AcBe neutron sources can be applied in a neutron probe – a standard device for measuring the quantity of water present in soil, as well as moisture/density for quality control in highway construction. Such probes are also used in well logging applications, in neutron radiography, tomography and other radiochemical investigations.
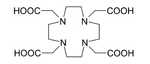
Ac is applied in medicine to produce Template:Bismuth in a reusable generator or can be used alone as an agent for radiation therapy, in particular targeted alpha therapy (TAT). This isotope has a half-life of 10 days that makes it much more suitable for radiation therapy than Bi (half-life 46 minutes). Not only Ac itself, but also its decay products emit alpha particles which kill cancer cells in the body. The major difficulty with application of Ac was that intravenous injection of simple actinium complexes resulted in their accumulation in the bones and liver for a period of tens of years. As a result, after the cancer cells were quickly killed by alpha particles from Ac, the radiation from the actinium and its decay products might induce new mutations. To solve this problem, Ac was bound to a chelating agent, such as citrate, ethylenediaminetetraacetic acid (EDTA) or diethylene triamine pentaacetic acid (DTPA). This reduced actinium accumulation in the bones, but the excretion from the body remained slow. Much better results were obtained with such chelating agents as HEHA or DOTA (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid) coupled to trastuzumab, a monoclonal antibody that interferes with the HER2/neu receptor. The latter delivery combination was tested on mice and proved to be effective against leukemia, lymphoma, breast, ovarian, neuroblastoma and prostate cancers.
The medium half-life of Ac (21.77 years) makes it very convenient radioactive isotope in modeling the slow vertical mixing of oceanic waters. The associated processes cannot be studied with the required accuracy by direct measurements of current velocities (of the order 50 meters per year). However, evaluation of the concentration depth-profiles for different isotopes allows estimating the mixing rates. The physics behind this method is as follows: oceanic waters contain homogeneously dispersed U. Its decay product, Pa, gradually precipitates to the bottom, so that its concentration first increases with depth and then stays nearly constant. Pa decays to Ac; however, the concentration of the latter isotope does not follow the Pa depth profile, but instead increases toward the sea bottom. This occurs because of the mixing processes which raise some additional Ac from the sea bottom. Thus analysis of both Pa and Ac depth profiles allows to model the mixing behavior.
Precautions
Ac is highly radioactive and experiments with it are carried out in a specially designed laboratory equipped with a glove box and radiation shielding. When actinium trichloride is administered intravenously to rats, about 33% of actinium is deposited into the bones and 50% into the liver. Its toxicity is comparable, but slightly lower than that of americium and plutonium.
See also
Portal:Actinium at Misplaced Pages's sister projects: Definitions from Wiktionary
Definitions from Wiktionary Media from Commons
Media from Commons
References
- Wall, Greg (8 September 2003). "C&EN: It's Elemental: The Periodic Table - Actinium". C&EN: It's Elemental: The Periodic Table. Chemical and Engineering News. Retrieved 2 June 2011.
- ^ Kirby, Harold W.; Morss, Lester R. (2006). "Actinium". The Chemistry of the Actinide and Transactinide Elements. p. 18. doi:10.1007/1-4020-3598-5_2. ISBN 978-1-4020-3555-5.
- Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3): 030001. doi:10.1088/1674-1137/abddae.
- Debierne, André-Louis (1899). "Sur un nouvelle matière radio-active". Comptes rendus (in French). 129: 593–595.
- Debierne, André-Louis (1900–1901). "Sur un nouvelle matière radio-actif – l'actinium". Comptes rendus (in French). 130: 906–908.
- Giesel, Friedrich Oskar (1902). "Ueber Radium und radioactive Stoffe". Berichte der Deutschen Chemische Geselschaft (in German). 35 (3): 3608–3611. doi:10.1002/cber.190203503187.
- Giesel, Friedrich Oskar (1904). "Ueber den Emanationskörper (Emanium)". Berichte der Deutschen Chemische Geselschaft (in German). 37 (2): 1696–1699. doi:10.1002/cber.19040370280.
- Debierne, André-Louis (1904). "Sur l'actinium". Comptes rendus (in French). 139: 538–540.
- Giesel, Friedrich Oskar (1904). "Ueber Emanium". Berichte der Deutschen Chemische Geselschaft (in German). 37 (2): 1696–1699. doi:10.1002/cber.19040370280.
- Giesel, Friedrich Oskar (1905). "Ueber Emanium". Berichte der Deutschen Chemische Geselschaft (in German). 38 (1): 775–778. doi:10.1002/cber.190503801130.
- Kirby, H. W. (1971). "The Discovery of Actinium". Isis. 62 (3): 290–308. doi:10.1086/350760. JSTOR 229943.
- Adloff, J. P. (2000). "The centenary of a controversial discovery: actinium". Radiochim. Acta. 88 (3–4_2000): 123–128. doi:10.1524/ract.2000.88.3-4.123.
- Kirby, Harold W.; Morss, Lester R. (2006). "Actinium": 18. doi:10.1007/1-4020-3598-5_2.
{{cite journal}}: Cite journal requires|journal=(help) - ^ C. R. Hammond. The Elements, in Handbook of Chemistry and Physics 81st edition. CRC press. ISBN 0849304857.
- ^ Stites, Joseph G.; Salutsky, Murrell L.; Stone, Bob D. (1955). "Preparation of Actinium Metal". J. Am. Chem. Soc. 77 (1): 237–240. doi:10.1021/ja01606a085.
- ^ "Actinium" Encyclopedia Britannica, 15th edition, 1995, p. 70
- Frederick Seitz, David Turnbull Solid state physics: advances in research and applications, Academic Press, 1964 ISBN 0126077169 pp. 289–291
- Katz, J. J.; Manning, W M (1952). "Chemistry of the Actinide Elements Annual Review of Nuclear Science". Annual Review of Nuclear Science. 1: 245–262. doi:10.1146/annurev.ns.01.120152.001333.
- Seaborg, Glenn T. (1946). "The Transuranium Elements". Science. 104 (2704): 379–386. doi:10.1126/science.104.2704.379. JSTOR 1675046. PMID 17842184.
- ^ Actinium, Great Soviet Encyclopedia (in Russian)
- Sherman, Fried; Hagemann, French; Zachariasen, W. H. (1950). "The Preparation and Identification of Some Pure Actinium Compounds". Journal of the American Chemical Society. 72 (2): 771–775. doi:10.1021/ja01158a034.
- ^ Fried, Sherman; Hagemann, French; Zachariasen, W. H. (1950). "The Preparation and Identification of Some Pure Actinium Compounds". Journal of the American Chemical Society. 72 (2): 771. doi:10.1021/ja01158a034.
- ^ Farr, J (1961). "The crystal structure of actinium metal and actinium hydride". Journal of Inorganic and Nuclear Chemistry. 18: 42. doi:10.1016/0022-1902(61)80369-2.
- ^ Zachariasen, W. H. (1949). "Crystal chemical studies of the 5f-series of elements. XII. New compounds representing known structure types". Acta Crystallographica. 2 (6): 388. doi:10.1107/S0365110X49001016.
- Zachariasen, W. H. (1949). "Crystal chemical studies of the 5f-series of elements. VI. The Ce2S3-Ce3S4 type of structure". Acta Crystallographica. 2: 57. doi:10.1107/S0365110X49000126.
- Meyer, p. 71
- ^ Zachariasen, W. H. (1948). "Crystal chemical studies of the 5f-series of elements. I. New structure types". Acta Crystallographica. 1 (5): 265. doi:10.1107/S0365110X48000703.
- ^ Meyer, pp. 87–88
- Meyer, p. 43
- ^ Audi, Georges (2003). "The NUBASE Evaluation of Nuclear and Decay Properties". Nuclear Physics A. 729. Atomic Mass Data Center: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001.
- ^ Hagemann, French (1950). "The Isolation of Actinium". Journal of the American Chemical Society. 72 (2): 768. doi:10.1021/ja01158a033.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 946. ISBN 978-0-08-037941-8.
- ^ Bolla, Rose A.; Malkemus, D; Mirzadeh, S (2005). "Production of actinium-225 for alpha particle mediated radioimmunotherapy". Applied Radiation and Isotopes. 62 (5): 667–679. doi:10.1016/j.apradiso.2004.12.003. PMID 15763472.
- Melville, G; Allen, Bj (2009). "Cyclotron and linac production of Ac-225". Applied radiation and isotopes. 67 (4): 549–55. doi:10.1016/j.apradiso.2008.11.012. PMID 19135381.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Pamela J. Russell, Paul Jackson, Elizabeth Anne Kingsley Prostate cancer methods and protocols, Humana Press, 2003, ISBN 0896039781, p. 336
- Alan M. Russell, Kok Loong Lee Structure-property relations in nonferrous metals, Wiley, 2005, ISBN 047164952X, pp. 470–471
- D. K. Majumdar Irrigation Water Management: Principles and Practice, 2004 ISBN 8120317297 p. 108
- H. Chandrasekharan, Navindu Gupta Fundamentals of Nuclear Science – Application in Agriculture, 2006 ISBN 8172112009 pp. 202 ff
- Dixon, W.R. (1957). "Neutron Spectrum of an Actinium–Beryllium Source". Can. J. Phys./Rev. Can. Phys. 35 (6): 699–702.
- "Improved in Vivo Stability of Actinium-225 Macrocyclic Complexes".
{{cite journal}}: Cite journal requires|journal=(help) - McDevitt, MR; Ma, D; Lai, LT; Simon, J; Borchardt, P; Frank, RK; Wu, K; Pellegrini, V; Curcio, MJ (2001). "Tumor Therapy with Targeted Atomic Nanogenerators" (PDF). Science. 294 (5546): 1537–40. doi:10.1126/science.1064126. PMID 11711678.
- Borchardt, Paul E.; et al. (2003). "Targeted Actinium-225 in Vivo Generators for Therapy of Ovarian Cancer" (PDF). Cancer Research. 63 (16): 5084–5090. PMID 12941838.
{{cite journal}}: Explicit use of et al. in:|author=(help) - Ballangrud, AM; Yang, WH; Palm, S; Enmon, R; Borchardt, PE; Pellegrini, VA; McDevitt, MR; Scheinberg, DA; Sgouros, G (2004). "Alpha-particle emitting atomic generator (Actinium-225)-labeled trastuzumab (herceptin) targeting of breast cancer spheroids: efficacy versus HER2/neu expression". Clinical cancer research : an official journal of the American Association for Cancer Research. 10 (13): 4489–97. doi:10.1158/1078-0432.CCR-03-0800. PMID 15240541.
- Nozaki, Yoshiyuki (1984). "Excess 227Ac in deep ocean water". Nature. 310 (5977): 486. Bibcode:1984Natur.310..486N. doi:10.1038/310486a0.
- Geibert, W.; Rutgers Van Der Loeff, M.M.; Hanfland, C.; Dauelsberg, H.-J. (2002). "Actinium-227 as a deep-sea tracer: sources, distribution and applications". Earth and Planetary Science Letters. 198: 147. Bibcode:2002E&PSL.198..147G. doi:10.1016/S0012-821X(02)00512-5.
- Langham, W.; Storer, J. (1952). "Toxicology of Actinium Equilibrium Mixture". Los Alamos Scientific Lab.: Technical Report. doi:10.2172/4406766.
Bibliography
- Meyer, Gerd and Morss, Lester R. Synthesis of lanthanide and actinide compounds, Springer, 1991, ISBN 0792310187
External links
- NLM Hazardous Substances Databank – Actinium, Radioactive
- Actinium in Haire, Richard G. (2006). Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer. ISBN 1-4020-3555-1.
| Periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Template:Link GA Template:Link FA
Categories:



