| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name 1,3-Thiazole | |||
| Other names Thiazole | |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.475 | ||
| PubChem CID | |||
| UNII | |||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
| Chemical formula | C3H3NS | ||
| Molar mass | 85.12 g·mol | ||
| Boiling point | 116 to 118 °C (241 to 244 °F; 389 to 391 K) | ||
| Acidity (pKa) | 2.5 (of conjugate acid) | ||
| Magnetic susceptibility (χ) | -50.55·10 cm/mol | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Thiazole (/ˈθaɪ.əzoʊl/), or 1,3-thiazole, is a 5-membered heterocyclic compound that contains both sulfur and nitrogen. The term 'thiazole' also refers to a large family of derivatives. Thiazole itself is a pale yellow liquid with a pyridine-like odor and the molecular formula C3H3NS. The thiazole ring is notable as a component of the vitamin thiamine (B1).
Molecular and electronic structure
Thiazoles are members of the azoles, heterocycles that include imidazoles and oxazoles. Thiazole can also be considered a functional group when part of a larger molecule.
Being planar thiazoles are characterized by significant pi-electron delocalization and have some degree of aromaticity, more so than the corresponding oxazoles. This aromaticity is evidenced by the H NMR chemical shift of the ring protons, which absorb between 7.27 and 8.77 ppm, indicating a strong diamagnetic ring current. The calculated pi-electron density marks C5 as the primary site for electrophilic substitution, and C2-H as susceptible to deprotonation.
Occurrence of thiazoles and thiazolium salts
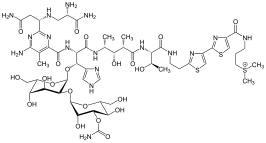
Thiazoles are found in a variety of specialized products, often fused with benzene derivatives, the so-called benzothiazoles. In addition to vitamin B1, the thiazole ring is found in epothilone. Other important thiazole derivatives are benzothiazoles, for example, the firefly chemical luciferin. Whereas thiazoles are well represented in biomolecules, oxazoles are not. It is found in naturally occurring peptides, and utilised in the development of peptidomimetics (i.e. molecules that mimic the function and structure of peptides).
Commercial significant thiazoles include mainly dyes and fungicides. Thifluzamide, Tricyclazole, and Thiabendazole are marketed for control of various agricultural pests. Another widely used thiazole derivative is the non-steroidal anti-inflammatory drug Meloxicam. The following anthroquinone dyes contain benzothiazole subunits: Algol Yellow 8 (CAS# ), Algol Yellow GC (CAS# ), Indanthren Rubine B (CAS# ), Indanthren Blue CLG (CAS# , and Indanthren Blue CLB (CAS#). These thiazole dye are used for dyeing cotton.
Synthesis
Various laboratory methods exist for the organic synthesis of thiazoles. Prominent is the Hantzsch thiazole synthesis, which is a reaction between haloketones and thioamides. For example, 2,4-dimethylthiazole is synthesized from thioacetamide and chloroacetone. In the Cook-Heilbron synthesis, thiazoles arise by the condensation of α-aminonitrile with carbon disulfide. Thiazoles can be accessed by acylation of 2-aminothiolates, often available by the Herz reaction.
Biosynthesis
Thiazoles are generally formed via reactions of cysteine, which provides the N-C-C-S backbone of the ring. Thiamine does not fit this pattern however. Several biosynthesis routes lead to the thiazole ring as required for the formation of thiamine. Sulfur of the thiazole is derived from cysteine. In anaerobic bacteria, the CN group is derived from dehydroglycine.
Reactions
With a pKa of 2.5 for the conjugate acid, thiazoles are far less basic than imidazole (pKa =7).
Deprotonation with strong bases occurs at C2-H. The negative charge on this position is stabilized as an ylide. Hauser bases and organolithium compounds react at this site, replacing the proton. 2-Lithiothiazoles are also generated by metal-halogen exchange from 2-bromothiazole.
Electrophilic aromatic substitution at C5 but require activating groups such as a methyl group, as illustrated in bromination:
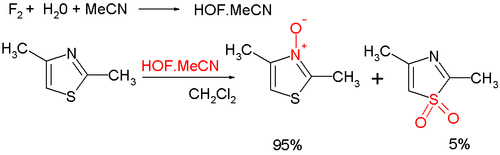
Oxidation at nitrogen gives the aromatic thiazole N-oxide; many oxidizing agents exist, such as mCPBA; a novel one is hypofluorous acid prepared from fluorine and water in acetonitrile; some of the oxidation takes place at sulfur, leading to non-aromatic sulfoxide/sulfone: Thiazole N-oxides are useful in Palladium-catalysed C-H arylations, where the N-oxide is able to shift the reactivity to reliably favor the 2-position, and allows for these reactions to be carried out under much more mild conditions.
- Thiazoles are formyl synthons; conversion of R-thia to the R-CHO aldehyde takes place with, respectively, methyl iodide (N-methylation), organic reduction with sodium borohydride, and hydrolysis with Mercury(II) chloride in water.
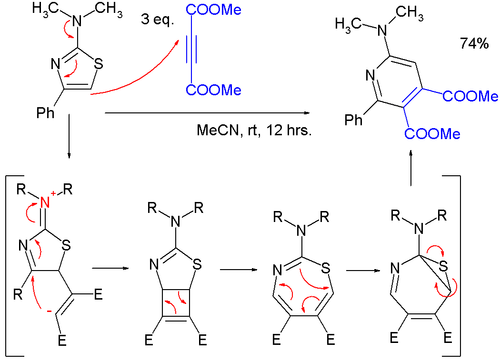
- Thiazoles can react in cycloadditions, but in general at high temperatures due to favorable aromatic stabilization of the reactant; Diels-Alder reactions with alkynes are followed by extrusion of sulfur, and the endproduct is a pyridine; in one study, a very mild reaction of a 2-(dimethylamino)thiazole with dimethyl acetylenedicarboxylate (DMAD) to a pyridine was found to proceed through a zwitterionic intermediate in a formal cycloaddition to a cyclobutene, then to a 1,3-thiazepine in a 4-electron electrocyclic ring opening and then to a 7-thia-2-azanorcaradiene in a 6-electron electrocyclic ring, closing before extruding the sulfur atom.
Thiazolium salts
Alkylation of thiazoles at nitrogen forms a thiazolium cation. Thiazolium salts are catalysts in the Stetter reaction and the Benzoin condensation. Deprotonation of N-alkyl thiazolium salts give the free carbenes and transition metal carbene complexes.
Alagebrium is a thiazolium-based drug.
References
- Zoltewicz, J. A.; Deady, L. W. (1978). "Quaternization of Heteroaromatic Compounds: Quantitative Aspects". Advances in Heterocyclic Chemistry Volume 22. Vol. 22. pp. 71–121. doi:10.1016/S0065-2725(08)60103-8. ISBN 9780120206223.
- Eicher, T.; Hauptmann, S. (2003). The Chemistry of Heterocycles: Structure, Reactions, Syntheses, and Applications. Wiley. ISBN 978-3-527-30720-3.
- Mak, Jeffrey Y. W.; Xu, Weijun; Fairlie, David P. (2015-01-01). Peptidomimetics I (PDF). Topics in Heterocyclic Chemistry. Vol. 48. Springer Berlin Heidelberg. pp. 235–266. doi:10.1007/7081_2015_176. ISBN 978-3-319-49117-2.
- George Schwarz (1945). "2,4-Dimethylthiazole". Organic Syntheses. 25: 35. doi:10.15227/orgsyn.025.0035.
- Kriek, M.; Martins, F.; Leonardi, R.; Fairhurst, S. A.; Lowe, D. J.; Roach, P. L. (2007). "Thiazole Synthase from Escherichia coli: An Investigation of the Substrates and Purified Proteins Required for Activity in vitro" (PDF). J. Biol. Chem. 282 (24): 17413–17423. doi:10.1074/jbc.M700782200. PMID 17403671.
- Thomas L. Gilchrist (1997). Heterocyclic Chemistry (3 ed.). Essex, England: Addison Wesley. p. 414. ISBN 0-582-27843-0.
- ^ Dondoni, A.; Merino, P. (1995). "Diastereoselective Homologation of D-(R)-Glyceraldehyde Acetonide using 2-(Trimethylsilyl)thiazole". 72: 21. doi:10.15227/orgsyn.072.0021.
{{cite journal}}: Cite journal requires|journal=(help) - Amir, E.; Rozen, S. (2006). "Easy Access to the Family of Thiazole N-oxides using HOF·CH3CN". Chemical Communications. 2006 (21): 2262–2264. doi:10.1039/b602594c. PMID 16718323.
- Campeau, Louis-Charles; Bertrand-Laperle, Mégan; Leclerc, Jean-Philippe; Villemure, Elisia; Gorelsky, Serge; Fagnou, Keith (2008-03-01). "C2, C5, and C4 Azole N -Oxide Direct Arylation Including Room-Temperature Reactions". Journal of the American Chemical Society. 130 (11): 3276–3277. doi:10.1021/ja7107068. ISSN 0002-7863. PMID 18302383.
- Alajarín, M.; Cabrera, J.; Pastor, A.; Sánchez-Andrada, P.; Bautista, D. (2006). "On the Cycloaddition of 2-Aminothiazoles and Dimethyl Acetylenedicarboxylate. Experimental and Computational Evidence of a Thermal Disrotatory Ring Opening of Fused Cyclobutenes". J. Org. Chem. 71 (14): 5328–5339. doi:10.1021/jo060664c. PMID 16808523.
- Arduengo, A. J.; Goerlich, J. R.; Marshall, W. J. (1997). "A Stable Thiazol-2-ylidene and Its Dimer". Liebigs Annalen. 1997 (2): 365–374. doi:10.1002/jlac.199719970213.