| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name (Dichloromethyl)benzene | |||
Other names
| |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| Beilstein Reference | 1099407 | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.463 | ||
| EC Number |
| ||
| KEGG | |||
| MeSH | Compounds Benzylidene Compounds | ||
| PubChem CID | |||
| RTECS number |
| ||
| UNII | |||
| UN number | 1886 2810 | ||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
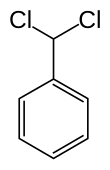
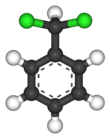
| Chemical formula | C7H6Cl2 | ||
| Molar mass | 161.03 g/mol | ||
| Appearance | Colorless liquid | ||
| Density | 1.254 g/cm, liquid | ||
| Melting point | −17 to −15 °C (1 to 5 °F; 256 to 258 K) | ||
| Boiling point | 205 °C (401 °F; 478 K) (82 °C at 10 mmHg) | ||
| Solubility in water | 0.25 g/L at 39 °C | ||
| Vapor pressure | 0.6 kPa (45 °C) | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms |    
| ||
| Signal word | Danger | ||
| Hazard statements | H302, H315, H318, H331, H335, H351 | ||
| Precautionary statements | P201, P202, P261, P264, P270, P271, P280, P281, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P311, P312, P321, P330, P332+P313, P362, P403+P233, P405, P501 | ||
| Flash point | 93 °C (199 °F; 366 K) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Benzal chloride is an organic compound with the formula C6H5CHCl2. This colourless liquid is a lachrymator and is used as a building block in organic synthesis.
Preparation and usage
Benzal chloride is produced by the free radical chlorination of toluene, being preceded in the process by benzyl chloride (C6H5CH2Cl) and followed by benzotrichloride (C6H5CCl3):
- C6H5CH3 + Cl2 → C6H5CH2Cl + HCl
- C6H5CH2Cl + Cl2 → C6H5CHCl2 + HCl
- C6H5CHCl2 + Cl2 → C6H5CCl3 + HCl
Benzylic halides are typically strong alkylating agents, and for this reason benzal chloride is treated as a hazardous compound.
Treatment of benzal chloride with sodium gives stilbene.
Most benzal chloride main industrial use is as a precursor to benzaldehyde. This conversion involves hydrolysis in the presence of base:
- C6H5CHCl2 + H2O → C6H5CHO + 2 HCl
References
- "BENZAL CHLORIDE". International Programme on Chemical Safety. Retrieved 2007-10-30.
- Lipper, Karl-August; Löser, Eckhard (2011). "Benzyl Chloride and Other Side-Chain Chlorinated Aromatic Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.o04_o01. ISBN 978-3527306732.