Pharmaceutical compound
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Focalin, Focalin XR, others |
| Other names | d-threo-methylphenidate (D-TMP) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603014 |
| License data | |
| Dependence liability | Physical: None; Psychological: High |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 11–52% |
| Protein binding | 30% |
| Metabolism | Liver |
| Elimination half-life | 4 hours |
| Excretion | Kidney |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
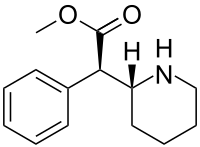
| Formula | C14H19NO2 |
| Molar mass | 233.311 g·mol |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
| (what is this?) (verify) | |
Dexmethylphenidate, sold under the brand name Focalin among others, is a potent central nervous system (CNS) stimulant used to treat attention deficit hyperactivity disorder (ADHD) in those over the age of five years. It is taken by mouth. The immediate release formulation lasts up to five hours while the extended release formulation lasts up to twelve hours. It is the more active enantiomer of methylphenidate.
Common side effects include abdominal pain, loss of appetite, and fever. Serious side effects may include psychosis, sudden cardiac death, mania, anaphylaxis, seizures, and dangerously prolonged erection. Safety during pregnancy and breastfeeding is unclear. Dexmethylphenidate is a central nervous system (CNS) stimulant. How it works in ADHD is unclear.
Dexmethylphenidate was approved for medical use in the United States in 2001. It is available as a generic medication. In 2022, it was the 109th most commonly prescribed medication in the United States, with more than 5 million prescriptions.
Medical uses
Dexmethylphenidate is used as a treatment for ADHD, usually along with psychological, educational, behavioral or other forms of treatment. It is proposed that stimulants help ameliorate the symptoms of ADHD by making it easier for the user to concentrate, avoid distraction, and control behavior. Placebo-controlled trials have shown that once-daily dexmethylphenidate XR was effective and generally well tolerated.
Improvements in ADHD symptoms in children were significantly greater for dexmethylphenidate XR versus placebo. It also showed greater efficacy than osmotic controlled-release oral delivery system (OROS) methylphenidate over the first half of the laboratory classroom day but assessments late in the day favoured OROS methylphenidate.
Contraindications
This section is transcluded from Methylphenidate. (edit | history)Methylphenidate is contraindicated for individuals with agitation, tics, glaucoma, heart defects or a hypersensitivity to any ingredients contained in methylphenidate pharmaceuticals.
Pregnant women are advised to only use the medication if the benefits outweigh the potential risks. Not enough human studies have been conducted to conclusively demonstrate an effect of methylphenidate on fetal development. In 2018, a review concluded that it has not been teratogenic in rats and rabbits, and that it "is not a major human teratogen".
Adverse effects
Part of this section is transcluded from Methylphenidate. (edit | history)Products containing dexmethylphenidate have a side effect profile comparable to those containing methylphenidate.

The most common side effects associated with methylphenidate (in standard and extended-release formulations) are appetite loss, dry mouth, anxiety/nervousness, nausea, and insomnia. Gastrointestinal adverse effects may include abdominal pain and weight loss. Nervous system adverse effects may include akathisia (agitation/restlessness), irritability, dyskinesia (tics), lethargy (drowsiness/fatigue), and dizziness. Cardiac adverse effects may include palpitations, changes in blood pressure, and heart rate (typically mild), and tachycardia (rapid heart rate). Ophthalmologic adverse effects may include blurred vision caused by pupil dilatation and dry eyes, with less frequent reports of diplopia and mydriasis.
Results from a 2024 systematic review showed that methylphenidate significantly improves ADHD symptoms and broadband measures but can cause appetite suppression and other adverse events in children and adolescents. Smokers with ADHD who take methylphenidate may increase their nicotine dependence, and smoke more often than before they began using methylphenidate, with increased nicotine cravings and an average increase of 1.3 cigarettes per day.
There is some evidence of mild reductions in height with prolonged treatment in children. This has been estimated at 1 centimetre (0.4 in) or less per year during the first three years with a total decrease of 3 centimetres (1.2 in) over 10 years.
Hypersensitivity (including skin rash, urticaria, and fever) is sometimes reported when using transdermal methylphenidate. The Daytrana patch has a much higher rate of skin reactions than oral methylphenidate.
Methylphenidate can worsen psychosis in people who are psychotic, and in very rare cases it has been associated with the emergence of new psychotic symptoms. It should be used with extreme caution in people with bipolar disorder due to the potential induction of mania or hypomania. There have been very rare reports of suicidal ideation, but some authors claim that evidence does not support a link. Logorrhea is occasionally reported and visual hallucinations are very rarely reported. Priapism is a very rare adverse event that can be potentially serious.
U.S. Food and Drug Administration-commissioned studies in 2011 indicate that in children, young adults, and adults, there is no association between serious adverse cardiovascular events (sudden death, heart attack, and stroke) and the medical use of methylphenidate or other ADHD stimulants.
Because some adverse effects may only emerge during chronic use of methylphenidate, a constant watch for adverse effects is recommended.
A 2018 Cochrane review found that methylphenidate might be associated with serious side effects such as heart problems, psychosis, and death. The certainty of the evidence was stated as very low.
The same review found tentative evidence that it may cause both serious and non-serious adverse effects in children.
Overdose
The symptoms of a moderate acute overdose of methylphenidate primarily arise from central nervous system overstimulation; these symptoms include: vomiting, nausea, agitation, tremors, hyperreflexia, muscle twitching, euphoria, confusion, hallucinations, delirium, hyperthermia, sweating, flushing, headache, tachycardia, heart palpitations, cardiac arrhythmias, hypertension, mydriasis, and dryness of mucous membranes. A severe overdose may involve symptoms such as hyperpyrexia, sympathomimetic toxidrome, convulsions, paranoia, stereotypy (a repetitive movement disorder), rhabdomyolysis, coma, and circulatory collapse. A methylphenidate overdose is rarely fatal with appropriate care. Following injection of methylphenidate tablets into an artery, severe toxic reactions involving abscess formation and necrosis have been reported.
Treatment of a methylphenidate overdose typically involves the administration of benzodiazepines, with antipsychotics, α-adrenoceptor agonists and propofol serving as second-line therapies.

Addiction and dependence
Methylphenidate is a stimulant with an addiction liability and dependence liability similar to amphetamine. It has moderate liability among addictive drugs; accordingly, addiction and psychological dependence are possible and likely when methylphenidate is used at high doses as a recreational drug. When used above the medical dose range, stimulants are associated with the development of stimulant psychosis.
Biomolecular mechanisms
Further information: Addiction § Biomolecular mechanismsMethylphenidate has the potential to induce euphoria due to its pharmacodynamic effect (i.e., dopamine reuptake inhibition) in the brain's reward system. At therapeutic doses, ADHD stimulants do not sufficiently activate the reward system; consequently, when taken as directed in doses that are commonly prescribed for the treatment of ADHD, methylphenidate use lacks the capacity to cause an addiction.
Interactions
This section is transcluded from Methylphenidate. (edit | history)Methylphenidate may inhibit the metabolism of vitamin K anticoagulants, certain anticonvulsants, and some antidepressants (tricyclic antidepressants, and selective serotonin reuptake inhibitors). Concomitant administration may require dose adjustments, possibly assisted by monitoring of plasma drug concentrations. There are several case reports of methylphenidate inducing serotonin syndrome with concomitant administration of antidepressants.
When methylphenidate is coingested with ethanol, a metabolite called ethylphenidate is formed via hepatic transesterification, not unlike the hepatic formation of cocaethylene from cocaine and ethanol. The reduced potency of ethylphenidate and its minor formation means it does not contribute to the pharmacological profile at therapeutic doses, and even in overdose cases, ethylphenidate concentrations remain negligible.
Coingestion of alcohol also increases the blood plasma levels of d-methylphenidate by up to 40%.
Liver toxicity from methylphenidate is extremely rare, but limited evidence suggests that intake of β-adrenergic agonists with methylphenidate may increase the risk of liver toxicity.
Mode of activity
Methylphenidate is a catecholamine reuptake inhibitor that indirectly increases catecholaminergic neurotransmission by inhibiting the dopamine transporter (DAT) and norepinephrine transporter (NET), which are responsible for clearing catecholamines from the synapse, particularly in the striatum and meso-limbic system. Moreover, it is thought to "increase the release of these monoamines into the extraneuronal space."
Although four stereoisomers of methylphenidate (MPH) are possible, only the threo diastereoisomers are used in modern practice. There is a high eudysmic ratio between the SS and RR enantiomers of MPH. Dexmethylphenidate (d-threo-methylphenidate) is a preparation of the RR enantiomer of methylphenidate. In theory, D-TMP (d-threo-methylphenidate) can be anticipated to be twice the strength of the racemic product.
| Compd | DAT (Ki) | DA (IC50) | NET (Ki) | NE (IC50) |
|---|---|---|---|---|
| D-TMP | 161 | 23 | 206 | 39 |
| L-TMP | 2250 | 1600 | >10K | 980 |
| DL-TMP | 121 | 20 | 788 | 51 |
Pharmacology
Main article: Methylphenidate § PharmacologyDexmethylphenidate has a 4–6 hour duration of effect. A long-acting formulation, Focalin XR, which spans 12 hours is also available and has been shown to be as effective as DL (dextro-, levo-)-TMP (threo-methylphenidate) XR (extended release) (Concerta, Ritalin LA), with flexible dosing and good tolerability. It has also been demonstrated to reduce ADHD symptoms in both children and adults. d-MPH has a similar side-effect profile to MPH and can be administered without regard to food intake.
CTx-1301 is an experimental medication that is an extended-release formulation of dexmethylphenidate that has a half life more than an hour longer than extended-release dexmethylphenidate (d-MPH-ER). It is under development for ADHD.
Notes
- "Our findings suggest that methylphenidate may be associated with a number of serious adverse events as well as a large number of non-serious adverse events in children" "Concerning adverse events associated with the treatment, our systematic review of randomised clinical trials (RCTs) demonstrated no increase in serious adverse events, but a high proportion of participants suffered a range of non-serious adverse events."
- The management of amphetamine, dextroamphetamine, and methylphenidate overdose is largely supportive, with a focus on interruption of the sympathomimetic syndrome with judicious use of benzodiazepines. In cases where agitation, delirium, and movement disorders are unresponsive to benzodiazepines, second-line therapies include antipsychotics such as ziprasidone or haloperidol, central alpha-adrenoreceptor agonists such as dexmedetomidine, or propofol. ... However, fatalities are rare with appropriate care.
References
- ^ "Focalin- dexmethylphenidate hydrochloride tablet". DailyMed. 24 June 2020. Retrieved 15 November 2020.
- ^ "Focalin XR- dexmethylphenidate hydrochloride capsule, extended release". DailyMed. 27 June 2020. Retrieved 15 November 2020.
- ^ "Dexmethylphenidate Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 15 April 2019.
- Mosby's Drug Reference for Health Professions - E-Book. Elsevier Health Sciences. 2013. p. 455. ISBN 9780323187602.
- "Dexmethylphenidate Use During Pregnancy". Drugs.com. Retrieved 15 April 2019.
- ^ Moen MD, Keam SJ (December 2009). "Dexmethylphenidate extended release: a review of its use in the treatment of attention-deficit hyperactivity disorder". CNS Drugs. 23 (12): 1057–83. doi:10.2165/11201140-000000000-00000. PMID 19958043. S2CID 24975170.
- "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- "Dexmethylphenidate Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ "Concerta- methylphenidate hydrochloride tablet, extended release". DailyMed. 1 July 2021. Archived from the original on 26 March 2017. Retrieved 26 March 2022.
- "Methylphenidate: Use During Pregnancy and Breastfeeding". Drugs.com. Archived from the original on 2 January 2018.
- Humphreys C, Garcia-Bournissen F, Ito S, Koren G (July 2007). "Exposure to attention deficit hyperactivity disorder medications during pregnancy". Canadian Family Physician. 53 (7): 1153–1155. PMC 1949295. PMID 17872810.
- Ornoy A (February 2018). "Pharmacological Treatment of Attention Deficit Hyperactivity Disorder During Pregnancy and Lactation". Pharmaceutical Research. 35 (3): 46. doi:10.1007/s11095-017-2323-z. PMID 29411149. S2CID 3663423.
- ^ Keating GM, Figgitt DP (2002). "Dexmethylphenidate". Drugs. 62 (13): 1899–904, discussion 1905–8. doi:10.2165/00003495-200262130-00009. PMID 12215063. S2CID 249894173.
- Nutt D, King LA, Saulsbury W, Blakemore C (March 2007). "Development of a rational scale to assess the harm of drugs of potential misuse". Lancet. 369 (9566): 1047–1053. doi:10.1016/S0140-6736(07)60464-4. PMID 17382831. S2CID 5903121.
- Coghill D, Banaschewski T, Zuddas A, Pelaz A, Gagliano A, Doepfner M (September 2013). "Long-acting methylphenidate formulations in the treatment of attention-deficit/hyperactivity disorder: a systematic review of head-to-head studies". BMC Psychiatry. 13 (1). Springer Science and Business Media LLC: 237. doi:10.1186/1471-244x-13-237. PMC 3852277. PMID 24074240.
- "Ritalin LA (methylphenidate hydrochloride) extended-release capsules" (PDF). Novartis. Archived from the original (PDF) on 20 July 2011.
- ^ de Sousa A, Kalra G (January 2012). "Drug therapy of attention deficit hyperactivity disorder: current trends". Mens Sana Monographs. 10 (1): 45–69. doi:10.4103/0973-1229.87261 (inactive 2 November 2024). PMC 3353606. PMID 22654382.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - Jaanus SD (1992). "Ocular side effects of selected systemic drugs". Optometry Clinics. 2 (4): 73–96. PMID 1363080.
- Peterson BS, Trampush J, Maglione M, Bolshakova M, Rozelle M, Miles J, et al. (April 2024). "Treatments for ADHD in Children and Adolescents: A Systematic Review". Pediatrics. 153 (4). doi:10.1542/peds.2024-065787. PMID 38523592.
- Bron TI, Bijlenga D, Kasander MV, Spuijbroek AT, Beekman AT, Kooij JJ (June 2013). "Long-term relationship between methylphenidate and tobacco consumption and nicotine craving in adults with ADHD in a prospective cohort study". European Neuropsychopharmacology. 23 (6): 542–554. doi:10.1016/j.euroneuro.2012.06.004. PMID 22809706. S2CID 23148548.
- ^ Cortese S, Holtmann M, Banaschewski T, Buitelaar J, Coghill D, Danckaerts M, et al. (March 2013). "Practitioner review: current best practice in the management of adverse events during treatment with ADHD medications in children and adolescents". Journal of Child Psychology and Psychiatry, and Allied Disciplines. 54 (3): 227–246. doi:10.1111/jcpp.12036. PMID 23294014.
- Poulton A (August 2005). "Growth on stimulant medication; clarifying the confusion: a review". Archives of Disease in Childhood. 90 (8): 801–806. doi:10.1136/adc.2004.056952. PMC 1720538. PMID 16040876.
- Hinshaw SP, Arnold LE (January 2015). "ADHD, Multimodal Treatment, and Longitudinal outcome: Evidence, paradox, and challenge". Wiley Interdisciplinary Reviews. Cognitive Science. 6 (1): 39–52. doi:10.1002/wcs.1324. PMC 4280855. PMID 25558298.
- Findling RL, Dinh S (March 2014). "Transdermal therapy for attention-deficit hyperactivity disorder with the methylphenidate patch (MTS)". CNS Drugs. 28 (3): 217–228. doi:10.1007/s40263-014-0141-y. PMC 3933749. PMID 24532028.
- Kraemer M, Uekermann J, Wiltfang J, Kis B (July 2010). "Methylphenidate-induced psychosis in adult attention-deficit/hyperactivity disorder: report of 3 new cases and review of the literature". Clinical Neuropharmacology. 33 (4): 204–206. doi:10.1097/WNF.0b013e3181e29174. PMID 20571380. S2CID 34956456.
- Wingo AP, Ghaemi SN (2008). "Frequency of stimulant treatment and of stimulant-associated mania/hypomania in bipolar disorder patients". Psychopharmacology Bulletin. 41 (4): 37–47. PMID 19015628.
- "Methylphenidate ADHD medications: Drug safety communication – risk of long-lasting erections". U.S. Food and Drug Administration (FDA). 17 December 2013. Archived from the original on 17 December 2013. Retrieved 17 December 2013.
- "FDA drug safety communication: Safety review update of medications used to treat attention-deficit / hyperactivity disorder (ADHD) in children and young adults". U.S. Food and Drug Administration (FDA). 20 December 2011. Archived from the original on 30 October 2013. Retrieved 4 November 2013. Cooper WO, Habel LA, Sox CM, Chan KA, Arbogast PG, Cheetham TC, et al. (November 2011). "ADHD drugs and serious cardiovascular events in children and young adults". The New England Journal of Medicine. 365 (20): 1896–1904. doi:10.1056/NEJMoa1110212. PMC 4943074. PMID 22043968. "FDA Drug Safety Communication: Safety Review Update of Medications used to treat Attention-Deficit/Hyperactivity Disorder (ADHD) in adults". U.S. Food and Drug Administration (FDA). 15 December 2011. Archived from the original on 30 October 2013. Retrieved 4 November 2013. Habel LA, Cooper WO, Sox CM, Chan KA, Fireman BH, Arbogast PG, et al. (December 2011). "ADHD medications and risk of serious cardiovascular events in young and middle-aged adults". JAMA. 306 (24): 2673–2683. doi:10.1001/jama.2011.1830. PMC 3350308. PMID 22161946.
- Gordon N (1999). "Attention deficit hyperactivity disorder: Possible causes and treatment". International Journal of Clinical Practice. 53 (7): 524–528. doi:10.1111/j.1742-1241.1999.tb11794.x. PMID 10692738. S2CID 27462347.
- ^ Storebø OJ, Pedersen N, Ramstad E, Kielsholm ML, Nielsen SS, Krogh HB, et al. (May 2018). "Methylphenidate for attention deficit hyperactivity disorder (ADHD) in children and adolescents - assessment of adverse events in non-randomised studies". The Cochrane Database of Systematic Reviews (Systematic Review). 5 (5): CD012069. doi:10.1002/14651858.CD012069.pub2. PMC 6494554. PMID 29744873.
- ^ "Daytrana- methylphenidate patch". DailyMed. 15 June 2021. Retrieved 26 March 2022.
- ^ Heedes G, Ailakis J. "Methylphenidate hydrochloride (PIM 344)". INCHEM. International Programme on Chemical Safety. Archived from the original on 23 June 2015. Retrieved 23 June 2015.
- ^ Spiller HA, Hays HL, Aleguas A (July 2013). "Overdose of drugs for attention-deficit hyperactivity disorder: Clinical presentation, mechanisms of toxicity, and management". CNS Drugs. 27 (7): 531–543. doi:10.1007/s40263-013-0084-8. PMID 23757186. S2CID 40931380.
- Bruggisser M, Bodmer M, Liechti ME (2011). "Severe toxicity due to injected but not oral or nasal abuse of methylphenidate tablets". Swiss Medical Weekly. 141: w13267. doi:10.4414/smw.2011.13267. PMID 21984207.
- Morton WA, Stockton GG (October 2000). "Methylphenidate Abuse and Psychiatric Side Effects". Primary Care Companion to the Journal of Clinical Psychiatry. 2 (5): 159–164. doi:10.4088/PCC.v02n0502. PMC 181133. PMID 15014637.
- ^ Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 15: Reinforcement and addictive disorders". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A foundation for clinical neuroscience (2nd ed.). New York: McGraw-Hill Medical. p. 368. ISBN 978-0-07-148127-4.
- Auger RR, Goodman SH, Silber MH, Krahn LE, Pankratz VS, Slocumb NL (June 2005). "Risks of high-dose stimulants in the treatment of disorders of excessive somnolence: A case-control study". Sleep. 28 (6): 667–672. doi:10.1093/sleep/28.6.667. PMID 16477952.
- Ishii M, Tatsuzawa Y, Yoshino A, Nomura S (April 2008). "Serotonin syndrome induced by augmentation of SSRI with methylphenidate". Psychiatry and Clinical Neurosciences. 62 (2): 246. doi:10.1111/j.1440-1819.2008.01767.x. PMID 18412855. S2CID 5659107.
- Türkoğlu S (2015). "Serotonin syndrome with sertraline and methylphenidate in an adolescent". Clinical Neuropharmacology. 38 (2): 65–66. doi:10.1097/WNF.0000000000000075. PMID 25768857. S2CID 38523209.
- Park YM, Jung YK (May 2010). "Manic switch and serotonin syndrome induced by augmentation of paroxetine with methylphenidate in a patient with major depression". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 34 (4): 719–720. doi:10.1016/j.pnpbp.2010.03.016. PMID 20298736. S2CID 31984813.
- Bodner RA, Lynch T, Lewis L, Kahn D (February 1995). "Serotonin syndrome". Neurology. 45 (2): 219–223. doi:10.1212/wnl.45.2.219. PMID 7854515. S2CID 35190429.
- Patrick KS, González MA, Straughn AB, Markowitz JS (January 2005). "New methylphenidate formulations for the treatment of attention-deficit/hyperactivity disorder". Expert Opinion on Drug Delivery. 2 (1): 121–143. doi:10.1517/17425247.2.1.121. PMID 16296740. S2CID 25026467.
- ^ Markowitz JS, DeVane CL, Boulton DW, Nahas Z, Risch SC, Diamond F, et al. (June 2000). "Ethylphenidate formation in human subjects after the administration of a single dose of methylphenidate and ethanol". Drug Metabolism and Disposition. 28 (6): 620–624. PMID 10820132.
- Markowitz JS, Logan BK, Diamond F, Patrick KS (August 1999). "Detection of the novel metabolite ethylphenidate after methylphenidate overdose with alcohol coingestion". Journal of Clinical Psychopharmacology. 19 (4): 362–366. doi:10.1097/00004714-199908000-00013. PMID 10440465.
- Patrick KS, Straughn AB, Minhinnett RR, Yeatts SD, Herrin AE, DeVane CL, et al. (March 2007). "Influence of ethanol and gender on methylphenidate pharmacokinetics and pharmacodynamics". Clinical Pharmacology and Therapeutics. 81 (3): 346–353. doi:10.1038/sj.clpt.6100082. PMC 3188424. PMID 17339864.
- Roberts SM, DeMott RP, James RC (1997). "Adrenergic modulation of hepatotoxicity". Drug Metabolism Reviews. 29 (1–2): 329–353. doi:10.3109/03602539709037587. PMID 9187524.
- ^ Markowitz JS, Patrick KS (June 2008). "Differential pharmacokinetics and pharmacodynamics of methylphenidate enantiomers: does chirality matter?". Journal of Clinical Psychopharmacology. 28 (3 Suppl 2): S54-61. doi:10.1097/JCP.0b013e3181733560. PMID 18480678.
- Schweri MM, Skolnick P, Rafferty MF, Rice KC, Janowsky AJ, Paul SM (October 1985). "Threo-(+/-)-methylphenidate binding to 3,4-dihydroxyphenylethylamine uptake sites in corpus striatum: correlation with the stimulant properties of ritalinic acid esters". Journal of Neurochemistry. 45 (4): 1062–70. doi:10.1111/j.1471-4159.1985.tb05524.x. PMID 4031878. S2CID 28720285.
- Ding YS, Fowler JS, Volkow ND, Dewey SL, Wang GJ, Logan J, et al. (May 1997). "Chiral drugs: comparison of the pharmacokinetics of [11C]d-threo and L-threo-methylphenidate in the human and baboon brain". Psychopharmacology. 131 (1): 71–8. doi:10.1007/s002130050267. PMID 9181638. S2CID 26046917.
- Ding YS, Gatley SJ, Thanos PK, Shea C, Garza V, Xu Y, et al. (September 2004). "Brain kinetics of methylphenidate (Ritalin) enantiomers after oral administration". Synapse. 53 (3): 168–75. CiteSeerX 10.1.1.514.7833. doi:10.1002/syn.20046. PMID 15236349. S2CID 11664668.
- Davids E, Zhang K, Tarazi FI, Baldessarini RJ (February 2002). "Stereoselective effects of methylphenidate on motor hyperactivity in juvenile rats induced by neonatal 6-hydroxydopamine lesioning". Psychopharmacology. 160 (1): 92–8. doi:10.1007/s00213-001-0962-5. PMID 11862378. S2CID 8037050.
- Williard RL, Middaugh LD, Zhu HJ, Patrick KS (February 2007). "Methylphenidate and its ethanol transesterification metabolite ethylphenidate: brain disposition, monoamine transporters and motor activity". Behavioural Pharmacology. 18 (1): 39–51. doi:10.1097/FBP.0b013e3280143226. PMID 17218796. S2CID 20232871.
- McGough JJ, Pataki CS, Suddath R (July 2005). "Dexmethylphenidate extended-release capsules for attention deficit hyperactivity disorder". Expert Review of Neurotherapeutics. 5 (4): 437–41. doi:10.1586/14737175.5.4.437. PMID 16026226. S2CID 6561452.
- Silva R, Tilker HA, Cecil JT, Kowalik S, Khetani V, Faleck H, et al. (2004). "Open-label study of dexmethylphenidate hydrochloride in children and adolescents with attention deficit hyperactivity disorder". Journal of Child and Adolescent Psychopharmacology. 14 (4): 555–63. doi:10.1089/cap.2004.14.555. PMID 15662147.
- Arnold LE, Lindsay RL, Conners CK, Wigal SB, Levine AJ, Johnson DE, et al. (Winter 2004). "A double-blind, placebo-controlled withdrawal trial of dexmethylphenidate hydrochloride in children with attention deficit hyperactivity disorder". Journal of Child and Adolescent Psychopharmacology. 14 (4): 542–54. doi:10.1089/cap.2004.14.542. PMID 15662146.
- Spencer TJ, Adler LA, McGough JJ, Muniz R, Jiang H, Pestreich L (June 2007). "Efficacy and safety of dexmethylphenidate extended-release capsules in adults with attention-deficit/hyperactivity disorder". Biological Psychiatry. 61 (12): 1380–7. doi:10.1016/j.biopsych.2006.07.032. PMID 17137560. S2CID 45976373.
- Teo SK, Scheffler MR, Wu A, Stirling DI, Thomas SD, Stypinski D, et al. (February 2004). "A single-dose, two-way crossover, bioequivalence study of dexmethylphenidate HCl with and without food in healthy subjects". Journal of Clinical Pharmacology. 44 (2): 173–8. doi:10.1177/0091270003261899. PMID 14747426. S2CID 20694072.
- Brady LS, Lisanby SH, Gordon JA (3 August 2023). "New directions in psychiatric drug development: promising therapeutics in the pipeline". Expert Opinion on Drug Discovery. 18 (8): 835–850. doi:10.1080/17460441.2023.2224555. PMID 37352473. S2CID 259240509.
- Childress AC, Beltran N, Supnet C, Weiss MD (March 2021). "Reviewing the role of emerging therapies in the ADHD armamentarium". Expert Opinion on Emerging Drugs. 26 (1): 1–16. doi:10.1080/14728214.2020.1846718. PMID 33143485. S2CID 226251694.
- Ryst E, Childress A (2023). "An updated safety review of the current drugs for managing ADHD in children". Expert Opinion on Drug Safety. 22 (11): 1025–1040. doi:10.1080/14740338.2023.2271392. PMID 37843488. S2CID 264144450.
- Harris E (1 February 2018). "Industry update: what is new in the field of therapeutic delivery?". Therapeutic Delivery. 9 (3): 155–161. doi:10.4155/tde-2017-0117.
- Childress AC, Komolova M, Sallee FR (November 2019). "An update on the pharmacokinetic considerations in the treatment of ADHD with long-acting methylphenidate and amphetamine formulations". Expert Opinion on Drug Metabolism & Toxicology. 15 (11): 937–974. doi:10.1080/17425255.2019.1675636. PMID 31581854. S2CID 203660100.
| ADHD pharmacotherapies | |
|---|---|
| CNSTooltip central nervous system stimulants | |
| Non-classical CNS stimulants | |
| α2-adrenoceptor agonists | |
| Antidepressants | |
| Miscellaneous/others | |
| Related articles |
|
| Stimulants | |
|---|---|
| Adamantanes | |
| Adenosine antagonists | |
| Alkylamines | |
| Ampakines | |
| Arylcyclohexylamines | |
| Benzazepines | |
| Cathinones |
|
| Cholinergics |
|
| Convulsants | |
| Eugeroics | |
| Oxazolines | |
| Phenethylamines |
|
| Phenylmorpholines | |
| Piperazines | |
| Piperidines |
|
| Pyrrolidines | |
| Racetams | |
| Tropanes |
|
| Tryptamines | |
| Others |
|
| ATC code: N06B | |
| Monoamine reuptake inhibitors | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DATTooltip Dopamine transporter (DRIsTooltip Dopamine reuptake inhibitors) |
| ||||||||||||||
| NETTooltip Norepinephrine transporter (NRIsTooltip Norepinephrine reuptake inhibitors) |
| ||||||||||||||
| SERTTooltip Serotonin transporter (SRIsTooltip Serotonin reuptake inhibitors) |
| ||||||||||||||
| VMATsTooltip Vesicular monoamine transporters | |||||||||||||||
| Others |
| ||||||||||||||
| See also: Receptor/signaling modulators • Monoamine releasing agents • Adrenergics • Dopaminergics • Serotonergics • Monoamine metabolism modulators • Monoamine neurotoxins | |||||||||||||||
| Sigma receptor modulators | |
|---|---|
| σ1 |
|
| σ2 |
|
| Unsorted |
|
| See also: Receptor/signaling modulators | |
| Phenethylamines | |
|---|---|
| Phenethylamines |
|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|