| |
| Names | |
|---|---|
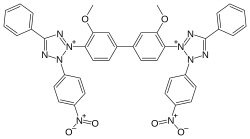
| IUPAC name 2,2'-bis(4-Nitrophenyl)-5,5'-diphenyl-3,3'-(3,3'-dimethoxy-4,4'-diphenylene)ditetrazolium chloride | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.517 |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C40H30Cl2N10O6 |
| Molar mass | 817.64 g/mol |
| Appearance | yellow crystalline powder |
| Melting point | 200 °C (392 °F; 473 K) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
| Main hazards | may be reactive based on presence of tetrazole group, nitro group and contiguous nitrogen atoms |
| Flash point | not available |
| Lethal dose or concentration (LD, LC): | |
| LD50 (median dose) | 2 g/kg |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Nitro blue tetrazolium is a chemical compound composed of two tetrazole moieties. It is used in immunology for sensitive detection of alkaline phosphatase (with BCIP). NBT serves as the oxidant and BCIP is the AP-substrate (and gives also dark blue dye).
Clinical significance
In immunohistochemistry the alkaline phosphatase is often used as a marker, conjugated to an antibody. The colored product can either be of the NBT/BCIP reaction reveals where the antibody is bound, or can be used in immunofluorescence.
The NBT/BCIP reaction is also used for colorimetric/spectrophotometric activity assays of oxidoreductases. One application is in activity stains in gel electrophoresis, such as with the mitochondrial electron transport chain complexes.
Nitro blue tetrazolium is used in a diagnostic test, particularly for chronic granulomatous disease and other diseases of phagocyte function. When there is an NADPH oxidase defect, the phagocyte is unable to make reactive oxygen species or radicals required for bacterial killing. As a result, bacteria may thrive within the phagocyte. The higher the blue score, the better the cell is at producing reactive oxygen species.
References
- Trinh le A, McCutchen MD, Bonner-Fraser M, Fraser SE, Bumm LA, McCauley DW (June 2007). "Fluorescent in situ hybridization employing the conventional NBT/BCIP chromogenic stain". BioTechniques. 42 (6): 756–9. doi:10.2144/000112476. PMID 17612300.
- Nisimoto Y, Wilson E, Heyl BL, Lambeth JD (5 January 1986). "NADH dehydrogenase from bovine neutrophil membranes. Purification and properties". J. Biol. Chem. 261 (1): 285–90. doi:10.1016/S0021-9258(17)42467-7. PMID 3941077.
- Freeman, R; King B (October 1972). "Technique for the performance of the nitro-blue tetrazolium (NBT) test". Journal of Clinical Pathology. 25 (10): 912–914. doi:10.1136/jcp.25.10.912. PMC 477548. PMID 4119008.
- Nathan DG, Baehner RL, Weaver DK (October 1969). "Failure of nitro blue tetrazolium reduction in the phagocytic vacuoles of leukocytes in chronic granulomatous disease". J. Clin. Invest. 48 (10): 1895–904. doi:10.1172/JCI106156. PMC 322426. PMID 5387730.
| Hematology blood tests | |
|---|---|
| Complete blood count | |
| Other tests of red blood cells | |
| Coagulation | |
| Other | |
Categories: