A slow-wave potential is a rhythmic electrophysiological event in the gastrointestinal tract. The normal conduction of slow waves is one of the key regulators of gastrointestinal motility. Slow waves are generated and propagated by a class of pacemaker cells called the interstitial cells of Cajal, which also act as intermediates between nerves and smooth muscle cells. Slow waves generated in interstitial cells of Cajal spread to the surrounding smooth muscle cells and control motility.
Description
In the human enteric nervous system, the slow-wave threshold is the slow-wave potential which must be reached before a slow wave can be propagated in gut wall smooth muscle. Slow waves themselves seldom cause any smooth muscle contraction (Except for, probably in the stomach). When the amplitude of slow waves in smooth muscle cells reaches the slow-wave threshold — the L-type Ca channels are activated, resulting in calcium influx and initiation of motility. Slow waves are generated at unique intrinsic frequencies by the interstitial cells of Cajal, even within the same organ. Entrainment of these different intrinsic frequencies through electrical coupling allows these unique intrinsic frequencies to occur at a single frequency within the stomach and segments of the small intestine. Electron microscopic and dye coupling studies to date have confirmed gap junctions as the major coupling mechanisms between interstitial cells of Cajal.
Coupling between ICC and smooth muscle cells is uncertain. Gap junctions have been demonstrated in rare circumstances as one coupling mechanism between ICC and smooth muscle cells. Another potential coupling mechanism is the "Peg and Socket" theory which demonstrates that the membranes of smooth muscle cells have the ability either form physical narrow "sockets" or "pegs" to lock onto other smooth muscle cells and/or interstitial cells of Cajal.
Types
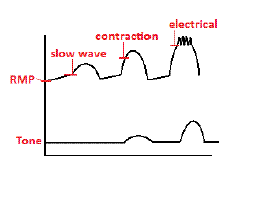
Gastric slow waves occur at around 3 cycles-per-minute in humans and exhibit significance variances in both amplitudes and propagation velocities in the stomach due to the existence of a gradient of resting membrane potential gradient, interstitial cells of Cajal distributions, and gastric wall thickness. Gastric slow waves frequency, propagation velocity, and amplitude demonstrate significant inter-species differences. Extracellular bioelectrical recording studies have demonstrated that gastric slow waves originate from a pacemaker region located on the greater curvature of the stomach. Human gastric slow waves propagate slower in the corpus than in the pacemaker region and antrum of the stomach. Up to four simultaneous slow wave wavefronts can occur in the human stomach.
Intestinal slow waves occur at around 12 cycles-per-minute in the duodenum, and decreases in frequency towards the colon. Entrainment of intestinal slow waves forms "frequency plateaus" in a piece-wise manner along the intestine. Similar to the stomach, intestinal slow waves frequency, propagation velocity, and amplitude also demonstrate significant inter-species differences.
In uterine smooth muscle, slow waves have not been consistently observed. Uterine muscle seems to generate action potentials spontaneously.
In gastrointestinal smooth muscle, the slow-wave threshold can be altered by input from endogenous and exogenous innervation, as well as excitatory (acetylcholine and Substance P) and inhibitory (vasoactive intestinal peptide and nitric oxide) compounds.
References
- Huizinga, J. D.; Lammers, W. J. E. P. (2008). "Gut peristalsis is governed by a multitude of cooperating mechanisms". AJP: Gastrointestinal and Liver Physiology. 296 (1): G1–8. doi:10.1152/ajpgi.90380.2008. PMID 18988693.
- Hanani, Menachem; Farrugia, Gianrico; Komuro, Terumasa (2004). Intercellular Coupling of Interstitial Cells of Cajal in the Digestive Tract. International Review of Cytology. Vol. 242. pp. 249–82. doi:10.1016/S0074-7696(04)42006-3. ISBN 978-0-12-364646-0. PMID 15598471.
- Thorneloe, Kevin S.; Nelson, Mark T. (2005). "Ion channels in smooth muscle: Regulators of intracellular calcium and contractility". Canadian Journal of Physiology and Pharmacology. 83 (3): 215–42. doi:10.1139/y05-016. PMID 15870837.
- Horiguchi, K; Komuro, T (1998). "Ultrastructural characterization of interstitial cells of Cajal in the rat small intestine using control and Ws/Ws mutant rats". Cell and Tissue Research. 293 (2): 277–84. doi:10.1007/s004410051119. PMID 9662650. S2CID 26179257.
- Zamir, O.; Hanani, M. (1990). "Intercellular dye-coupling in intestinal smooth muscle. Are gap junctions required for intercellular coupling?". Experientia. 46 (10): 1002–5. doi:10.1007/BF01940654. PMID 2226711. S2CID 30692665.
- Ishikawa, Koichi; Komuro, Terumasa (1996). "Characterization of the interstitial cells associated with the submuscular plexus of the guinea-pig colon". Anatomy and Embryology. 194 (1): 49–55. doi:10.1007/BF00196314. PMID 8800422. S2CID 23410156.
- Thuneberg, Lars; Peters, Susan (2001). "Toward a concept of stretch-coupling in smooth muscle. I. Anatomy of intestinal segmentation and sleeve contractions". The Anatomical Record. 262 (1): 110–24. doi:10.1002/1097-0185(20010101)262:1<110::AID-AR1016>3.0.CO;2-0. PMID 11146434. S2CID 34906117.
- ^ O'Grady, G.; Du, P.; Cheng, L. K.; Egbuji, J. U.; Lammers, W. J. E. P.; Windsor, J. A.; Pullan, A. J. (2010). "Origin and propagation of human gastric slow-wave activity defined by high-resolution mapping". AJP: Gastrointestinal and Liver Physiology. 299 (3): G585–92. doi:10.1152/ajpgi.00125.2010. PMC 2950696. PMID 20595620.
- ^ Egbuji, J. U.; o’Grady, G.; Du, P.; Cheng, L. K.; Lammers, W. J. E. P.; Windsor, J. A.; Pullan, A. J. (2010). "Origin, propagation and regional characteristics of porcine gastric slow wave activity determined by high-resolution mapping". Neurogastroenterology & Motility. 22 (10): e292–300. doi:10.1111/j.1365-2982.2010.01538.x. PMC 4110485. PMID 20618830.
- ^ Lammers, W. J. E. P.; Ver Donck, L.; Stephen, B.; Smets, D.; Schuurkes, J. A. J. (2009). "Origin and propagation of the slow wave in the canine stomach: The outlines of a gastric conduction system". AJP: Gastrointestinal and Liver Physiology. 296 (6): G1200–10. doi:10.1152/ajpgi.90581.2008. PMID 19359425.
- Farrugia, G.; Lei, S.; Lin, X.; Miller, S. M.; Nath, K. A.; Ferris, C. D.; Levitt, M.; Szurszewski, J. H. (2003). "A major role for carbon monoxide as an endogenous hyperpolarizing factor in the gastrointestinal tract". Proceedings of the National Academy of Sciences. 100 (14): 8567–70. Bibcode:2003PNAS..100.8567F. doi:10.1073/pnas.1431233100. PMC 166269. PMID 12832617.
- Angeli, Timothy R; O'Grady, Gregory; Paskaranandavadivel, Niranchan; Erickson, Jonathan C; Du, Peng; Pullan, Andrew J; Bissett, Ian P; Cheng, Leo K (2013). "Experimental and Automated Analysis Techniques for High-resolution Electrical Mapping of Small Intestine Slow Wave Activity". Journal of Neurogastroenterology and Motility. 19 (2): 179–91. doi:10.5056/jnm.2013.19.2.179. PMC 3644654. PMID 23667749.
- Lammers, W. J. E. P.; Stephen, B. (2007). "Origin and propagation of individual slow waves along the intact feline small intestine". Experimental Physiology. 93 (3): 334–46. doi:10.1113/expphysiol.2007.039180. PMID 18156170.
- Aguilar, H. N.; Mitchell, B. F. (2010). "Physiological pathways and molecular mechanisms regulating uterine contractility". Human Reproduction Update. 16 (6): 725–44. doi:10.1093/humupd/dmq016. PMID 20551073.
- Pathophysiology. Porth. 7th Ed. pg.875–878
Textbook of Medical Physiology - Gyton and Hall (12th edition)
Categories: