| Revision as of 07:32, 6 September 2011 editCurb Chain (talk | contribs)18,691 edits WP:HEAD← Previous edit | Revision as of 00:56, 2 November 2011 edit undo68.119.215.170 (talk)No edit summaryNext edit → | ||
| Line 49: | Line 49: | ||
| }} | }} | ||
| }} | }} | ||
| '''Potassium oxide''' is an ionic ] of ] and ]. This pale yellow solid, the simplest oxide of potassium, is a rarely encountered, highly reactive |
'''Potassium oxide''' is an ionic ] of ] and ]. This pale yellow solid, the simplest oxide of potassium, is a rarely encountered, highly reactive compound. Some materials of commerce, such as fertilizers and cements, are assayed assuming the percent composition that would be equivalent to K<sub>2</sub>O. | ||
| ==Production== | ==Production== | ||
Revision as of 00:56, 2 November 2011
| |
| Names | |
|---|---|
| IUPAC name Potassium oxide | |
| Other names Potassium monoxide | |
| Identifiers | |
| CAS Number | |
| ECHA InfoCard | 100.032.012 |
| UN number | 2033 |
| CompTox Dashboard (EPA) | |
| Properties | |
| Chemical formula | K2O |
| Molar mass | 94.20 g/mol |
| Appearance | pale yellow solid |
| Density | 2.35 g/cm |
| Melting point | >350 °C decomp. |
| Solubility in water | Reacts forming KOH |
| Structure | |
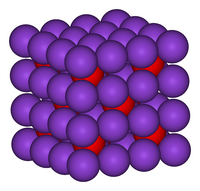
| Crystal structure | Antifluorite (cubic), cF12 |
| Space group | Fm3m, No. 225 |
| Coordination geometry | Tetrahedral (K); cubic (O) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
| Main hazards | Corrosive, reacts violently with water |
| Flash point | Non-flammable |
| Related compounds | |
| Other anions | Potassium sulfide |
| Other cations | Lithium oxide Sodium oxide Rubidium oxide Caesium oxide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Potassium oxide is an ionic compound of potassium and oxygen. This pale yellow solid, the simplest oxide of potassium, is a rarely encountered, highly reactive compound. Some materials of commerce, such as fertilizers and cements, are assayed assuming the percent composition that would be equivalent to K2O.
Production
Potassium oxide is produced from the reaction of oxygen and potassium; this reaction affords potassium peroxide, K2O2. Treatment of the peroxide with potassium produces the oxide:
- K2O2 + 2 K → 2 K2O
Alternatively and more conveniently, K2O is synthesized by heating potassium nitrate with metallic potassium:
- 2 KNO3 + 10 K → 6 K2O + N2
Potassium hydroxide cannot be further dehydrated to the oxide.
Properties and reactions
K2O crystallises in the antifluorite structure. In this motif the positions of the anions and cations are reversed relative to their positions in CaF2, with potassium ions coordinated to 4 oxide ions and oxide ions coordinated to 8 potassium. K2O is a basic oxide and reacts with water violently to produce the caustic potassium hydroxide. It is deliquescent and will absorb water from the atmosphere, initiating this vigorous reaction.
Fertilizers
The chemical formula K2O is used in the N-P-K (nitrogen-phosphorus-potassium) numbers on the labels of fertilizers. Although K2O is the correct formula for potassium oxide, potassium oxide is not used as a fertilizer in these products. Normally, potassium chloride, potassium sulfate, or potassium carbonate is used as a fertilizer source for potassium. The percentage of K2O given on the label only represents the amount of potassium in the fertilizer if it was in the form of potassium oxide. Potassium oxide is about 83% potassium by weight, but potassium chloride, for instance, is only 52% potassium by weight. Potassium chloride provides less potassium than an equal amount of potassium oxide. Thus, if a fertilizer is 30% potassium chloride by weight, its standard potassium rating, based on potassium oxide, would be only 19%.
References
- Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- Zintl, E.; Harder, A.; Dauth B. (1934), "Gitterstruktur der oxyde, sulfide, selenide und telluride des lithiums, natriums und kaliums", Z. Elektrochem. Angew. Phys. Chem., 40: 588–93
{{citation}}: CS1 maint: multiple names: authors list (link) - Wells, A.F. (1984) Structural Inorganic Chemistry, Oxford: Clarendon Press. ISBN 0-19-855370-6.
| Potassium compounds | |
|---|---|
| H, (pseudo)halogens | |
| chalcogens | |
| pnictogens | |
| B, C group | |
| transition metals | |
| organic | |