| Inoviridae | |
|---|---|

| |
| Electron micrograph of shadowed filamentous bacteriophage (inovirus) | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Monodnaviria |
| Kingdom: | Loebvirae |
| Phylum: | Hofneiviricota |
| Class: | Faserviricetes |
| Order: | Tubulavirales |
| Family: | Inoviridae |
| Genera | |
Filamentous bacteriophages are a family of viruses (Inoviridae) that infect bacteria, or bacteriophages. They are named for their filamentous shape, a worm-like chain (long, thin, and flexible, reminiscent of a length of cooked spaghetti), about 6 nm in diameter and about 1000-2000 nm long. This distinctive shape reflects their method of replication: the coat of the virion comprises five types of viral protein, which are located in the inner membrane of the host bacterium during phage assembly, and these proteins are added to the nascent virion's DNA as it is extruded through the membrane. The simplicity of filamentous phages makes them an appealing model organism for research in molecular biology, and they have also shown promise as tools in nanotechnology and immunology.
Characteristics

Filamentous bacteriophages are among the simplest living organisms known, with far fewer genes than the classical tailed bacteriophages studied by the phage group in the mid-20th century. The family contains 29 defined species, divided among 23 genera. However, mining of genomic and metagenomic datasets using a machine learning approach led to the discovery of 10,295 inovirus-like sequences in nearly all bacterial phyla across virtually every ecosystem, indicating that this group of viruses is much more diverse and widespread than originally appreciated.
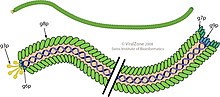
Three filamentous bacteriophages, fd, f1, and M13, were isolated and characterized by three different research groups in the early 1960s, but they are so similar that they are sometimes grouped under the common name "Ff", which are members of the genus Inovirus, as acknowledged by the International Committee on Taxonomy of Viruses (ICTV). The molecular structure of Ff phages was determined using a number of physical techniques, especially X-ray fiber diffraction, solid-state NMR and cryo-electron microscopy. The structures of the phage capsid and of some other phage proteins are available from the Protein Data Bank. The single-stranded Ff phage DNA runs down the central core of the phage, and is protected by a cylindrical protein coat built from thousands of identical α-helical major coat protein subunits coded by phage gene 8. The gene 8 protein is inserted into the plasma membrane as an early step in phage assembly. Some strains of phage have a "leader sequence" on the gene 8 protein to promote membrane insertion, but others do not seem to need the leader sequence. The two ends of the phage are capped by a few copies of proteins that are important for infection of the host bacteria, and also for assembly of nascent phage particles. These proteins are the products of phage genes 3 and 6 at one end of the phage, and phage genes 7 and 9 at the other end. The fiber diffraction studies identified two structural classes of phage, differing in the details of the arrangement of the gene 8 protein. Class I has a rotation axis relating the gene 8 coat proteins, whereas for Class II this rotation axis is replaced by a helix axis. This technical difference has little noticeable effect on the overall phage structure, but the extent of independent diffraction data is greater for symmetry Class II than for Class I. This assisted the determination of the Class II phage Pf1 structure, and by extension the Class I structure.
Structural Class I includes strains fd, f1, M13 of genus Inovirus as well as If1 (of ICTV's species Escherichia virus If1, genus Infulavirus) and IKe (of ICTV's species Salmonella virus IKe, genus Lineavirus), whereas Class II includes strains Pf1 (of ICTV's species Pseudomonas virus Pf1 of genus Primolicivirus), and perhaps also Pf3 (of ICTV's species Pseudomonas virus Pf3 of genus Tertilicivirus), Pf4 and PH75 (of NCBI's proposed species Thermus phage PH75, incertae sedis within Inoviridae).
The DNA isolated from fd phage (of genus Inovirus) is single-stranded, and topologically a circle. That is, the DNA single strand extends from one end of the phage particle to the other and then back again to close the circle, although the two strands are not base-paired. This topology was assumed to extend to all other filamentous phages, but it is not the case for phage Pf4, for which the DNA in the phage is single-stranded but topologically linear, not circular. During fd phage assembly, the phage DNA is first packaged into a linear intracellular nucleoprotein complex with many copies of the phage gene 5 replication/assembly protein. The gene 5 protein is then displaced by the gene 8 coat protein as the nascent phage is extruded across the bacterial plasma membrane without killing the bacterial host. This protein also binds with high affinity to G-quadruplex structures (although they are not present in the phage DNA) and to similar hairpin structures in phage DNA.
The p1 protein of Ff phage (i. e. genus Inovirus), which is required for phage assembly at the membrane, has a membrane-spanning hydrophobic domain with the N-terminal portion in the cytoplasm and the C-terminal portion in the periplasm (the reverse of the orientation of the gene 8 coat protein). Adjacent to the cytoplasmic side of the membrane-spanning domain is a 13- residue sequence of p1 having a pattern of basic residues closely matching the pattern of basic residues near the C terminus of p8, but inverted with respect to the sequence. This assembly mechanism makes this phage a valuable system with which to study transmembrane proteins. Gene 1, coding for an ATPase, is a conserved marker gene that (along with three additional genetic features) was used to automatically detect inovirus sequences.
Life cycle
Viral replication is cytoplasmic. Entry into the host cell is achieved by pilus-mediated adsorption into the host cell. Replication follows the ssDNA rolling circle model. DNA-templated transcription is the method of transcription. The virus exits the host cell by viral extrusion. Viral assembly occurs at the inner membrane (in case of Gram-negative bacteria), mediated by a membrane-embedded motor protein complex. This multimeric assembly complex, including p1 encoded by gene 1 (referred to as ZOT, zonula occludens toxin by researchers on Vibrio cholerae phage CTXΦ) is an ATPase containing functional and essential Walker motifs that are thought to mediate the hydrolysis of ATP providing the energy for the assembly of the phage filament. Filamentous phage Cf1t from Xanthomonas campestris (of NCBI's proposed species Xanthomonas phage Cf1t, incertae sedis within Inoviridae, likely misspelled as Cflt), was shown in 1987 to integrate into the host bacterial genome, and further such temperate filamentous phages have since been reported, many of which have been implicated in pathogenesis.
Taxonomy
The following genera are recognized:
- Affertcholeramvirus
- Bifilivirus
- Capistrivirus
- Coriovirus
- Fibrovirus
- Habenivirus
- Infulavirus
- Inovirus
- Lineavirus
- Parhipatevirus
- Primolicivirus
- Psecadovirus
- Restivirus
- Saetivirus
- Scuticavirus
- Staminivirus
- Subteminivirus
- Tertilicivirus
- Thomixvirus
- Versovirus
- Vicialiavirus
- Villovirus
- Xylivirus
Phylogenetic trees and clades have been increasingly used to study taxonomy of Inoviridae.
On base of metagenomical data, the family has been proposed to be split into new families Amplinoviridae, Protoinoviridae, Photinoviridae, Vespertilinoviridae, Densinoviridae, and Paulinoviridae, all within order Tubulavirales, of course.
Notable members
- genus Inovirus aka Ff phages – these infect Escherichia coli carrying the F episome.
- species Escherichia virus M13
- species Filamentous bacteriophage fd (proposal)
- fd phage
- genus Affertcholeramvirus
- species Vibrio virus CTXphi
- Vibrio phage CTXphi (aka CTXφ bacteriophage)
- genus Infulavirus)
- species Escherichia virus If1
- If1 phage
- genus Lineavirus
- species Salmonella virus IKe
- IKe phage
- genus Primolicivirus)
- species Pseudomonas virus Pf1
- Pf1 phage
- genus Tertilicivirus)
- species Pseudomonas virus Pf3 — bacteriophages that infect Pseudomonas aeruginosa
- Pf3 phage
Further notable proposed species are:
- species Thermus phage PH75
- PH75 phage
- species Xanthomonas phage Cf1t (likely misspelled as Cflt)
- Cf1t phage
History
The filamentous particle seen in electron micrographs was initially incorrectly interpreted as contaminating bacterial pilus, but ultrasonic degradation, which breaks flexible filaments roughly in half, inactivated infectivity as predicted for a filamentous bacteriophage morphology. Three filamentous bacteriophages, fd, f1 and M13, were isolated and characterized by three different research groups in the early 1960s. Since these three phages differ by less than 2 percent in their DNA sequences, corresponding to changes in only a few dozen codons in the whole genome, for many purposes they can be considered to be identical. Further independent characterization over the subsequent half-century was shaped by the interests of these research groups and their followers.
Filamentous phages, unlike most other phages, are continually extruded through the bacterial membrane without killing the host. Genetic studies on M13 using conditional lethal mutants, initiated by David Pratt and colleagues, led to description of phage gene functions. Notably, the protein product of gene 5, which is required for synthesis of progeny single-stranded DNA, is made in large amounts in the infected bacteria, and it binds to the nascent DNA to form a linear intracellular complex. (The simple numbering of genes using Arabic numerals 1,2,3,4... introduced by the Pratt group is sometimes displaced by the practice of using Roman numerals I, II, III, IV... but the gene numbers defined by the two systems are the same).
Longer (or shorter) DNA can be included in fd phage, since more (or fewer) protein subunits can be added during assembly as required to protect the DNA, making the phage convenient for genetic studies. The length of the phage is also affected by the positive charge per length on the inside surface of the phage capsid. The genome of fd was one of the first complete genomes to be sequenced.
The taxonomy of filamentous bacteriophage was defined by Andre Lwoff and Paul Tournier as family Inophagoviridae, genus I. inophagovirus, species Inophagovirus bacterii (Inos=fiber or filament in Greek), with phage fd (Hoffmann-Berling) as the type species. "Phagovirus" is tautological, and the name of the family was altered to Inoviridae and the type genus to Inovirus. This nomenclature persisted for many decades, although the definition of fd as type species was replaced as M13 became more widely used for genetic manipulation, and for studies of p8 in membrane mimetic environments. The number of known filamentous bacteriophages has multiplied many-fold by using a machine-learning approach, and it has been suggested that "the former Inoviridae family should be reclassified as an order, provisionally divided into 6 candidate families and 212 candidate subfamilies". Phages fd, f1, M13 and other related phages are Ff phages, for F specific (they infect Escherichia coli carrying the F-episome) filamentous phages, using the concept of vernacular name.
Filamentous bacteriophage engineered to display immunogenic peptides are useful in immunology and wider biological applications. George Smith and Greg Winter used f1 and fd for their work on phage display for which they were awarded a share of the 2018 Nobel Prize in Chemistry. The creation and exploitation of many derivatives of M13 for a wide range of purposes, especially in materials science, has been employed by Angela Belcher and colleagues. Filamentous bacteriophage can promote antibiotic tolerance by forming liquid crystalline domains around bacterial cells.
References
- ^ Hay ID, Lithgow T (June 2019). "Filamentous phages: masters of a microbial sharing economy". EMBO Reports. 20 (6): e47427. doi:10.15252/embr.201847427. PMC 6549030. PMID 30952693.
- ^ Straus SK, Bo HE (2018). "Filamentous Bacteriophage Proteins and Assembly". In Bhella JR, Harris D (eds.). Virus Protein and Nucleoprotein Complexes. Subcellular Biochemistry. Vol. 88. Springer Singapore. pp. 261–279. doi:10.1007/978-981-10-8456-0_12. ISBN 978-981-10-8455-3. PMID 29900501.
- ^ Mai-Prochnow A, Hui JG, Kjelleberg S, Rakonjac J, McDougald D, Rice SA (July 2015). "Big things in small packages: the genetics of filamentous phage and effects on fitness of their host". FEMS Microbiology Reviews. 39 (4): 465–87. doi:10.1093/femsre/fuu007. hdl:10453/65260. PMID 25670735.
- ^ Rakonjac J, Russel M, Khanum S, Brooke SJ, Rajič M (2017). "Filamentous Phage: Structure and Biology". In Lim TS (ed.). Recombinant Antibodies for Infectious Diseases. Advances in Experimental Medicine and Biology. Vol. 1053. Springer International Publishing. pp. 1–20. doi:10.1007/978-3-319-72077-7_1. ISBN 978-3-319-72076-0. PMID 29549632.
- ^ Roux S, Krupovic M, Daly RA, Borges AL, Nayfach S, Schulz F, et al. (November 2019). "Cryptic inoviruses revealed as pervasive in bacteria and archaea across Earth's biomes". Nature Microbiology. 4 (11): 1895–1906. doi:10.1038/s41564-019-0510-x. PMC 6813254. PMID 31332386.
- ^ "Inoviridae ~ ViralZone". viralzone.expasy.org. Retrieved 31 March 2021.
- ^ ICTV. "Virus Taxonomy: 2019 Release". Retrieved 4 July 2020.
- ^ NCBI: Inovirus (genus)
- ^ ICTV: ICTV Taxonomy history: Inovirus. 2019 EC 51, Berlin, Germany, July 2019; Email ratification March 2020 (MSL #35).
- ^ Tarafder AK, von Kügelgen A, Mellul AJ, Schulze U, Aarts DG, Bharat TA (March 2020). "Phage liquid crystalline droplets form occlusive sheaths that encapsulate and protect infectious rod-shaped bacteria". Proceedings of the National Academy of Sciences of the United States of America. 117 (9): 4724–4731. Bibcode:2020PNAS..117.4724T. doi:10.1073/pnas.1917726117. PMC 7060675. PMID 32071243.
- ^ NCBI: Infulavirus (genus)
- ^ NCBI: Lineavirus (genus)
- ^ NCBI: Primolicivirus (genus)
- ^ NCBI: Tertilicivirus (genus)
- ^ Secor, Patrick R.; Burgener, Elizabeth B.; Kinnersley, M.; Jennings, Laura K.; Roman-Cruz, Valery; Popescu, Medeea; Van Belleghem, Jonas D.; Haddock, Naomi; Copeland, Conner; Michaels, Lia A.; de Vries, Christiaan R. (2020). "Pf Bacteriophage and Their Impact on Pseudomonas Virulence, Mammalian Immunity, and Chronic Infections". Frontiers in Immunology. 11: 244. doi:10.3389/fimmu.2020.00244. ISSN 1664-3224. PMC 7047154. PMID 32153575.
- ^ NCBI: Thermus phage PH75 (species)
- ^ Pratt D, Laws P, Griffith J (February 1974). "Complex of bacteriophage M13 single-stranded DNA and gene 5 protein". Journal of Molecular Biology. 82 (4): 425–39. doi:10.1016/0022-2836(74)90239-3. PMID 4594145.
- Gray CW (July 1989). "Three-dimensional structure of complexes of single-stranded DNA-binding proteins with DNA. IKe and fd gene 5 proteins form left-handed helices with single-stranded DNA". Journal of Molecular Biology. 208 (1): 57–64. doi:10.1016/0022-2836(89)90087-9. PMID 2671388.
- ^ Hoffmann Berling H, Maze R (March 1964). "Release of male-specific bacteriophages from surviving host bacteria". Virology. 22 (3): 305–13. doi:10.1016/0042-6822(64)90021-2. PMID 14127828.
- Wen JD, Gray DM (March 2004). "Ff gene 5 single-stranded DNA-binding protein assembles on nucleotides constrained by a DNA hairpin". Biochemistry. 43 (9): 2622–34. doi:10.1021/bi030177g. PMID 14992600.
- Rapoza, M.P.; Webster, R. L. (1995). "The Products of Gene I and the Overlapping in-Frame Gene XI are Required for Filamentous Phage Assembly". J. Mol. Biol. 248 (3): 627–638. doi:10.1006/jmbi.1995.0247. PMID 7752229.
- ^ Loh, Belinda; Haase, Maximilian; Mueller, Lukas; Kuhn, Andreas; Leptihn, Sebastian (9 April 2017). "The Transmembrane Morphogenesis Protein gp1 of Filamentous Phages Contains Walker A and Walker B Motifs Essential for Phage Assembly". Viruses. 9 (4): 73. doi:10.3390/v9040073. PMC 5408679. PMID 28397779.
- ^ Loh, Belinda; Kuhn, Andreas; Leptihn, Sebastian (May 2019). "The fascinating biology behind phage display: filamentous phage assembly". Molecular Microbiology. 111 (5): 1132–1138. doi:10.1111/mmi.14187. PMID 30556628.
- ^ NCBI: Xanthomonas phage Cf1t (species)
- International Committee on Taxonomy of Viruses Executive Committee (May 2020). "The new scope of virus taxonomy: partitioning the virosphere into 15 hierarchical ranks". Nature Microbiology. 5 (5): 668–674. doi:10.1038/s41564-020-0709-x. PMC 7186216. PMID 32341570.
- Kazlauskas D, Varsani A, Koonin EV, Krupovic M (July 2019). "Multiple origins of prokaryotic and eukaryotic single-stranded DNA viruses from bacterial and archaeal plasmids". Nature Communications. 10 (1): 3425. Bibcode:2019NatCo..10.3425K. doi:10.1038/s41467-019-11433-0. PMC 6668415. PMID 31366885.
- Simon Roux, Mart Krupovic, Rebecca A. Daly, Adair L. Borges, Stephen Nayfach, Frederik Schulz, Emiley A. Eloe-Fadrosh et al.: Cryptic inoviruses revealed as pervasive in bacteria and archaea across Earth’s biomes. In: Nature Microbiology, 22 July 2019, doi:10.1038/s41564-019-0510-x
- NCBI: Affertcholeramvirus (genus)
- Freifelder D, Davison PF (May 1962). "Studies on the sonic degradation of deoxyribonucleic acid". Biophysical Journal. 2 (3): 235–47. Bibcode:1962BpJ.....2..235F. doi:10.1016/S0006-3495(62)86852-0. PMC 1366369. PMID 13894963.
- Marvin DA, Hoffmann-Berling H (1963). "Physical and Chemical Properties of Two New Small Bacteriophages". Nature. 197 (4866): 517–518. Bibcode:1963Natur.197..517M. doi:10.1038/197517b0. hdl:11858/00-001M-0000-002D-442F-1. S2CID 4224468.
- Morag O, Abramov G, Goldbourt A (December 2011). "Similarities and differences within members of the Ff family of filamentous bacteriophage viruses". The Journal of Physical Chemistry B. 115 (51): 15370–9. doi:10.1021/jp2079742. PMID 22085310.
- Pratt D, Tzagoloff H, Erdahl WS (November 1966). "Conditional lethal mutants of the small filamentous coliphage M13. I. Isolation, complementation, cell killing, time of cistron action". Virology. 30 (3): 397–410. doi:10.1016/0042-6822(66)90118-8. PMID 5921643.
- Pratt D, Tzagoloff H, Beaudoin J (September 1969). "Conditional lethal mutants of the small filamentous coliphage M13. II. Two genes for coat proteins". Virology. 39 (1): 42–53. doi:10.1016/0042-6822(69)90346-8. PMID 5807970.
- Pratt D, Erdahl WS (October 1968). "Genetic control of bacteriophage M13 DNA synthesis". Journal of Molecular Biology. 37 (1): 181–200. doi:10.1016/0022-2836(68)90082-X. PMID 4939035.
- Henry TJ, Pratt D (March 1969). "The proteins of bacteriophage M13". Proceedings of the National Academy of Sciences of the United States of America. 62 (3): 800–7. Bibcode:1969PNAS...62..800H. doi:10.1073/pnas.62.3.800. PMC 223669. PMID 5257006.
- Alberts B, Frey L, Delius H (July 1972). "Isolation and characterization of gene 5 protein of filamentous bacterial viruses". Journal of Molecular Biology. 68 (1): 139–52. doi:10.1016/0022-2836(72)90269-0. PMID 4115107.
- Herrmann R, Neugebauer K, Zentgraf H, Schaller H (February 1978). "Transposition of a DNA sequence determining kanamycin resistance into the single-stranded genome of bacteriophage fd". Molecular & General Genetics. 159 (2): 171–8. doi:10.1007/bf00270890. PMID 345091. S2CID 22923713.
- Sattar, Sadia; Bennett, Nicholas J.; Wen, Wesley X.; Guthrie, Jenness M.; Blackwell, Len F.; Conway, James F.; Rakonjac, Jasna (2015). "Ff-nano, short functionalized nanorods derived from Ff (f1, fd, or M13) filamentous bacteriophage". Frontiers in Microbiology. 6: 316. doi:10.3389/fmicb.2015.00316. ISSN 1664-302X. PMC 4403547. PMID 25941520.
- Greenwood J, Hunter GJ, Perham RN (January 1991). "Regulation of filamentous bacteriophage length by modification of electrostatic interactions between coat protein and DNA". Journal of Molecular Biology. 217 (2): 223–7. doi:10.1016/0022-2836(91)90534-d. PMID 1992159.
- Beck E, Sommer R, Auerswald EA, Kurz C, Zink B, Osterburg G, et al. (December 1978). "Nucleotide sequence of bacteriophage fd DNA". Nucleic Acids Research. 5 (12): 4495–503. doi:10.1093/nar/5.12.4495. PMC 342768. PMID 745987.
- Lwoff A, Tournier P (1966). "The classification of viruses". Annual Review of Microbiology. 20 (1): 45–74. doi:10.1146/annurev.mi.20.100166.000401. PMID 5330240.
- Matthews RE (1982). "Classification and nomenclature of viruses. Fourth report of the International Committee on Taxonomy of Viruses". Intervirology. 17 (1–3): 1–199. doi:10.1159/000149278. PMID 6811498.
- Messing, Joachim (1991). "Cloning in M13 phage or how to use biology at its best". Gene. 100: 3–12. doi:10.1016/0378-1119(91)90344-B. ISSN 0378-1119. PMID 2055478.
- Messing, Joachim (1996). "Cloning Single-Stranded DNA". Molecular Biotechnology. 5 (1): 39–47. doi:10.1007/BF02762411. PMID 8853015. S2CID 33495106.
- Gibbs AJ, Harrison BD, Watson DH, Wildy P (January 1966). "What's in a virus name?". Nature. 209 (5022): 450–4. Bibcode:1966Natur.209..450G. doi:10.1038/209450a0. PMID 5919575. S2CID 4288812.
- Smith GP (June 1985). "Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface". Science. 228 (4705): 1315–7. Bibcode:1985Sci...228.1315S. doi:10.1126/science.4001944. PMID 4001944.
- Prisco A, De Berardinis P (24 April 2012). "Filamentous bacteriophage fd as an antigen delivery system in vaccination". International Journal of Molecular Sciences. 13 (4): 5179–94. doi:10.3390/ijms13045179. PMC 3344273. PMID 22606037.
- Sioud M (April 2019). "Phage Display Libraries: From Binders to Targeted Drug Delivery and Human Therapeutics". Molecular Biotechnology. 61 (4): 286–303. doi:10.1007/s12033-019-00156-8. PMID 30729435. S2CID 73434013.
- ^ Henry KA, Arbabi-Ghahroudi M, Scott JK (2015). "Beyond phage display: non-traditional applications of the filamentous bacteriophage as a vaccine carrier, therapeutic biologic, and bioconjugation scaffold". Frontiers in Microbiology. 6: 755. doi:10.3389/fmicb.2015.00755. PMC 4523942. PMID 26300850.
- Casey JP, Barbero RJ, Heldman N, Belcher AM (November 2014). "Versatile de novo enzyme activity in capsid proteins from an engineered M13 bacteriophage library". Journal of the American Chemical Society. 136 (47): 16508–14. doi:10.1021/ja506346f. PMID 25343220.
- Oh D, Qi J, Han B, Zhang G, Carney TJ, Ohmura J, et al. (August 2014). "M13 virus-directed synthesis of nanostructured metal oxides for lithium-oxygen batteries". Nano Letters. 14 (8): 4837–45. Bibcode:2014NanoL..14.4837O. doi:10.1021/nl502078m. PMID 25058851.
- Dorval Courchesne NM, Klug MT, Huang KJ, Weidman MC, Cantú VJ, Chen PY, et al. (June 2015). "Constructing Multifunctional Virus-Templated Nanoporous Composites for Thin Film Solar Cells: Contributions of Morphology and Optics to Photocurrent Generation". The Journal of Physical Chemistry C. 119 (25): 13987–4000. doi:10.1021/acs.jpcc.5b00295. hdl:1721.1/102981. ISSN 1932-7447.
- Dogic Z (30 June 2016). "Filamentous Phages As a Model System in Soft Matter Physics". Frontiers in Microbiology. 7: 1013. doi:10.3389/fmicb.2016.01013. PMC 4927585. PMID 27446051.
- Secor PR, Jennings LK, Michaels LA, Sweere JM, Singh PK, Parks WC, Bollyky PL (December 2015). "Pseudomonas aeruginosa biofilm matrix into a liquid crystal". Microbial Cell. 3 (1): 49–52. doi:10.15698/mic2016.01.475 (inactive 2 November 2024). PMC 5354590. PMID 28357315.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link)
External links
| Taxon identifiers | |
|---|---|
| Inoviridae | |