| |
| |
| Names | |
|---|---|
| Preferred IUPAC name Pentanedioic acid | |
| Other names
Glutaric acid Propane-1,3-dicarboxylic acid 1,3-Propanedicarboxylic acid Pentanedioic acid n-Pyrotartaric acid 1,5-Pentanedioic acid | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.471 |
| EC Number |
|
| KEGG | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
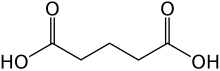
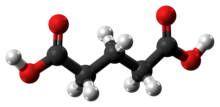
| Chemical formula | C5H8O4 |
| Molar mass | 132.12 g/mol |
| Melting point | 95 to 98 °C (203 to 208 °F; 368 to 371 K) |
| Boiling point | 200 °C (392 °F; 473 K) /20 mmHg |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Glutaric acid is the organic compound with the formula C3H6(COOH)2. Although the related "linear" dicarboxylic acids adipic and succinic acids are water-soluble only to a few percent at room temperature, the water-solubility of glutaric acid is over 50% (w/w).
Biochemistry
Glutaric acid is naturally produced in the body during the metabolism of some amino acids, including lysine and tryptophan. Defects in this metabolic pathway can lead to a disorder called glutaric aciduria, where toxic byproducts build up and can cause severe encephalopathy.
Production
Glutaric acid can be prepared by the ring-opening of butyrolactone with potassium cyanide to give the potassium salt of the carboxylate-nitrile that is hydrolyzed to the diacid. Alternatively hydrolysis, followed by oxidation of dihydropyran gives glutaric acid. It can also be prepared from reacting 1,3-dibromopropane with sodium or potassium cyanide to obtain the dinitrile, followed by hydrolysis. Using periodate, it is obtained from oxidation of 1,3-cyclohexanedione, which proceeds with decarboxylation.
Uses
- 1,5-Pentanediol, a common plasticizer and precursor to polyesters is manufactured by hydrogenation of glutaric acid and its derivatives.
- Glutaric acid itself has been used in the production of polymers such as polyester polyols, polyamides. The odd number of carbon atoms (i.e. 5) is useful in decreasing polymer elasticity.
- Pyrogallol can be produced from glutaric diester.
Safety
Glutaric acid may cause irritation to the skin and eyes. Acute hazards include the fact that this compound may be harmful by ingestion, inhalation or skin absorption.
References
- G. Paris, L. Berlinguet, R. Gaudry, J. English, Jr. and J. E. Dayan (1957). "Glutaric Acid and Glutaramide". Organic Syntheses: 47. doi:10.15227/orgsyn.037.0047
{{cite journal}}: CS1 maint: multiple names: authors list (link). - Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 1736, ISBN 978-0-471-72091-1
- Peter Werle and Marcus Morawietz "Alcohols, Polyhydric" in Ullmann's Encyclopedia of Industrial Chemistry: 2002, Wiley-VCH: Weinheim. DOI 10.1002/14356007.a01_305
- "Glutaric acid, Pentanedioic acid, 99%". Chemkits.eu. Retrieved 2020-09-29.
- US 4046817, Shipchandler, Mohammed T., "Method of synthesis of pyrogallol", published 1977-09-06, assigned to IMC Chemical Group
- ^ Glutaric acid, cameochemicals.com
External links
| Linear saturated dicarboxylic acids (HO2C-R-CO2H) | |
|---|---|
| |
| Category:Dicarboxylic acids |