| |
| Names | |
|---|---|
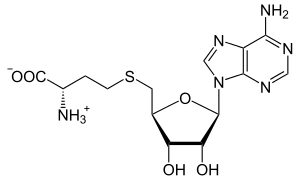
| IUPAC name S-(5′-Deoxyadenos-5′-yl)-L-homocysteine | |
| Systematic IUPAC name (2S)-2-Amino-4-({methyl}sulfanyl)butanoic acid | |
| Other names
AdoHcy, 2-S-adenosyl-L-homocysteine, 5′-S-(3-Amino-3-carboxypropyl)-5′-thioadenosine S-adenosylhomocysteine, SAH | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.012.328 |
| IUPHAR/BPS | |
| KEGG | |
| MeSH | S-Adenosylhomocysteine |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C14H20N6O5S |
| Molar mass | 384.41 g·mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
S-Adenosyl-L-homocysteine (SAH) is the biosynthetic precursor to homocysteine. SAH is formed by the demethylation of S-adenosyl-L-methionine. Adenosylhomocysteinase converts SAH into homocysteine and adenosine.
Biological role
DNA methyltransferases are inhibited by SAH. Two S-adenosyl-L-homocysteine cofactor products can bind the active site of DNA methyltransferase 3B and prevent the DNA duplex from binding to the active site, which inhibits DNA methylation.
References
- Finkelstein JD (2000). "Pathways and regulation of homocysteine metabolism in mammals". Seminars in Thrombosis and Hemostasis. 26 (3): 219–225. doi:10.1055/s-2000-8466. PMID 11011839.
- Ribbe MW, Hu Y, Hodgson KO, Hedman B (April 2014). "Biosynthesis of nitrogenase metalloclusters". Chemical Reviews. 114 (8): 4063–4080. doi:10.1021/cr400463x. PMC 3999185. PMID 24328215.
- James SJ, Melnyk S, Pogribna M, Pogribny IP, Caudill MA (August 2002). "Elevation in S-adenosylhomocysteine and DNA hypomethylation: potential epigenetic mechanism for homocysteine-related pathology". The Journal of Nutrition. 132 (8 Suppl): 2361S–2366S. doi:10.1093/jn/132.8.2361S. PMID 12163693.
- Kumar R, Srivastava R, Singh RK, Surolia A, Rao DN (March 2008). "Activation and inhibition of DNA methyltransferases by S-adenosyl-L-homocysteine analogues". Bioorganic & Medicinal Chemistry. 16 (5): 2276–2285. doi:10.1016/j.bmc.2007.11.075. PMID 18083524.
- Lin CC, Chen YP, Yang WZ, Shen JC, Yuan HS (April 2020). "Structural insights into CpG-specific DNA methylation by human DNA methyltransferase 3B". Nucleic Acids Research. 48 (7): 3949–3961. doi:10.1093/nar/gkaa111. PMC 7144912. PMID 32083663.
External links
This biochemistry article is a stub. You can help Misplaced Pages by expanding it. |