| Revision as of 20:11, 22 February 2011 editCheMoBot (talk | contribs)Bots141,565 edits Updating {{drugbox}} (no changed fields - added verified revid - updated 'UNII_Ref', 'ChemSpiderID_Ref', 'StdInChI_Ref', 'StdInChIKey_Ref', 'ChEMBL_Ref', 'KEGG_Ref') per Chem/Drugbox validation (← Previous edit | Revision as of 10:38, 9 May 2011 edit undoRod57 (talk | contribs)Autopatrolled, Extended confirmed users, Pending changes reviewers32,793 edits →Clinical use: reword out of date textNext edit → | ||
| Line 47: | Line 47: | ||
| {{main|Cephalosporin}} | {{main|Cephalosporin}} | ||
| Ceftaroline is a novel cephalosporin that has activity against MRSA with ] for complicated skin and skin structure infections with reported non-inferior efficacy against MRSA compared to ] and ]<ref name="corey2008">{{cite conference| first = Corey| last = R| coauthors = Wilcox M, Talbot GH, et al.| title = CANVAS-1: Randomized, Double-blinded, Phase 3 Study (P903-06) of the Efficacy and Safety of Ceftaroline vs. Vancomycin plus Aztreonam in Complicated Skin and Skin Structure Infections (cSSSI).| conference = 2008 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference}}</ref><ref name="pmid19207097">{{cite journal| author = Kanafani ZA, Corey GR| title = Ceftaroline: a cephalosporin with expanded Gram-positive activity| journal = Future Microbiology| volume = 4| pages = 25–33| year = 2009| month = February| pmid = 19207097| doi = 10.2217/17460913.4.1.25| url = http://www.futuremedicine.com/doi/abs/10.2217/17460913.4.1.25?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%3dncbi.nlm.nih.gov}}</ref> Recently,{{When|date=February 2011}} ceftaroline has completed phase III clinical trials for community-acquired pneumonia comparing it against ] with non-inferior results and similar adverse reaction profile.<ref name="eckberg2009">{{cite conference| first = Eckberg| last = P| coauthors = Friedland HD, et al.| title = FOCUS 1 and 2: Randomized, Double-blinded, Multicenter Phase 3 Trials of the Efficacy and Safety of Ceftaroline (CPT) vs. Ceftriaxone (CRO) in Community-acquired pneumonia (CAP).| conference = 2009 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference}}</ref> However there are only published results for Phase II clinical trials in treatment of complicated skin and skin structure infections.<ref name="pmid17682094">{{cite journal| author = Talbot GH, Thye D, Das A, Ge Y| title = Phase 2 study of ceftaroline versus standard therapy in treatment of complicated skin and skin structure infections| journal = ]| volume = 51| issue = 10| pages = 3612–6| year = 2007| month = October| pmid = 17682094| pmc = 2043268| doi = 10.1128/AAC.00590-07| url = http://aac.asm.org/cgi/pmidlookup?view=long&pmid=17682094}}</ref> |
Ceftaroline is a novel cephalosporin that has activity against MRSA with ] for complicated skin and skin structure infections with reported non-inferior efficacy against MRSA compared to ] and ]<ref name="corey2008">{{cite conference| first = Corey| last = R| coauthors = Wilcox M, Talbot GH, et al.| title = CANVAS-1: Randomized, Double-blinded, Phase 3 Study (P903-06) of the Efficacy and Safety of Ceftaroline vs. Vancomycin plus Aztreonam in Complicated Skin and Skin Structure Infections (cSSSI).| conference = 2008 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference}}</ref><ref name="pmid19207097">{{cite journal| author = Kanafani ZA, Corey GR| title = Ceftaroline: a cephalosporin with expanded Gram-positive activity| journal = Future Microbiology| volume = 4| pages = 25–33| year = 2009| month = February| pmid = 19207097| doi = 10.2217/17460913.4.1.25| url = http://www.futuremedicine.com/doi/abs/10.2217/17460913.4.1.25?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%3dncbi.nlm.nih.gov}}</ref> Recently,{{When|date=February 2011}} ceftaroline has completed phase III clinical trials for ] comparing it against ] with non-inferior results and similar adverse reaction profile.<ref name="eckberg2009">{{cite conference| first = Eckberg| last = P| coauthors = Friedland HD, et al.| title = FOCUS 1 and 2: Randomized, Double-blinded, Multicenter Phase 3 Trials of the Efficacy and Safety of Ceftaroline (CPT) vs. Ceftriaxone (CRO) in Community-acquired pneumonia (CAP).| conference = 2009 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference}}</ref> However there are only published results for Phase II clinical trials in treatment of complicated skin and skin structure infections.<ref name="pmid17682094">{{cite journal| author = Talbot GH, Thye D, Das A, Ge Y| title = Phase 2 study of ceftaroline versus standard therapy in treatment of complicated skin and skin structure infections| journal = ]| volume = 51| issue = 10| pages = 3612–6| year = 2007| month = October| pmid = 17682094| pmc = 2043268| doi = 10.1128/AAC.00590-07| url = http://aac.asm.org/cgi/pmidlookup?view=long&pmid=17682094}}</ref> Sept 2009 : Phase III trials results reported.<ref name="forest2009">{{cite press release |title=Forest Laboratories Presents Analysis of Two Positive Pivotal Phase III Studies of Ceftaroline for the Treatment of Community-Acquired Pneumonia (CAP) at ICAAC |url=http://www.businesswire.com/news/home/20090912005009/en |publisher=BUSINESS WIRE |date=2009-09-12 |accessdate=2009-10-19}}</ref> | ||
| On September 8, 2010 the FDA Advisory Committee recommended approval for the treatment of community acquired bacterial pneumonia and complicated skin and skin structure infections.<ref>{{Cite web |url=http://www.drugs.com/newdrugs.html |title=New Drug Approvals |date=2010-10-29 |accessdate=2010-11-08}}</ref> | |||
| In Oct 2010 the FDA approval was gained for ] (CABP) and acute bacterial ]s (ABSSSI), including methicillin-resistant Staphylococcus aureus (MRSA).<ref>{{cite news |url=http://www.ibtimes.com/articles/77358/20101030/fda-teflaro-forests-laboratories-inc-bacterial-infections.htm |title=FDA approves Teflaro for treatment of bacterial infections |date=30 Oct 2010 }}</ref> | |||
| On November 1, 2010 the FDA approved ceftaroline for bacterial infections. Also, on September 8, 2010 the FDA Advisory Committee recommended approval for the treatment of community acquired bacterial pneumonia and complicated skin and skin structure infections.<ref>{{Cite web |url=http://www.drugs.com/newdrugs.html |title=New Drug Approvals |date=2010-10-29 |accessdate=2010-11-08}}</ref> | |||
| == Safety == | == Safety == | ||
Revision as of 10:38, 9 May 2011
Pharmaceutical compound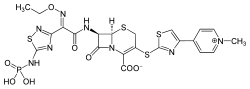 | |
| Clinical data | |
|---|---|
| Other names | PPI 0903, TAK-599 |
| License data |
|
| Routes of administration | Intravenous, Intramuscular |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 2.13-2.89 hours |
| Excretion | 46.4% Kidney, renal |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H25N8O10PS4 |
| Molar mass | 744.736661 g/mol g·mol |
| 3D model (JSmol) | |
SMILES
| |
| (verify) | |
Ceftaroline fosamil (INN) (Template:Pron-en, brand name Teflaro) is an advanced generation cephalosporin antibiotic. It is active against methicillin-resistant Staphylococcus aureus (MRSA) and Gram positive bacteria. It retains the activity of later generation cephalosporins having broad spectrum activity against Gram negative bacteria. It is currently being investigated for community-acquired pneumonia and complicated skin and skin structure infection.
Ceftaroline is being developed by Forest Laboratories, under a license from Takeda. Ceftaroline has received approval from the U.S. Food and Drug Administration for the treatment of community-acquired bacterial pneumonia and acute bacterial skin infections on October 29, 2010. In vitro studies show that it has a similar spectrum to ceftobiprole, the only other fifth-generation cephalosporin to date, although no head to head clinical trials have been conducted. Currently, ceftaroline and ceftobiprole are on an unnamed subclass of cephalosporins by the Clinical and Laboratory Standards Institute (CLSI).
Mechanism of action
Beta-lactam antibiotics inhibit bacterial peptidoglycan synthesis by binding the penicillin binding proteins (PBPs) in the bacterial cell wall. Inhibition of PBPs leads to irregularities in cell wall structures, such as elongation, lesions, loss of selective permeability, and eventual cell death and lysis. In particular, ceftaroline can effectively bind to and inhibit PBP-2a, the type of PBP produced by MRSA and not well inhibited by other antibiotics currently in clinical use.
Clinical use
Main article: CephalosporinCeftaroline is a novel cephalosporin that has activity against MRSA with phase III clinical trials for complicated skin and skin structure infections with reported non-inferior efficacy against MRSA compared to vancomycin and aztreonam Recently, ceftaroline has completed phase III clinical trials for community-acquired pneumonia comparing it against ceftriaxone with non-inferior results and similar adverse reaction profile. However there are only published results for Phase II clinical trials in treatment of complicated skin and skin structure infections. Sept 2009 : Phase III trials results reported. On September 8, 2010 the FDA Advisory Committee recommended approval for the treatment of community acquired bacterial pneumonia and complicated skin and skin structure infections. In Oct 2010 the FDA approval was gained for community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI), including methicillin-resistant Staphylococcus aureus (MRSA).
Safety
The clinical studies indicated that ceftaroline was well-tolerated. The overall rate of adverse events was comparable between the two treatment groups (The CANVAS I and CANVAS II trials evaluated ceftaroline monotherapy versus vancomycin plus aztreonam in adult patients with complicated skin and skin structure infections caused by gram-positive and gram-negative bacteria.). The overall discontinuation rate for ceftaroline-treated patients was 2.7% compared to a rate of 3.7% for the comparator group-treated patients. The most common adverse reactions occurring in > 2% of patients receiving ceftaroline in the pooled Phase 3 clinical trials were diarrhea, nausea, and rash.:
Contraindications
- Known serious hypersensitivity to ceftaroline or other members of the cephalosporin class.
- Anaphylaxis and anaphylactoid reactions have been reported with ceftaroline.
Warnings and precautions
The warnings and precautions associated with ceftaroline include:
Hypersensitivity reactions
Serious hypersensitivity (anaphylactic) reactions and serious skin reactions have been reported with beta-lactam antibiotics, including ceftaroline. Exercise caution in patients with known hypersensitivity to beta-lactam antibiotics including ceftaroline. Before therapy with ceftaroline is instituted, careful inquiry about previous hypersensitivity reactions to other cephalosporins, penicillins, or carbapenems should be made. If this product is to be given to penicillin or other beta-lactam-allergic patient, caution should be exercised because cross sensitivity among beta-lactam antibacterial agents has been clearly established. If an allergic reaction to ceftaroline occurs, the drug should be discontinued. Serious acute hypersensitivity (anaphylactic) reactions require emergency treatment with epinephrine and other emergency measures, that may include airway management, oxygen, intravenous fluids, antihistamines, corticosteroids, and vasopressors as clinically indicated.
Clostridium difficile associated diarrhea
Clostridium difficile associated diarrhea (CDAD) has been reported for nearly all antibacterial agents including ceftaroline, and may range in severity from mild diarrhea to fatal colitis. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents. If CDAD is suspected or confirmed, antibacterials not directed against C. difficile should be discontinued, if possible.
Development of drug-resistant bacteria
Prescribing ceftaroline in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Direct Coombs test seroconversion
In the pooled Phase 3 CABP trials, 51/520 (9.8%) of patients treated with ceftaroline compared to 24/534 (4.5%) of patients treated with ceftriaxone seroconverted from a negative to a positive direct Coombs' test result. No clinical adverse reactions representing hemolytic anemia were reported in any treatment group. If anemia develops during or after treatment with ceftaroline, drug-induced hemolytic anemia should be considered. If drug-induced hemolytic anemia is suspected, discontinuation of ceftaroline should be considered and supportive care should be administered to the patient if clinically indicated.
Interactions
No clinical drug-drug interaction studies have been conducted with ceftaroline fosamil. In vitro studies in human liver microsomes indicated that neither ceftaroline fosamil nor ceftaroline inhibits the major cytochrome P450 isoenzymes. Therefore neither ceftaroline fosamil nor ceftaroline are expected to inhibit or induce the clearance of drugs that are metabolized by these metabolic pathways in a clinically relevant manner.
Use in specific populations
For pregnant or nursing mothers, ceftaroline fosamil should only be used if the potential benefit outweighs the potential risk to the fetus or child.
Safety and effectiveness in pediatric patients has not been studied.
Because elderly patients greater-than or equal to 65 years of age are more likely to have decreased renal function and ceftaroline is excreted primarily by the kidney, care should be taken in dose selection in this age group as in younger patients with impaired renal function
Dosage adjustment is required in patients with moderately (30 to ‰¤ 50 mL/min) or severely (< 30 mL/min) impaired renal function
The pharmacokinetics of ceftaroline in patients with hepatic impairment have not been established
Side effects
No adverse reactions occurred in greater than 5% of patients receiving ceftaroline. The most common adverse reactions occurring in > 2% of patients receiving ceftaroline in the pooled Phase 3 clinical trials were:
Chemistry
Ceftaroline fosamil is a prodrug that is converted to active metabolite ceftaroline and inactive metabolite ceftaroline-M1. Initial in vitro and in vivo animal studies referred to ceftaroline fosamil acetate as PPI-0903.
Characteristic of cephalosporins, ceftaroline has a bicyclic ring with four member β-lactam ring fused to a six member cephem ring. Ceftaroline is thought to have activity against MRSA with its 1,3 thiazole ring.
References
- Kollef MH (2009). "New antimicrobial agents for methicillin-resistant Staphylococcus aureus". Crit Care Resusc. 11 (4): 282–6. PMID 20001879.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ P, Eckberg. FOCUS 1 and 2: Randomized, Double-blinded, Multicenter Phase 3 Trials of the Efficacy and Safety of Ceftaroline (CPT) vs. Ceftriaxone (CRO) in Community-acquired pneumonia (CAP). 2009 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference.
{{cite conference}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ R, Corey. CANVAS-1: Randomized, Double-blinded, Phase 3 Study (P903-06) of the Efficacy and Safety of Ceftaroline vs. Vancomycin plus Aztreonam in Complicated Skin and Skin Structure Infections (cSSSI). 2008 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference.
{{cite conference}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Kanafani ZA, Corey GR (2009). "Ceftaroline: a cephalosporin with expanded Gram-positive activity". Future Microbiology. 4: 25–33. doi:10.2217/17460913.4.1.25. PMID 19207097.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Parish D, Scheinfeld N (2008). "Ceftaroline fosamil, a cephalosporin derivative for the potential treatment of MRSA infection". Current Opinion in Investigational Drugs (London, England : 2000). 9 (2): 201–9. PMID 18246523.
{{cite journal}}: Unknown parameter|month=ignored (help) - "Forest Announces FDA Approval of Teflaro(TM) (ceftaroline fosamil) for the Treatment of Community-Acquired Bacterial Pneumonia and Acute Bacterial Skin and Skin Structure Infection" (Press release). Forest Laboratories. 2010-10-29. Retrieved October 30, 2010.
- Villegas-Estrada A, Lee M, Hesek D, Vakulenko SB, Mobashery S (2008). "Co-opting the cell wall in fighting methicillin-resistant Staphylococcus aureus: potent inhibition of PBP 2a by two anti-MRSA beta-lactam antibiotics". Journal of the American Chemical Society. 130 (29): 9212–3. doi:10.1021/ja8029448. PMC 2747592. PMID 18582062.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - Zhanel GG, Sniezek G, Schweizer F, Zelenitsky S, Lagacé-Wiens PR, Rubinstein E, Gin AS, Hoban DJ, Karlowsky JA (2009). "Ceftaroline: a novel broad-spectrum cephalosporin with activity against meticillin-resistant Staphylococcus aureus". Drugs. 69 (7): 809–31. doi:10.2165/00003495-200969070-00003. PMID 19441869.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Talbot GH, Thye D, Das A, Ge Y (2007). "Phase 2 study of ceftaroline versus standard therapy in treatment of complicated skin and skin structure infections". Antimicrobial Agents and Chemotherapy. 51 (10): 3612–6. doi:10.1128/AAC.00590-07. PMC 2043268. PMID 17682094.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - "Forest Laboratories Presents Analysis of Two Positive Pivotal Phase III Studies of Ceftaroline for the Treatment of Community-Acquired Pneumonia (CAP) at ICAAC" (Press release). BUSINESS WIRE. 2009-09-12. Retrieved 2009-10-19.
- "New Drug Approvals". 2010-10-29. Retrieved 2010-11-08.
- "FDA approves Teflaro for treatment of bacterial infections". 30 Oct 2010.
- ^ "Teflaro". 2010-10-29. Retrieved 2010-11-08.
- Y, Ge. The pharmacokinetics and safety of ceftaroline (PPI-0903) in healthy subjects receiving multiple-dose intravenous infusions. 2006 Interscience Conference on Antimicrobial Agents and Chemotherapy / Infectious Disease Society of America Conference.
{{cite conference}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - Yukihiro I, Junko B (2008). "Stability and Stabilization Studies of TAK-599 (Ceftaroline Fosamil) a Novel N-Phosphono Type Prodrug of Anti-methicillin Resistant Staphylococcus aureus Cephalosporin T-91825". Chem Pharm Bull. 56 (10): 1406–11. doi:10.1248/cpb.56.1406. PMID 18827379.
- Ishikawa t, Nobuyuki M; et al. (2003). "TAK-599, a novel N-phosphono type prodrug of anti-MRSA cephalosporin T-91825: Synthesis, physicochemical and pharmacological properties". Bioorg Med Chem. 11 (11): 2427–2437. PMID 12735989.
{{cite journal}}: Explicit use of et al. in:|author=(help)