
Micro ribonucleic acid (microRNA, miRNA, μRNA) are small, single-stranded, non-coding RNA molecules containing 21–23 nucleotides. Found in plants, animals, and even some viruses, miRNAs are involved in RNA silencing and post-transcriptional regulation of gene expression. miRNAs base-pair to complementary sequences in messenger RNA (mRNA) molecules, then silence said mRNA molecules by one or more of the following processes:
- Cleaving the mRNA strand into two pieces.
- Destabilizing the mRNA by shortening its poly(A) tail.
- Reducing translation of the mRNA into proteins.
In cells of humans and other animals, miRNAs primarily act by destabilizing the mRNA.
miRNAs resemble the small interfering RNAs (siRNAs) of the RNA interference (RNAi) pathway, except miRNAs derive from regions of RNA transcripts that fold back on themselves to form short hairpins, whereas siRNAs derive from longer regions of double-stranded RNA. The human genome may encode over 1900 miRNAs, However, only about 500 human miRNAs represent bona fide miRNAs in the manually curated miRNA gene database MirGeneDB.
miRNAs are abundant in many mammalian cell types. They appear to target about 60% of the genes of humans and other mammals. Many miRNAs are evolutionarily conserved, which implies that they have important biological functions. For example, 90 families of miRNAs have been conserved since at least the common ancestor of mammals and fish, and most of these conserved miRNAs have important functions, as shown by studies in which genes for one or more members of a family have been knocked out in mice.
In 2024, American scientists Victor Ambros and Gary Ruvkun were awarded the Nobel Prize in Physiology or Medicine for their work on the discovery of miRNA and its role in post-transcriptional gene regulation.
History
The first miRNA was discovered in the early 1990s. However, they were not recognized as a distinct class of biological regulators until the early 2000s. Research revealed different sets of miRNAs expressed in different cell types and tissues and multiple roles for miRNAs in plant and animal development and in many other biological processes. Aberrant miRNA expression are implicated in disease states. MiRNA-based therapies are under investigation.
The first miRNA was discovered in 1993 by a group led by Victor Ambros and including Lee and Feinbaum. However, additional insight into its mode of action required simultaneously published work by Gary Ruvkun's team, including Wightman and Ha. These groups published back-to-back papers on the lin-4 gene, which was known to control the timing of C. elegans larval development by repressing the lin-14 gene. When Lee et al. isolated the lin-4 miRNA, they found that instead of producing an mRNA encoding a protein, it produced short non-coding RNAs, one of which was a ~22-nucleotide RNA that contained sequences partially complementary to multiple sequences in the 3' UTR of the lin-14 mRNA. This complementarity was proposed to inhibit the translation of the lin-14 mRNA into the LIN-14 protein. At the time, the lin-4 small RNA was thought to be a nematode idiosyncrasy.
In 2000, a second small RNA was characterized: let-7 RNA, which represses lin-41 to promote a later developmental transition in C. elegans. The let-7 RNA was found to be conserved in many species, leading to the suggestion that let-7 RNA and additional "small temporal RNAs" might regulate the timing of development in diverse animals, including humans.
A year later, the lin-4 and let-7 RNAs were found to be part of a large class of small RNAs present in C. elegans, Drosophila and human cells. The many RNAs of this class resembled the lin-4 and let-7 RNAs, except their expression patterns were usually inconsistent with a role in regulating the timing of development. This suggested that most might function in other types of regulatory pathways. At this point, researchers started using the term "microRNA" to refer to this class of small regulatory RNAs.
The first human disease associated with deregulation of miRNAs was chronic lymphocytic leukemia. In this disorder, the miRNAs have a dual role working as both tumor suppressors and oncogenes.
Nomenclature
Under a standard nomenclature system, names are assigned to experimentally confirmed miRNAs before publication. The prefix "miR" is followed by a dash and a number, the latter often indicating order of naming. For example, miR-124 was named and likely discovered prior to miR-456. A capitalized "miR-" refers to the mature form of the miRNA, while the uncapitalized "mir-" refers to the pre-miRNA and the pri-miRNA. The genes encoding miRNAs are also named using the same three-letter prefix according to the conventions of the organism gene nomenclature. For examples, the official miRNAs gene names in some organisms are "mir-1 in C. elegans and Drosophila, Mir1 in Rattus norvegicus and MIR25 in human.
miRNAs with nearly identical sequences except for one or two nucleotides are annotated with an additional lower case letter. For example, miR-124a is closely related to miR-124b. For example:
- hsa-miR-181a: aacauucaACgcugucggugAgu
- hsa-miR-181b: aacauucaUUgcugucggugGgu
Pre-miRNAs, pri-miRNAs and genes that lead to 100% identical mature miRNAs but that are located at different places in the genome are indicated with an additional dash-number suffix. For example, the pre-miRNAs hsa-mir-194-1 and hsa-mir-194-2 lead to an identical mature miRNA (hsa-miR-194) but are from genes located in different genome regions.
Species of origin is designated with a three-letter prefix, e.g., hsa-miR-124 is a human (Homo sapiens) miRNA and oar-miR-124 is a sheep (Ovis aries) miRNA. Other common prefixes include "v" for viral (miRNA encoded by a viral genome) and "d" for Drosophila miRNA (a fruit fly commonly studied in genetic research).
When two mature microRNAs originate from opposite arms of the same pre-miRNA and are found in roughly similar amounts, they are denoted with a -3p or -5p suffix. (In the past, this distinction was also made with "s" (sense) and "as" (antisense)). However, the mature microRNA found from one arm of the hairpin is usually much more abundant than that found from the other arm, in which case, an asterisk following the name indicates the mature species found at low levels from the opposite arm of a hairpin. For example, miR-124 and miR-124* share a pre-miRNA hairpin, but much more miR-124 is found in the cell.
Targets
Plant miRNAs usually have near-perfect pairing with their mRNA targets, which induces gene repression through cleavage of the target transcripts. In contrast, animal miRNAs are able to recognize their target mRNAs by using as few as 6–8 nucleotides (the seed region) at the 5' end of the miRNA, which is not enough pairing to induce cleavage of the target mRNAs. Combinatorial regulation is a feature of miRNA regulation in animals. A given miRNA may have hundreds of different mRNA targets, and a given target might be regulated by multiple miRNAs.
Estimates of the average number of unique messenger RNAs that are targets for repression by a typical miRNA vary, depending on the estimation method, but multiple approaches show that mammalian miRNAs can have many unique targets. For example, an analysis of the miRNAs highly conserved in vertebrates shows that each has, on average, roughly 400 conserved targets. Likewise, experiments show that a single miRNA species can reduce the stability of hundreds of unique messenger RNAs. Other experiments show that a single miRNA species may repress the production of hundreds of proteins, but that this repression often is relatively mild (much less than 2-fold).
Biogenesis

As many as 40% of miRNA genes may lie in the introns or even exons of other genes. These are usually, though not exclusively, found in a sense orientation, and thus usually are regulated together with their host genes.
The DNA template is not the final word on mature miRNA production: 6% of human miRNAs show RNA editing (IsomiRs), the site-specific modification of RNA sequences to yield products different from those encoded by their DNA. This increases the diversity and scope of miRNA action beyond that implicated from the genome alone.
Transcription
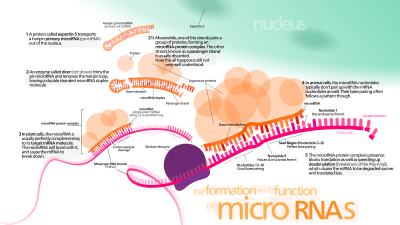
miRNA genes are usually transcribed by RNA polymerase II (Pol II). The polymerase often binds to a promoter found near the DNA sequence, encoding what will become the hairpin loop of the pre-miRNA. The resulting transcript is capped with a specially modified nucleotide at the 5' end, polyadenylated with multiple adenosines (a poly(A) tail), and spliced. Animal miRNAs are initially transcribed as part of one arm of an ~80 nucleotide RNA stem-loop that in turn forms part of a several hundred nucleotide-long miRNA precursor termed a pri-miRNA. When a stem-loop precursor is found in the 3' UTR, a transcript may serve as a pri-miRNA and a mRNA. RNA polymerase III (Pol III) transcribes some miRNAs, especially those with upstream Alu sequences, transfer RNAs (tRNAs), and mammalian wide interspersed repeat (MWIR) promoter units.
Nuclear processing

A single pri-miRNA may contain from one to six miRNA precursors. These hairpin loop structures are composed of about 70 nucleotides each. Each hairpin is flanked by sequences necessary for efficient processing.
The double-stranded RNA (dsRNA) structure of the hairpins in a pri-miRNA is recognized by a nuclear protein known as DiGeorge Syndrome Critical Region 8 (DGCR8 or "Pasha" in invertebrates), named for its association with DiGeorge Syndrome. DGCR8 associates with the enzyme Drosha, a protein that cuts RNA, to form the Microprocessor complex. In this complex, DGCR8 orients the catalytic RNase III domain of Drosha to liberate hairpins from pri-miRNAs by cleaving RNA about eleven nucleotides from the hairpin base (one helical dsRNA turn into the stem). The product resulting has a two-nucleotide overhang at its 3' end; it has 3' hydroxyl and 5' phosphate groups. It is often termed as a pre-miRNA (precursor-miRNA). Sequence motifs downstream of the pre-miRNA that are important for efficient processing have been identified.
Pre-miRNAs that are spliced directly out of introns, bypassing the Microprocessor complex, are known as "mirtrons." Mirtrons have been found in Drosophila, C. elegans, and mammals.
As many as 16% of pre-miRNAs may be altered through nuclear RNA editing. Most commonly, enzymes known as adenosine deaminases acting on RNA (ADARs) catalyze adenosine to inosine (A to I) transitions. RNA editing can halt nuclear processing (for example, of pri-miR-142, leading to degradation by the ribonuclease Tudor-SN) and alter downstream processes including cytoplasmic miRNA processing and target specificity (e.g., by changing the seed region of miR-376 in the central nervous system).
Nuclear export

Pre-miRNA hairpins are exported from the nucleus in a process involving the nucleocytoplasmic shuttler Exportin-5. This protein, a member of the karyopherin family, recognizes a two-nucleotide overhang left by the RNase III enzyme Drosha at the 3' end of the pre-miRNA hairpin. Exportin-5-mediated transport to the cytoplasm is energy-dependent, using guanosine triphosphate (GTP) bound to the Ran protein.
Cytoplasmic processing
In the cytoplasm, the pre-miRNA hairpin is cleaved by the RNase III enzyme Dicer. This endoribonuclease interacts with 5' and 3' ends of the hairpin and cuts away the loop joining the 3' and 5' arms, yielding an imperfect miRNA:miRNA* duplex about 22 nucleotides in length. Overall hairpin length and loop size influence the efficiency of Dicer processing. The imperfect nature of the miRNA:miRNA* pairing also affects cleavage. Some of the G-rich pre-miRNAs can potentially adopt the G-quadruplex structure as an alternative to the canonical stem-loop structure. For example, human pre-miRNA 92b adopts a G-quadruplex structure which is resistant to the Dicer mediated cleavage in the cytoplasm. Although either strand of the duplex may potentially act as a functional miRNA, only one strand is usually incorporated into the RNA-induced silencing complex (RISC) where the miRNA and its mRNA target interact.
While the majority of miRNAs are located within the cell, some miRNAs, commonly known as circulating miRNAs or extracellular miRNAs, have also been found in extracellular environment, including various biological fluids and cell culture media.
Biogenesis in plants
miRNA biogenesis in plants differs from animal biogenesis mainly in the steps of nuclear processing and export. Instead of being cleaved by two different enzymes, once inside and once outside the nucleus, both cleavages of the plant miRNA are performed by a Dicer homolog, called Dicer-like1 (DL1). DL1 is expressed only in the nucleus of plant cells, which indicates that both reactions take place inside the nucleus. Before plant miRNA:miRNA* duplexes are transported out of the nucleus, its 3' overhangs are methylated by a RNA methyltransferaseprotein called Hua-Enhancer1 (HEN1). The duplex is then transported out of the nucleus to the cytoplasm by a protein called Hasty (HST), an Exportin 5 homolog, where they disassemble and the mature miRNA is incorporated into the RISC.
RNA-induced silencing complex
Main article: RNA-induced silencing complexThe mature miRNA is part of an active RNA-induced silencing complex (RISC) containing Dicer and many associated proteins. RISC is also known as a microRNA ribonucleoprotein complex (miRNP); A RISC with incorporated miRNA is sometimes referred to as a "miRISC."
Dicer processing of the pre-miRNA is thought to be coupled with unwinding of the duplex. Generally, only one strand is incorporated into the miRISC, selected on the basis of its thermodynamic instability and weaker base-pairing on the 5' end relative to the other strand. The position of the stem-loop may also influence strand choice. The other strand, called the passenger strand due to its lower levels in the steady state, is denoted with an asterisk (*) and is normally degraded. In some cases, both strands of the duplex are viable and become functional miRNA that target different mRNA populations.
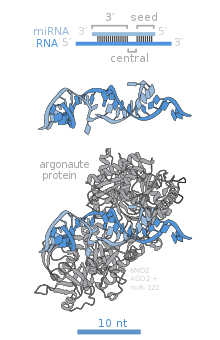
Members of the Argonaute (Ago) protein family are central to RISC function. Argonautes are needed for miRNA-induced silencing and contain two conserved RNA binding domains: a PAZ domain that can bind the single stranded 3' end of the mature miRNA and a PIWI domain that structurally resembles ribonuclease-H and functions to interact with the 5' end of the guide strand. They bind the mature miRNA and orient it for interaction with a target mRNA. Some argonautes, for example human Ago2, cleave target transcripts directly; argonautes may also recruit additional proteins to achieve translational repression. The human genome encodes eight argonaute proteins divided by sequence similarities into two families: AGO (with four members present in all mammalian cells and called E1F2C/hAgo in humans), and PIWI (found in the germline and hematopoietic stem cells).
Additional RISC components include TRBP , PACT (protein activator of the interferon-induced protein kinase), the SMN complex, fragile X mental retardation protein (FMRP), Tudor staphylococcal nuclease-domain-containing protein (Tudor-SN), the putative DNA helicase MOV10, and the RNA recognition motif containing protein TNRC6B.
Mode of silencing and regulatory loops
Gene silencing may occur either via mRNA degradation or preventing mRNA from being translated. For example, miR16 contains a sequence complementary to the AU-rich element found in the 3'UTR of many unstable mRNAs, such as TNF alpha or GM-CSF. It has been demonstrated that given complete complementarity between the miRNA and target mRNA sequence, Ago2 can cleave the mRNA and lead to direct mRNA degradation. In the absence of complementarity, silencing is achieved by preventing translation. The relation of miRNA and its target mRNA can be based on the simple negative regulation of a target mRNA, but it seems that a common scenario is the use of a "coherent feed-forward loop", "mutual negative feedback loop" (also termed double negative loop) and "positive feedback/feed-forward loop". Some miRNAs work as buffers of random gene expression changes arising due to stochastic events in transcription, translation and protein stability. Such regulation is typically achieved by the virtue of negative feedback loops or incoherent feed-forward loop uncoupling protein output from mRNA transcription.
Turnover
Turnover of mature miRNA is needed for rapid changes in miRNA expression profiles. During miRNA maturation in the cytoplasm, uptake by the Argonaute protein is thought to stabilize the guide strand, while the opposite (* or "passenger") strand is preferentially destroyed. In what has been called a "Use it or lose it" strategy, Argonaute may preferentially retain miRNAs with many targets over miRNAs with few or no targets, leading to degradation of the non-targeting molecules.
Decay of mature miRNAs in Caenorhabditis elegans is mediated by the 5'-to-3' exoribonuclease XRN2, also known as Rat1p. In plants, SDN (small RNA degrading nuclease) family members degrade miRNAs in the opposite (3'-to-5') direction. Similar enzymes are encoded in animal genomes, but their roles have not been described.
Several miRNA modifications affect miRNA stability. As indicated by work in the model organism Arabidopsis thaliana (thale cress), mature plant miRNAs appear to be stabilized by the addition of methyl moieties at the 3' end. The 2'-O-conjugated methyl groups block the addition of uracil (U) residues by uridyltransferase enzymes, a modification that may be associated with miRNA degradation. However, uridylation may also protect some miRNAs; the consequences of this modification are incompletely understood. Uridylation of some animal miRNAs has been reported. Both plant and animal miRNAs may be altered by addition of adenine (A) residues to the 3' end of the miRNA. An extra A added to the end of mammalian miR-122, a liver-enriched miRNA important in hepatitis C, stabilizes the molecule and plant miRNAs ending with an adenine residue have slower decay rates.
Cellular functions

The function of miRNAs appears to be in gene regulation. For that purpose, a miRNA is complementary to a part of one or more messenger RNAs (mRNAs). Animal miRNAs are usually complementary to a site in the 3' UTR whereas plant miRNAs are usually complementary to coding regions of mRNAs. Perfect or near perfect base pairing with the target RNA promotes cleavage of the RNA. This is the primary mode of plant miRNAs. In animals the match-ups are imperfect.
For partially complementary microRNAs to recognise their targets, nucleotides 2–7 of the miRNA (its 'seed region') must be perfectly complementary. Animal miRNAs inhibit protein translation of the target mRNA (this is present but less common in plants). Partially complementary microRNAs can also speed up deadenylation, causing mRNAs to be degraded sooner. While degradation of miRNA-targeted mRNA is well documented, whether or not translational repression is accomplished through mRNA degradation, translational inhibition, or a combination of the two is hotly debated. Recent work on miR-430 in zebrafish, as well as on bantam-miRNA and miR-9 in Drosophila cultured cells, shows that translational repression is caused by the disruption of translation initiation, independent of mRNA deadenylation.
miRNAs occasionally also cause histone modification and DNA methylation of promoter sites, which affects the expression of target genes.
Nine mechanisms of miRNA action are described and assembled in a unified mathematical model:
- Cap-40S initiation inhibition;
- 60S Ribosomal unit joining inhibition;
- Elongation inhibition;
- Ribosome drop-off (premature termination);
- Co-translational nascent protein degradation;
- Sequestration in P-bodies;
- mRNA decay (destabilisation);
- mRNA cleavage;
- Transcriptional inhibition through microRNA-mediated chromatin reorganization followed by gene silencing.
It is often impossible to discern these mechanisms using experimental data about stationary reaction rates. Nevertheless, they are differentiated in dynamics and have different kinetic signatures.
Unlike plant microRNAs, the animal microRNAs target diverse genes. However, genes involved in functions common to all cells, such as gene expression, have relatively fewer microRNA target sites and seem to be under selection to avoid targeting by microRNAs. There is a strong correlation between ITPR gene regulations and mir-92 and mir-19.
dsRNA can also activate gene expression, a mechanism that has been termed "small RNA-induced gene activation" or RNAa. dsRNAs targeting gene promoters can induce potent transcriptional activation of associated genes. This was demonstrated in human cells using synthetic dsRNAs termed small activating RNAs (saRNAs), but has also been demonstrated for endogenous microRNA.
Interactions between microRNAs and complementary sequences on genes and even pseudogenes that share sequence homology are thought to be a back channel of communication regulating expression levels between paralogous genes (genes having a similar structure indicating divergence from a common ancestral gene). Given the name "competing endogenous RNAs" (ceRNAs), these microRNAs bind to "microRNA response elements" on genes and pseudogenes and may provide another explanation for the persistence of non-coding DNA.
miRNAs are also found as extracellular circulating miRNAs. Circulating miRNAs are released into body fluids including blood and cerebrospinal fluid and have the potential to be available as biomarkers in a number of diseases. Some researches show that mRNA cargo of exosomes may have a role in implantation, they can savage an adhesion between trophoblast and endometrium or support the adhesion by down regulating or up regulating expression of genes involved in adhesion/invasion.
Moreover, miRNA as miR-183/96/182 seems to play a key role in circadian rhythm.
Evolution
miRNAs are well conserved in both plants and animals, and are thought to be a vital and evolutionarily ancient component of gene regulation. While core components of the microRNA pathway are conserved between plants and animals, miRNA repertoires in the two kingdoms appear to have emerged independently with different primary modes of action.
microRNAs are useful phylogenetic markers because of their apparently low rate of evolution. microRNAs' origin as a regulatory mechanism developed from previous RNAi machinery that was initially used as a defense against exogenous genetic material such as viruses. Their origin may have permitted the development of morphological innovation, and by making gene expression more specific and 'fine-tunable', permitted the genesis of complex organs and perhaps, ultimately, complex life. Rapid bursts of morphological innovation are generally associated with a high rate of microRNA accumulation.
New microRNAs are created in multiple ways. Novel microRNAs can originate from the random formation of hairpins in "non-coding" sections of DNA (i.e. introns or intergene regions), but also by the duplication and modification of existing microRNAs. microRNAs can also form from inverted duplications of protein-coding sequences, which allows for the creation of a foldback hairpin structure. The rate of evolution (i.e. nucleotide substitution) in recently originated microRNAs is comparable to that elsewhere in the non-coding DNA, implying evolution by neutral drift; however, older microRNAs have a much lower rate of change (often less than one substitution per hundred million years), suggesting that once a microRNA gains a function, it undergoes purifying selection. Individual regions within an miRNA gene face different evolutionary pressures, where regions that are vital for processing and function have higher levels of conservation. At this point, a microRNA is rarely lost from an animal's genome, although newer microRNAs (thus presumably non-functional) are frequently lost. In Arabidopsis thaliana, the net flux of miRNA genes has been predicted to be between 1.2 and 3.3 genes per million years. This makes them a valuable phylogenetic marker, and they are being looked upon as a possible solution to outstanding phylogenetic problems such as the relationships of arthropods. On the other hand, in multiple cases microRNAs correlate poorly with phylogeny, and it is possible that their phylogenetic concordance largely reflects a limited sampling of microRNAs.
microRNAs feature in the genomes of most eukaryotic organisms, from the brown algae to the animals. However, the difference in how these microRNAs function and the way they are processed suggests that microRNAs arose independently in plants and animals.
Focusing on the animals, the genome of Mnemiopsis leidyi appears to lack recognizable microRNAs, as well as the nuclear proteins Drosha and Pasha, which are critical to canonical microRNA biogenesis. It is the only animal thus far reported to be missing Drosha. MicroRNAs play a vital role in the regulation of gene expression in all non-ctenophore animals investigated thus far except for Trichoplax adhaerens, the first known member of the phylum Placozoa.
Across all species, in excess of 5000 different miRNAs had been identified by March 2010. Whilst short RNA sequences (50 – hundreds of base pairs) of a broadly comparable function occur in bacteria, bacteria lack true microRNAs.
Experimental detection and manipulation
While researchers focused on miRNA expression in physiological and pathological processes, various technical variables related to microRNA isolation emerged. The stability of stored miRNA samples has been questioned. microRNAs degrade much more easily than mRNAs, partly due to their length, but also because of ubiquitously present RNases. This makes it necessary to cool samples on ice and use RNase-free equipment.
microRNA expression can be quantified in a two-step polymerase chain reaction process of modified RT-PCR followed by quantitative PCR. Variations of this method achieve absolute or relative quantification. miRNAs can also be hybridized to microarrays, slides or chips with probes to hundreds or thousands of miRNA targets, so that relative levels of miRNAs can be determined in different samples. microRNAs can be both discovered and profiled by high-throughput sequencing methods (microRNA sequencing). The activity of an miRNA can be experimentally inhibited using a locked nucleic acid (LNA) oligo, a Morpholino oligo or a 2'-O-methyl RNA oligo. A specific miRNA can be silenced by a complementary antagomir. microRNA maturation can be inhibited at several points by steric-blocking oligos. The miRNA target site of an mRNA transcript can also be blocked by a steric-blocking oligo. For the "in situ" detection of miRNA, LNA or Morpholino probes can be used. The locked conformation of LNA results in enhanced hybridization properties and increases sensitivity and selectivity, making it ideal for detection of short miRNA.
High-throughput quantification of miRNAs is error prone, for the larger variance (compared to mRNAs) that comes with methodological problems. mRNA-expression is therefore often analyzed to check for miRNA-effects in their levels (e.g. in). Databases can be used to pair mRNA- and miRNA-data that predict miRNA-targets based on their base sequence. While this is usually done after miRNAs of interest have been detected (e. g. because of high expression levels), ideas for analysis tools that integrate mRNA- and miRNA-expression information have been proposed.
Human and animal diseases
Just as miRNA is involved in the normal functioning of eukaryotic cells, so has dysregulation of miRNA been associated with disease. A manually curated, publicly available database, miR2Disease, documents known relationships between miRNA dysregulation and human disease.
Inherited diseases
A mutation in the seed region of miR-96 causes hereditary progressive hearing loss.
A mutation in the seed region of miR-184 causes hereditary keratoconus with anterior polar cataract.
Deletion of the miR-17~92 cluster causes skeletal and growth defects.
Cancer
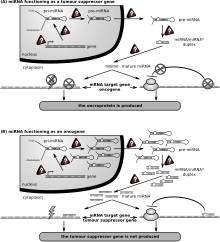
The first human disease known to be associated with miRNA deregulation was chronic lymphocytic leukemia. Many other miRNAs also have links with cancer and accordingly are sometimes referred to as "oncomirs". In malignant B cells miRNAs participate in pathways fundamental to B cell development like B-cell receptor (BCR) signalling, B-cell migration/adhesion, cell-cell interactions in immune niches and the production and class-switching of immunoglobulins. MiRNAs influence B cell maturation, generation of pre-, marginal zone, follicular, B1, plasma and memory B cells.
Another role for miRNA in cancers is to use their expression level for prognosis. In NSCLC samples, low miR-324a levels may serve as an indicator of poor survival. Either high miR-185 or low miR-133b levels may correlate with metastasis and poor survival in colorectal cancer.
Furthermore, specific miRNAs may be associated with certain histological subtypes of colorectal cancer. For instance, expression levels of miR-205 and miR-373 have been shown to be increased in mucinous colorectal cancers and mucin-producing Ulcerative Colitis-associated colon cancers, but not in sporadic colonic adenocarcinoma that lack mucinous components. In-vitro studies suggested that miR-205 and miR-373 may functionally induce different features of mucinous-associated neoplastic progression in intestinal epithelial cells.
Hepatocellular carcinoma cell proliferation may arise from miR-21 interaction with MAP2K3, a tumor repressor gene. Optimal treatment for cancer involves accurately identifying patients for risk-stratified therapy. Those with a rapid response to initial treatment may benefit from truncated treatment regimens, showing the value of accurate disease response measures. Cell-free circulating miRNAs (cimiRNAs) are highly stable in blood, are overexpressed in cancer and are quantifiable within the diagnostic laboratory. In classical Hodgkin lymphoma, plasma miR-21, miR-494, and miR-1973 are promising disease response biomarkers. Circulating miRNAs have the potential to assist clinical decision making and aid interpretation of positron emission tomography combined with computerized tomography. They can be performed at each consultation to assess disease response and detect relapse.
MicroRNAs have the potential to be used as tools or targets for treatment of different cancers. The specific microRNA, miR-506 has been found to work as a tumor antagonist in several studies. A significant number of cervical cancer samples were found to have downregulated expression of miR-506. Additionally, miR-506 works to promote apoptosis of cervical cancer cells, through its direct target hedgehog pathway transcription factor, Gli3.
DNA repair and cancer
Many miRNAs can directly target and inhibit cell cycle genes to control cell proliferation. A new strategy for tumor treatment is to inhibit tumor cell proliferation by repairing the defective miRNA pathway in tumors. Cancer is caused by the accumulation of mutations from either DNA damage or uncorrected errors in DNA replication. Defects in DNA repair cause the accumulation of mutations, which can lead to cancer. Several genes involved in DNA repair are regulated by microRNAs.
Germline mutations in DNA repair genes cause only 2–5% of colon cancer cases. However, altered expression of microRNAs, causing DNA repair deficiencies, are frequently associated with cancers and may be an important causal factor. Among 68 sporadic colon cancers with reduced expression of the DNA mismatch repair protein MLH1, most were found to be deficient due to epigenetic methylation of the CpG island of the MLH1 gene. However, up to 15% of MLH1-deficiencies in sporadic colon cancers appeared to be due to over-expression of the microRNA miR-155, which represses MLH1 expression.
In 29–66% of glioblastomas, DNA repair is deficient due to epigenetic methylation of the MGMT gene, which reduces protein expression of MGMT. However, for 28% of glioblastomas, the MGMT protein is deficient, but the MGMT promoter is not methylated. In glioblastomas without methylated MGMT promoters, the level of microRNA miR-181d is inversely correlated with protein expression of MGMT and the direct target of miR-181d is the MGMT mRNA 3'UTR (the three prime untranslated region of MGMT mRNA). Thus, in 28% of glioblastomas, increased expression of miR-181d and reduced expression of DNA repair enzyme MGMT may be a causal factor.
HMGA proteins (HMGA1a, HMGA1b and HMGA2) are implicated in cancer, and expression of these proteins is regulated by microRNAs. HMGA expression is almost undetectable in differentiated adult tissues, but is elevated in many cancers. HMGA proteins are polypeptides of ~100 amino acid residues characterized by a modular sequence organization. These proteins have three highly positively charged regions, termed AT hooks, that bind the minor groove of AT-rich DNA stretches in specific regions of DNA. Human neoplasias, including thyroid, prostatic, cervical, colorectal, pancreatic and ovarian carcinomas, show a strong increase of HMGA1a and HMGA1b proteins. Transgenic mice with HMGA1 targeted to lymphoid cells develop aggressive lymphoma, showing that high HMGA1 expression is associated with cancers and that HMGA1 can act as an oncogene. HMGA2 protein specifically targets the promoter of ERCC1, thus reducing expression of this DNA repair gene. ERCC1 protein expression was deficient in 100% of 47 evaluated colon cancers (though the extent to which HGMA2 was involved is not known).
Single Nucleotide polymorphisms (SNPs) can alter the binding of miRNAs on 3'UTRs for example the case of hsa-mir181a and hsa-mir181b on the CDON tumor suppressor gene.
Heart disease
The global role of miRNA function in the heart has been addressed by conditionally inhibiting miRNA maturation in the murine heart. This revealed that miRNAs play an essential role during its development. miRNA expression profiling studies demonstrate that expression levels of specific miRNAs change in diseased human hearts, pointing to their involvement in cardiomyopathies. Furthermore, animal studies on specific miRNAs identified distinct roles for miRNAs both during heart development and under pathological conditions, including the regulation of key factors important for cardiogenesis, the hypertrophic growth response and cardiac conductance. Another role for miRNA in cardiovascular diseases is to use their expression levels for diagnosis, prognosis or risk stratification. miRNA's in animal models have also been linked to cholesterol metabolism and regulation.
miRNA-712
Murine microRNA-712 is a potential biomarker (i.e. predictor) for atherosclerosis, a cardiovascular disease of the arterial wall associated with lipid retention and inflammation. Non-laminar blood flow also correlates with development of atherosclerosis as mechanosenors of endothelial cells respond to the shear force of disturbed flow (d-flow). A number of pro-atherogenic genes including matrix metalloproteinases (MMPs) are upregulated by d-flow, mediating pro-inflammatory and pro-angiogenic signals. These findings were observed in ligated carotid arteries of mice to mimic the effects of d-flow. Within 24 hours, pre-existing immature miR-712 formed mature miR-712 suggesting that miR-712 is flow-sensitive. Coinciding with these results, miR-712 is also upregulated in endothelial cells exposed to naturally occurring d-flow in the greater curvature of the aortic arch.
Origin
Pre-mRNA sequence of miR-712 is generated from the murine ribosomal RN45s gene at the internal transcribed spacer region 2 (ITS2). XRN1 is an exonuclease that degrades the ITS2 region during processing of RN45s. Reduction of XRN1 under d-flow conditions therefore leads to the accumulation of miR-712.
Mechanism
MiR-712 targets tissue inhibitor of metalloproteinases 3 (TIMP3). TIMPs normally regulate activity of matrix metalloproteinases (MMPs) which degrade the extracellular matrix (ECM). Arterial ECM is mainly composed of collagen and elastin fibers, providing the structural support and recoil properties of arteries. These fibers play a critical role in regulation of vascular inflammation and permeability, which are important in the development of atherosclerosis. Expressed by endothelial cells, TIMP3 is the only ECM-bound TIMP. A decrease in TIMP3 expression results in an increase of ECM degradation in the presence of d-flow. Consistent with these findings, inhibition of pre-miR712 increases expression of TIMP3 in cells, even when exposed to turbulent flow.
TIMP3 also decreases the expression of TNFα (a pro-inflammatory regulator) during turbulent flow. Activity of TNFα in turbulent flow was measured by the expression of TNFα-converting enzyme (TACE) in blood. TNFα decreased if miR-712 was inhibited or TIMP3 overexpressed, suggesting that miR-712 and TIMP3 regulate TACE activity in turbulent flow conditions.
Anti-miR-712 effectively suppresses d-flow-induced miR-712 expression and increases TIMP3 expression. Anti-miR-712 also inhibits vascular hyperpermeability, thereby significantly reducing atherosclerosis lesion development and immune cell infiltration.
Human homolog microRNA-205
The human homolog of miR-712 was found on the RN45s homolog gene, which maintains similar miRNAs to mice. MiR-205 of humans share similar sequences with miR-712 of mice and is conserved across most vertebrates. MiR-205 and miR-712 also share more than 50% of the cell signaling targets, including TIMP3.
When tested, d-flow decreased the expression of XRN1 in humans as it did in mice endothelial cells, indicating a potentially common role of XRN1 in humans.
Kidney disease
Targeted deletion of Dicer in the FoxD1-derived renal progenitor cells in a murine model resulted in a complex renal phenotype including expansion of nephron progenitors, fewer renin cells, smooth muscle arterioles, progressive mesangial loss and glomerular aneurysms. High throughput whole transcriptome profiling of the FoxD1-Dicer knockout mouse model revealed ectopic upregulation of pro-apoptotic gene, Bcl2L11 (Bim) and dysregulation of the p53 pathway with increase in p53 effector genes including Bax, Trp53inp1, Jun, Cdkn1a, Mmp2, and Arid3a. p53 protein levels remained unchanged, suggesting that FoxD1 stromal miRNAs directly repress p53-effector genes. Using a lineage tracing approach followed by Fluorescent-activated cell sorting, miRNA profiling of the FoxD1-derived cells not only comprehensively defined the transcriptional landscape of miRNAs that are critical for vascular development, but also identified key miRNAs that are likely to modulate the renal phenotype in its absence. These miRNAs include miRs-10a, 18a, 19b, 24, 30c, 92a, 106a, 130a, 152, 181a, 214, 222, 302a, 370, and 381 that regulate Bcl2L11 (Bim) and miRs-15b, 18a, 21, 30c, 92a, 106a, 125b-5p, 145, 214, 222, 296-5p and 302a that regulate p53-effector genes. Consistent with the profiling results, ectopic apoptosis was observed in the cellular derivatives of the FoxD1 derived progenitor lineage and reiterates the importance of renal stromal miRNAs in cellular homeostasis.
Nervous system
MiRNAs are crucial for the healthy development and function of the nervous system. Previous studies demonstrate that miRNAs can regulate neuronal differentiation and maturation at various stages. MiRNAs also play important roles in synaptic development (such as dendritogenesis or spine morphogenesis) and synaptic plasticity (contributing to learning and memory). Elimination of miRNA formation in mice by experimental silencing of Dicer has led to pathological outcomes, such as reduced neuronal size, motor abnormalities (when silenced in striatal neurons), and neurodegeneration (when silenced in forebrain neurons). Altered miRNA expression has been found in neurodegenerative diseases (such as Alzheimer's disease, Parkinson's disease, and Huntington's disease) as well as many psychiatric disorders (including epilepsy, schizophrenia, major depression, bipolar disorder, and anxiety disorders).
Stroke
According to the Center for Disease Control and Prevention, Stroke is one of the leading causes of death and long-term disability in America. 87% of the cases are ischemic strokes, which results from blockage in the artery of the brain that carries oxygen-rich blood. The obstruction of the blood flow means the brain cannot receive necessary nutrients, such as oxygen and glucose, and remove wastes, such as carbon dioxide. miRNAs plays a role in posttranslational gene silencing by targeting genes in the pathogenesis of cerebral ischemia, such as the inflammatory, angiogenesis, and apoptotic pathway.
Alcoholism
The vital role of miRNAs in gene expression is significant to addiction, specifically alcoholism. Chronic alcohol abuse results in persistent changes in brain function mediated in part by alterations in gene expression. miRNA global regulation of many downstream genes deems significant regarding the reorganization or synaptic connections or long term neural adaptations involving the behavioral change from alcohol consumption to withdrawal and/or dependence. Up to 35 different miRNAs have been found to be altered in the alcoholic post-mortem brain, all of which target genes that include the regulation of the cell cycle, apoptosis, cell adhesion, nervous system development and cell signaling. Altered miRNA levels were found in the medial prefrontal cortex of alcohol-dependent mice, suggesting the role of miRNA in orchestrating translational imbalances and the creation of differentially expressed proteins within an area of the brain where complex cognitive behavior and decision making likely originate.
miRNAs can be either upregulated or downregulated in response to chronic alcohol use. miR-206 expression increased in the prefrontal cortex of alcohol-dependent rats, targeting the transcription factor brain-derived neurotrophic factor (BDNF) and ultimately reducing its expression. BDNF plays a critical role in the formation and maturation of new neurons and synapses, suggesting a possible implication in synapse growth/synaptic plasticity in alcohol abusers. miR-155, important in regulating alcohol-induced neuroinflammation responses, was found to be upregulated, suggesting the role of microglia and inflammatory cytokines in alcohol pathophysiology. Downregulation of miR-382 was found in the nucleus accumbens, a structure in the basal forebrain significant in regulating feelings of reward that power motivational habits. miR-382 is the target for the dopamine receptor D1 (DRD1), and its overexpression results in the upregulation of DRD1 and delta fosB, a transcription factor that activates a series of transcription events in the nucleus accumbens that ultimately result in addictive behaviors. Alternatively, overexpressing miR-382 resulted in attenuated drinking and the inhibition of DRD1 and delta fosB upregulation in rat models of alcoholism, demonstrating the possibility of using miRNA-targeted pharmaceuticals in treatments.
Obesity
miRNAs play crucial roles in the regulation of stem cell progenitors differentiating into adipocytes. Studies to determine what role pluripotent stem cells play in adipogenesis, were examined in the immortalized human bone marrow-derived stromal cell line hMSC-Tert20. Decreased expression of miR-155, miR-221, and miR-222, have been found during the adipogenic programming of both immortalized and primary hMSCs, suggesting that they act as negative regulators of differentiation. Conversely, ectopic expression of the miRNAs 155, 221, and 222 significantly inhibited adipogenesis and repressed induction of the master regulators PPARγ and CCAAT/enhancer-binding protein alpha (CEBPA). This paves the way for possible genetic obesity treatments.
Another class of miRNAs that regulate insulin resistance, obesity, and diabetes, is the let-7 family. Let-7 accumulates in human tissues during the course of aging. When let-7 was ectopically overexpressed to mimic accelerated aging, mice became insulin-resistant, and thus more prone to high fat diet-induced obesity and diabetes. In contrast when let-7 was inhibited by injections of let-7-specific antagomirs, mice become more insulin-sensitive and remarkably resistant to high fat diet-induced obesity and diabetes. Not only could let-7 inhibition prevent obesity and diabetes, it could also reverse and cure the condition. These experimental findings suggest that let-7 inhibition could represent a new therapy for obesity and type 2 diabetes.
Hemostasis
miRNAs also play crucial roles in the regulation of complex enzymatic cascades including the hemostatic blood coagulation system. Large scale studies of functional miRNA targeting have recently uncovered rationale therapeutic targets in the hemostatic system. They have been directly linked to Calcium homeostasis in the endoplasmic reticulum, which is critical in cell differentiation in early development.
Plants
miRNAs are considered to be key regulators of many developmental, homeostatic, and immune processes in plants. Their roles in plant development include shoot apical meristem development, leaf growth, flower formation, seed production, or root expansion. In addition, they play a complex role in responses to various abiotic stresses comprising heat stress, low-temperature stress, drought stress, light stress, or gamma radiation exposure.
Viruses
Viral microRNAs play an important role in the regulation of gene expression of viral and/or host genes to benefit the virus. Hence, miRNAs play a key role in host–virus interactions and pathogenesis of viral diseases. The expression of transcription activators by human herpesvirus-6 DNA is believed to be regulated by viral miRNA.
Target prediction
See also: List of RNA structure prediction software § Inter molecular interactions: MicroRNA:UTRmiRNAs can bind to target messenger RNA (mRNA) transcripts of protein-coding genes and negatively control their translation or cause mRNA degradation. It is of key importance to identify the miRNA targets accurately. A comparison of the predictive performance of eighteen in silico algorithms is available. Large scale studies of functional miRNA targeting suggest that many functional miRNAs can be missed by target prediction algorithms.
See also
- Anti-miRNA oligonucleotides
- C19MC miRNA cluster
- Gene expression
- List of miRNA gene prediction tools
- List of miRNA target prediction tools
- MicroDNA
- MicroRNA Biosensors
- MiRNEST
- MIR222
- miR-324-5p
- Mir-M7 microRNA precursor family
- RNA interference
- Small interfering RNA
- Small nucleolar RNA-derived microRNA
References
- ^ Bartel DP (March 2018). "Metazoan MicroRNAs". Cell. 173 (1): 20–51. doi:10.1016/j.cell.2018.03.006. PMC 6091663. PMID 29570994.
- ^ Bartel DP (January 2004). "MicroRNAs: genomics, biogenesis, mechanism, and function". Cell. 116 (2): 281–297. doi:10.1016/S0092-8674(04)00045-5. PMID 14744438.
- Qureshi A, Thakur N, Monga I, Thakur A, Kumar M (January 2014). "VIRmiRNA: A comprehensive resource for experimentally validated viral miRNAs and their targets". Database. 2014: bau103. doi:10.1093/database/bau103. PMC 4224276. PMID 25380780.
- ^ Bartel DP (January 2009). "MicroRNAs: Target recognition and regulatory functions". Cell. 136 (2): 215–233. doi:10.1016/j.cell.2009.01.002. PMC 3794896. PMID 19167326.
- Jonas S, Izaurralde E (July 2015). "Towards a molecular understanding of microRNA-mediated gene silencing". Nature Reviews. Genetics. 16 (7): 421–433. doi:10.1038/nrg3965. PMID 26077373. S2CID 24892348.
- Jonas S, Izaurralde E (July 2015). "Towards a molecular understanding of microRNA-mediated gene silencing". Nature Reviews. Genetics. 16 (7): 421–433. doi:10.1038/nrg3965. PMID 26077373. S2CID 24892348.
- Guo H, Ingolia NT, Weissman JS, Bartel DP (August 2010). "Mammalian microRNAs predominantly act to decrease target mRNA levels". Nature. 466 (7308): 835–840. Bibcode:2010Natur.466..835G. doi:10.1038/nature09267. hdl:1721.1/72447. PMC 2990499. PMID 20703300.
- "Homo sapiens miRNAs". miRBase. Manchester, UK: Manchester University.
- Alles J, Fehlmann T, Fischer U, Backes C, Galata V, Minet M, et al. (April 2019). "An estimate of the total number of true human miRNAs". Nucleic Acids Research. 47 (7): 3353–3364. doi:10.1093/nar/gkz097. PMC 6468295. PMID 30820533.
{{cite journal}}: CS1 maint: overridden setting (link) - Fromm B, Domanska D, Høye E, Ovchinnikov V, Kang W, Aparicio-Puerta E, et al. (January 2020). "MirGeneDB 2.0: The metazoan microRNA complement". Nucleic Acids Research. 48 (D1): D132–D141. doi:10.1093/nar/gkz885. PMC 6943042. PMID 31598695.
{{cite journal}}: CS1 maint: overridden setting (link) - Lim LP, Lau NC, Weinstein EG, Abdelhakim A, Yekta S, Rhoades MW, et al. (April 2003). "The microRNAs of Caenorhabditis elegans". Genes & Development. 17 (8): 991–1008. doi:10.1101/gad.1074403. PMC 196042. PMID 12672692.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ Lagos-Quintana M, Rauhut R, Yalcin A, Meyer J, Lendeckel W, Tuschl T (April 2002). "Identification of tissue-specific microRNAs from mouse". Current Biology. 12 (9): 735–739. Bibcode:2002CBio...12..735L. doi:10.1016/S0960-9822(02)00809-6. PMID 12007417.
- ^ Lewis BP, Burge CB, Bartel DP (January 2005). "Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets". Cell. 120 (1): 15–20. doi:10.1016/j.cell.2004.12.035. PMID 15652477.
- ^ Friedman RC, Farh KK, Burge CB, Bartel DP (January 2009). "Most mammalian mRNAs are conserved targets of microRNAs". Genome Research. 19 (1): 92–105. doi:10.1101/gr.082701.108. PMC 2612969. PMID 18955434.
- Fromm B, Billipp T, Peck LE, Johansen M, Tarver JE, King BL, et al. (2015). "A uniform system for the annotation of vertebrate microRNA genes and the evolution of the human microRNAome". Annual Review of Genetics. 49 (1): 213–242. Bibcode:2015ARGen..49..213F. doi:10.1146/annurev-genet-120213-092023. PMC 4743252. PMID 26473382.
{{cite journal}}: CS1 maint: overridden setting (link) - "The Nobel Prize in Physiology or Medicine 2024".
- "The Nobel Prize in Physiology or Medicine 2024". NobelPrize.org (Press release). Retrieved 7 October 2024.
- Lewis T (7 October 2024). "Nobel Prize in Physiology or Medicine awarded for discovery of microRNA gene regulation". Scientific American. Retrieved 7 October 2024.
- ^ Lee RC, Feinbaum RL, Ambros V (December 1993). "The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14". Cell. 75 (5): 843–54. doi:10.1016/0092-8674(93)90529-Y. PMID 8252621.
- ^ Reinhart BJ, Slack FJ, Basson M, Pasquinelli AE, Bettinger JC, Rougvie AE, et al. (February 2000). "The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans". Nature. 403 (6772): 901–6. Bibcode:2000Natur.403..901R. doi:10.1038/35002607. PMID 10706289. S2CID 4384503.
- ^ Pasquinelli AE, Reinhart BJ, Slack F, Martindale MQ, Kuroda MI, Maller B, et al. (November 2000). "Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA". Nature. 408 (6808): 86–9. Bibcode:2000Natur.408...86P. doi:10.1038/35040556. PMID 11081512. S2CID 4401732.
- ^ Lagos-Quintana M, Rauhut R, Lendeckel W, Tuschl T (October 2001). "Identification of novel genes coding for small expressed RNAs". Science. 294 (5543): 853–8. Bibcode:2001Sci...294..853L. doi:10.1126/science.1064921. hdl:11858/00-001M-0000-0012-F65F-2. PMID 11679670. S2CID 18101169.
- ^ Lau NC, Lim LP, Weinstein EG, Bartel DP (October 2001). "An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans". Science. 294 (5543): 858–62. Bibcode:2001Sci...294..858L. doi:10.1126/science.1065062. PMID 11679671. S2CID 43262684.
- ^ Lee RC, Ambros V (October 2001). "An extensive class of small RNAs in Caenorhabditis elegans". Science. 294 (5543): 862–4. Bibcode:2001Sci...294..862L. doi:10.1126/science.1065329. PMID 11679672. S2CID 33480585.
- Wienholds E, Kloosterman WP, Miska E, Alvarez-Saavedra E, Berezikov E, de Bruijn E, et al. (July 2005). "MicroRNA expression in zebrafish embryonic development". Science. 309 (5732): 310–1. Bibcode:2005Sci...309..310W. doi:10.1126/science.1114519. PMID 15919954. S2CID 38939571.
- ^ Jones-Rhoades MW, Bartel DP, Bartel B (2006). "MicroRNAS and their regulatory roles in plants". Annual Review of Plant Biology. 57: 19–53. doi:10.1146/annurev.arplant.57.032905.105218. PMID 16669754.
- Brennecke J, Hipfner DR, Stark A, Russell RB, Cohen SM (April 2003). "bantam encodes a developmentally regulated microRNA that controls cell proliferation and regulates the proapoptotic gene hid in Drosophila". Cell. 113 (1): 25–36. doi:10.1016/S0092-8674(03)00231-9. PMID 12679032.
- Cuellar TL, McManus MT (December 2005). "MicroRNAs and endocrine biology". The Journal of Endocrinology. 187 (3): 327–32. doi:10.1677/joe.1.06426. PMID 16423811.
- Poy MN, Eliasson L, Krutzfeldt J, Kuwajima S, Ma X, Macdonald PE, et al. (November 2004). "A pancreatic islet-specific microRNA regulates insulin secretion". Nature. 432 (7014): 226–30. Bibcode:2004Natur.432..226P. doi:10.1038/nature03076. PMID 15538371. S2CID 4415988.
- Chen CZ, Li L, Lodish HF, Bartel DP (January 2004). "MicroRNAs modulate hematopoietic lineage differentiation". Science. 303 (5654): 83–6. Bibcode:2004Sci...303...83C. doi:10.1126/science.1091903. hdl:1721.1/7483. PMID 14657504. S2CID 7044929.
- Wilfred BR, Wang WX, Nelson PT (July 2007). "Energizing miRNA research: a review of the role of miRNAs in lipid metabolism, with a prediction that miR-103/107 regulates human metabolic pathways". Molecular Genetics and Metabolism. 91 (3): 209–17. doi:10.1016/j.ymgme.2007.03.011. PMC 1978064. PMID 17521938.
- Harfe BD, McManus MT, Mansfield JH, Hornstein E, Tabin CJ (August 2005). "The RNaseIII enzyme Dicer is required for morphogenesis but not patterning of the vertebrate limb". Proceedings of the National Academy of Sciences of the United States of America. 102 (31): 10898–903. Bibcode:2005PNAS..10210898H. doi:10.1073/pnas.0504834102. PMC 1182454. PMID 16040801.
- Trang P, Weidhaas JB, Slack FJ (December 2008). "MicroRNAs as potential cancer therapeutics". Oncogene. 27 (Suppl 2): S52–7. doi:10.1038/onc.2009.353. PMC 10033140. PMID 19956180.
- Li C, Feng Y, Coukos G, Zhang L (December 2009). "Therapeutic microRNA strategies in human cancer". The AAPS Journal. 11 (4): 747–57. doi:10.1208/s12248-009-9145-9. PMC 2782079. PMID 19876744.
- Fasanaro P, Greco S, Ivan M, Capogrossi MC, Martelli F (January 2010). "microRNA: emerging therapeutic targets in acute ischemic diseases". Pharmacology & Therapeutics. 125 (1): 92–104. doi:10.1016/j.pharmthera.2009.10.003. PMID 19896977.
- Hydbring P, Badalian-Very G (August 2013). "Clinical applications of microRNAs". F1000Research. 2: 136. doi:10.12688/f1000research.2-136.v2. PMC 3917658. PMID 24627783.
- Wightman B, Ha I, Ruvkun G (December 1993). "Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans". Cell. 75 (5): 855–62. doi:10.1016/0092-8674(93)90530-4. PMID 8252622.
- Giza DE, Calin GA (2015). "MicroRNA and Chronic Lymphocytic Leukemia". MicroRNA: Cancer. Advances in Experimental Medicine and Biology. Vol. 889. pp. 23–40. doi:10.1007/978-3-319-23730-5_2. ISBN 978-3-319-23729-9. PMID 26658994.
- Ambros V, Bartel B, Bartel DP, Burge CB, Carrington JC, Chen X, et al. (March 2003). "A uniform system for microRNA annotation". RNA. 9 (3): 277–9. doi:10.1261/rna.2183803. PMC 1370393. PMID 12592000.
- Griffiths-Jones S, Grocock RJ, van Dongen S, Bateman A, Enright AJ (January 2006). "miRBase: microRNA sequences, targets and gene nomenclature". Nucleic Acids Research. 34 (Database issue): D140–4. doi:10.1093/nar/gkj112. PMC 1347474. PMID 16381832.
- Wright MW, Bruford EA (January 2011). "Naming 'junk': human non-protein coding RNA (ncRNA) gene nomenclature". Human Genomics. 5 (2): 90–8. doi:10.1186/1479-7364-5-2-90. PMC 3051107. PMID 21296742.
- Hunt M, Banerjee S, Surana P, Liu M, Fuerst G, Mathioni S, et al. (2019). "Small RNA discovery in the interaction between barley and the powdery mildew pathogen". BMC Genomics. 20 (1): 19–53. doi:10.1186/s12864-019-5947-z. PMC 6657096. PMID 31345162.
- ^ Lewis BP, Shih IH, Jones-Rhoades MW, Bartel DP, Burge CB (December 2003). "Prediction of mammalian microRNA targets". Cell. 115 (7): 787–98. doi:10.1016/S0092-8674(03)01018-3. PMID 14697198.
- Ellwanger DC, Büttner FA, Mewes HW, Stümpflen V (May 2011). "The sufficient minimal set of miRNA seed types". Bioinformatics. 27 (10): 1346–50. doi:10.1093/bioinformatics/btr149. PMC 3087955. PMID 21441577.
- Rajewsky N (June 2006). "microRNA target predictions in animals". Nature Genetics. 38 (6s): S8–13. doi:10.1038/ng1798. PMID 16736023. S2CID 23496396.
- Krek A, Grün D, Poy MN, Wolf R, Rosenberg L, Epstein EJ, et al. (May 2005). "Combinatorial microRNA target predictions". Nature Genetics. 37 (5): 495–500. doi:10.1038/ng1536. PMID 15806104. S2CID 22672750.
- Thomson DW, Bracken CP, Goodall GJ (September 2011). "Experimental strategies for microRNA target identification". Nucleic Acids Research. 39 (16): 6845–53. doi:10.1093/nar/gkr330. PMC 3167600. PMID 21652644.
- ^ Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, et al. (February 2005). "Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs". Nature. 433 (7027): 769–73. Bibcode:2005Natur.433..769L. doi:10.1038/nature03315. PMID 15685193. S2CID 4430576.
- Selbach M, Schwanhäusser B, Thierfelder N, Fang Z, Khanin R, Rajewsky N (September 2008). "Widespread changes in protein synthesis induced by microRNAs". Nature. 455 (7209): 58–63. Bibcode:2008Natur.455...58S. doi:10.1038/nature07228. PMID 18668040. S2CID 4429008.
- Baek D, Villén J, Shin C, Camargo FD, Gygi SP, Bartel DP (September 2008). "The impact of microRNAs on protein output". Nature. 455 (7209): 64–71. Bibcode:2008Natur.455...64B. doi:10.1038/nature07242. PMC 2745094. PMID 18668037.
- ^ Rodriguez A, Griffiths-Jones S, Ashurst JL, Bradley A (October 2004). "Identification of mammalian microRNA host genes and transcription units". Genome Research. 14 (10A): 1902–10. doi:10.1101/gr.2722704. PMC 524413. PMID 15364901.
- ^ Cai X, Hagedorn CH, Cullen BR (December 2004). "Human microRNAs are processed from capped, polyadenylated transcripts that can also function as mRNAs". RNA. 10 (12): 1957–66. doi:10.1261/rna.7135204. PMC 1370684. PMID 15525708.
- Weber MJ (January 2005). "New human and mouse microRNA genes found by homology search". The FEBS Journal. 272 (1): 59–73. doi:10.1111/j.1432-1033.2004.04389.x. PMID 15634332. S2CID 32923462.
- Kim YK, Kim VN (February 2007). "Processing of intronic microRNAs". The EMBO Journal. 26 (3): 775–83. doi:10.1038/sj.emboj.7601512. PMC 1794378. PMID 17255951.
- Baskerville S, Bartel DP (March 2005). "Microarray profiling of microRNAs reveals frequent coexpression with neighboring miRNAs and host genes". RNA. 11 (3): 241–7. doi:10.1261/rna.7240905. PMC 1370713. PMID 15701730.
- ^ Lee Y, Kim M, Han J, Yeom KH, Lee S, Baek SH, et al. (October 2004). "MicroRNA genes are transcribed by RNA polymerase II". The EMBO Journal. 23 (20): 4051–60. doi:10.1038/sj.emboj.7600385. PMC 524334. PMID 15372072.
- Zhou X, Ruan J, Wang G, Zhang W (March 2007). "Characterization and identification of microRNA core promoters in four model species". PLOS Computational Biology. 3 (3): e37. Bibcode:2007PLSCB...3...37Z. doi:10.1371/journal.pcbi.0030037. PMC 1817659. PMID 17352530.
- Faller M, Guo F (November 2008). "MicroRNA biogenesis: there's more than one way to skin a cat". Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms. 1779 (11): 663–7. doi:10.1016/j.bbagrm.2008.08.005. PMC 2633599. PMID 18778799.
- Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J, et al. (September 2003). "The nuclear RNase III Drosha initiates microRNA processing". Nature. 425 (6956): 415–9. Bibcode:2003Natur.425..415L. doi:10.1038/nature01957. PMID 14508493. S2CID 4421030.
- Gregory RI, Chendrimada TP, Shiekhattar R (2006). "MicroRNA biogenesis: isolation and characterization of the microprocessor complex". MicroRNA Protocols. Methods in Molecular Biology. Vol. 342. pp. 33–47. doi:10.1385/1-59745-123-1:33. ISBN 978-1-59745-123-9. PMID 16957365.
- Han J, Lee Y, Yeom KH, Kim YK, Jin H, Kim VN (December 2004). "The Drosha-DGCR8 complex in primary microRNA processing". Genes & Development. 18 (24): 3016–27. doi:10.1101/gad.1262504. PMC 535913. PMID 15574589.
- Han J, Lee Y, Yeom KH, Nam JW, Heo I, Rhee JK, et al. (June 2006). "Molecular basis for the recognition of primary microRNAs by the Drosha-DGCR8 complex". Cell. 125 (5): 887–901. doi:10.1016/j.cell.2006.03.043. PMID 16751099.
- Conrad T, Marsico A, Gehre M, Orom UA (October 2014). "Microprocessor activity controls differential miRNA biogenesis in Vivo". Cell Reports. 9 (2): 542–54. doi:10.1016/j.celrep.2014.09.007. PMID 25310978.
- Auyeung VC, Ulitsky I, McGeary SE, Bartel DP (February 2013). "Beyond secondary structure: primary-sequence determinants license pri-miRNA hairpins for processing". Cell. 152 (4): 844–58. doi:10.1016/j.cell.2013.01.031. PMC 3707628. PMID 23415231.
- Ali PS, Ghoshdastider U, Hoffmann J, Brutschy B, Filipek S (November 2012). "Recognition of the let-7g miRNA precursor by human Lin28B". FEBS Letters. 586 (22): 3986–90. Bibcode:2012FEBSL.586.3986S. doi:10.1016/j.febslet.2012.09.034. PMID 23063642. S2CID 28899778.
- ^ Ruby JG, Jan CH, Bartel DP (July 2007). "Intronic microRNA precursors that bypass Drosha processing". Nature. 448 (7149): 83–86. Bibcode:2007Natur.448...83R. doi:10.1038/nature05983. PMC 2475599. PMID 17589500.
- Berezikov E, Chung WJ, Willis J, Cuppen E, Lai EC (October 2007). "Mammalian mirtron genes". Molecular Cell. 28 (2): 328–336. doi:10.1016/j.molcel.2007.09.028. PMC 2763384. PMID 17964270.
- ^ Kawahara Y, Megraw M, Kreider E, Iizasa H, Valente L, Hatzigeorgiou AG, et al. (September 2008). "Frequency and fate of microRNA editing in human brain". Nucleic Acids Research. 36 (16): 5270–80. doi:10.1093/nar/gkn479. PMC 2532740. PMID 18684997.
- Winter J, Jung S, Keller S, Gregory RI, Diederichs S (March 2009). "Many roads to maturity: microRNA biogenesis pathways and their regulation". Nature Cell Biology. 11 (3): 228–34. doi:10.1038/ncb0309-228. PMID 19255566. S2CID 205286318.
- Ohman M (October 2007). "A-to-I editing challenger or ally to the microRNA process". Biochimie. 89 (10): 1171–6. doi:10.1016/j.biochi.2007.06.002. PMID 17628290.
- ^ Murchison EP, Hannon GJ (June 2004). "miRNAs on the move: miRNA biogenesis and the RNAi machinery". Current Opinion in Cell Biology. 16 (3): 223–9. doi:10.1016/j.ceb.2004.04.003. PMID 15145345.

- ^ Lund E, Dahlberg JE (2006). "Substrate selectivity of exportin 5 and Dicer in the biogenesis of microRNAs". Cold Spring Harbor Symposia on Quantitative Biology. 71: 59–66. doi:10.1101/sqb.2006.71.050. PMID 17381281.
- Park JE, Heo I, Tian Y, Simanshu DK, Chang H, Jee D, et al. (July 2011). "Dicer recognizes the 5' end of RNA for efficient and accurate processing". Nature. 475 (7355): 201–5. doi:10.1038/nature10198. PMC 4693635. PMID 21753850.
- Ji X (2008). "The Mechanism of RNase III Action: How Dicer Dices". RNA Interference. Current Topics in Microbiology and Immunology. Vol. 320. pp. 99–116. doi:10.1007/978-3-540-75157-1_5. ISBN 978-3-540-75156-4. PMID 18268841.
- Mirihana Arachchilage G, Dassanayake AC, Basu S (February 2015). "A potassium ion-dependent RNA structural switch regulates human pre-miRNA 92b maturation". Chemistry & Biology. 22 (2): 262–72. doi:10.1016/j.chembiol.2014.12.013. PMID 25641166.
- Sohel MH (2016). "Extracellular/Circulating MicroRNAs: Release Mechanisms, Functions and Challenges". Achievements in the Life Sciences. 10 (2): 175–186. doi:10.1016/j.als.2016.11.007.
- ^ Boeckel JN, Reis SM, Leistner D, Thomé CE, Zeiher AM, Fichtlscherer S, et al. (April 2014). "From heart to toe: heart's contribution on peripheral microRNA levels". International Journal of Cardiology. 172 (3): 616–7. doi:10.1016/j.ijcard.2014.01.082. PMID 24508494.
- Lelandais-Brière C, Sorin C, Declerck M, Benslimane A, Crespi M, Hartmann C (March 2010). "Small RNA diversity in plants and its impact in development". Current Genomics. 11 (1): 14–23. doi:10.2174/138920210790217918. PMC 2851111. PMID 20808519.
- Rana TM (January 2007). "Illuminating the silence: understanding the structure and function of small RNAs". Nature Reviews Molecular Cell Biology. 8 (1): 23–36. doi:10.1038/nrm2085. PMID 17183358. S2CID 8966239.
- ^ Schwarz DS, Zamore PD (May 2002). "Why do miRNAs live in the miRNP?". Genes & Development. 16 (9): 1025–31. doi:10.1101/gad.992502. PMID 12000786.
- Krol J, Sobczak K, Wilczynska U, Drath M, Jasinska A, Kaczynska D, et al. (October 2004). "Structural features of microRNA (miRNA) precursors and their relevance to miRNA biogenesis and small interfering RNA/short hairpin RNA design". The Journal of Biological Chemistry. 279 (40): 42230–9. doi:10.1074/jbc.M404931200. PMID 15292246.
- Khvorova A, Reynolds A, Jayasena SD (October 2003). "Functional siRNAs and miRNAs exhibit strand bias". Cell. 115 (2): 209–16. doi:10.1016/S0092-8674(03)00801-8. PMID 14567918.
- Schwarz DS, Hutvágner G, Du T, Xu Z, Aronin N, Zamore PD (October 2003). "Asymmetry in the assembly of the RNAi enzyme complex". Cell. 115 (2): 199–208. doi:10.1016/S0092-8674(03)00759-1. PMID 14567917.
- Lin SL, Chang D, Ying SY (August 2005). "Asymmetry of intronic pre-miRNA structures in functional RISC assembly". Gene. 356: 32–8. doi:10.1016/j.gene.2005.04.036. PMC 1788082. PMID 16005165.
- Okamura K, Chung WJ, Lai EC (September 2008). "The long and short of inverted repeat genes in animals: microRNAs, mirtrons and hairpin RNAs". Cell Cycle. 7 (18): 2840–5. doi:10.4161/cc.7.18.6734. PMC 2697033. PMID 18769156.
- ^ Pratt AJ, MacRae IJ (July 2009). "The RNA-induced silencing complex: a versatile gene-silencing machine". The Journal of Biological Chemistry. 284 (27): 17897–901. doi:10.1074/jbc.R900012200. PMC 2709356. PMID 19342379.
- MacRae IJ, Ma E, Zhou M, Robinson CV, Doudna JA (January 2008). "In vitro reconstitution of the human RISC-loading complex". Proceedings of the National Academy of Sciences of the United States of America. 105 (2): 512–7. Bibcode:2008PNAS..105..512M. doi:10.1073/pnas.0710869105. PMC 2206567. PMID 18178619.
- Mourelatos Z, Dostie J, Paushkin S, Sharma A, Charroux B, Abel L, et al. (March 2002). "miRNPs: a novel class of ribonucleoproteins containing numerous microRNAs". Genes & Development. 16 (6): 720–8. doi:10.1101/gad.974702. PMC 155365. PMID 11914277.
- Meister G, Landthaler M, Peters L, Chen PY, Urlaub H, Lührmann R, et al. (December 2005). "Identification of novel argonaute-associated proteins". Current Biology. 15 (23): 2149–55. Bibcode:2005CBio...15.2149M. doi:10.1016/j.cub.2005.10.048. hdl:11858/00-001M-0000-0012-E763-B. PMID 16289642.
- Shaw G, Kamen R (29 August 1986). "A conserved AU sequence from the 3' untranslated region of GM-CSF mRNA mediates selective mRNA degradation". Cell. 46 (5): 659–667. doi:10.1016/0092-8674(86)90341-7. ISSN 0092-8674. PMID 3488815.
- Jing Q, Huang S, Guth S, Zarubin T, Motoyama A, Chen J, et al. (March 2005). "Involvement of microRNA in AU-rich element-mediated mRNA instability". Cell. 120 (5): 623–34. doi:10.1016/j.cell.2004.12.038. PMID 15766526.
- ^ Kai ZS, Pasquinelli AE (January 2010). "MicroRNA assassins: factors that regulate the disappearance of miRNAs". Nature Structural & Molecular Biology. 17 (1): 5–10. doi:10.1038/nsmb.1762. PMC 6417416. PMID 20051982.
- Chatterjee S, Grosshans H (September 2009). "Active turnover modulates mature microRNA activity in Caenorhabditis elegans". Nature. 461 (7263): 546–9. Bibcode:2009Natur.461..546C. doi:10.1038/nature08349. PMID 19734881. S2CID 4414841.
- ^ Morozova N, Zinovyev A, Nonne N, Pritchard LL, Gorban AN, Harel-Bellan A (September 2012). "Kinetic signatures of microRNA modes of action". RNA. 18 (9): 1635–55. doi:10.1261/rna.032284.112. PMC 3425779. PMID 22850425.
- Wang XJ, Reyes JL, Chua NH, Gaasterland T (2004). "Prediction and identification of Arabidopsis thaliana microRNAs and their mRNA targets". Genome Biology. 5 (9): R65. doi:10.1186/gb-2004-5-9-r65. PMC 522872. PMID 15345049.
- Kawasaki H, Taira K (2004). "MicroRNA-196 inhibits HOXB8 expression in myeloid differentiation of HL60 cells". Nucleic Acids Symposium Series. 48 (1): 211–2. doi:10.1093/nass/48.1.211. PMID 17150553.
- ^ Moxon S, Jing R, Szittya G, Schwach F, Rusholme Pilcher RL, Moulton V, et al. (October 2008). "Deep sequencing of tomato short RNAs identifies microRNAs targeting genes involved in fruit ripening". Genome Research. 18 (10): 1602–9. doi:10.1101/gr.080127.108. PMC 2556272. PMID 18653800.
- Mazière P, Enright AJ (June 2007). "Prediction of microRNA targets". Drug Discovery Today. 12 (11–12): 452–8. doi:10.1016/j.drudis.2007.04.002. PMID 17532529.
- Williams AE (February 2008). "Functional aspects of animal microRNAs". Cellular and Molecular Life Sciences. 65 (4): 545–62. doi:10.1007/s00018-007-7355-9. PMC 11131689. PMID 17965831. S2CID 5708394.
- Eulalio A, Huntzinger E, Nishihara T, Rehwinkel J, Fauser M, Izaurralde E (January 2009). "Deadenylation is a widespread effect of miRNA regulation". RNA. 15 (1): 21–32. doi:10.1261/rna.1399509. PMC 2612776. PMID 19029310.
- Bazzini AA, Lee MT, Giraldez AJ (April 2012). "Ribosome profiling shows that miR-430 reduces translation before causing mRNA decay in zebrafish". Science. 336 (6078): 233–7. Bibcode:2012Sci...336..233B. doi:10.1126/science.1215704. PMC 3547538. PMID 22422859.
- Djuranovic S, Nahvi A, Green R (April 2012). "miRNA-mediated gene silencing by translational repression followed by mRNA deadenylation and decay". Science. 336 (6078): 237–40. Bibcode:2012Sci...336..237D. doi:10.1126/science.1215691. PMC 3971879. PMID 22499947.
- Tan Y, Zhang B, Wu T, Skogerbø G, Zhu X, Guo X, et al. (February 2009). "Transcriptional inhibiton of Hoxd4 expression by miRNA-10a in human breast cancer cells". BMC Molecular Biology. 10 (1): 12. doi:10.1186/1471-2199-10-12. PMC 2680403. PMID 19232136.
- Hawkins PG, Morris KV (March 2008). "RNA and transcriptional modulation of gene expression". Cell Cycle. 7 (5): 602–7. doi:10.4161/cc.7.5.5522. PMC 2877389. PMID 18256543.
- Stark A, Brennecke J, Bushati N, Russell RB, Cohen SM (December 2005). "Animal MicroRNAs confer robustness to gene expression and have a significant impact on 3'UTR evolution". Cell. 123 (6): 1133–46. doi:10.1016/j.cell.2005.11.023. PMID 16337999.
- He L, Hannon GJ (July 2004). "MicroRNAs: small RNAs with a big role in gene regulation". Nature Reviews. Genetics. 5 (7): 522–531. doi:10.1038/nrg1379. PMID 15211354. S2CID 5270062.
- Li LC (2008). "Small RNA-Mediated Gene Activation". In Morris KV (ed.). RNA and the Regulation of Gene Expression: A Hidden Layer of Complexity. Horizon Scientific Press. ISBN 978-1-904455-25-7.
- Place RF, Li LC, Pookot D, Noonan EJ, Dahiya R (February 2008). "MicroRNA-373 induces expression of genes with complementary promoter sequences". Proceedings of the National Academy of Sciences of the United States of America. 105 (5): 1608–13. Bibcode:2008PNAS..105.1608P. doi:10.1073/pnas.0707594105. PMC 2234192. PMID 18227514. (Erratum: doi:10.1073/pnas.1803343115, PMID 29555737, Retraction Watch. If the erratum has been checked and does not affect the cited material, please replace
{{erratum|...}}with{{erratum|...|checked=yes}}.) - Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP (August 2011). "A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language?". Cell. 146 (3): 353–8. doi:10.1016/j.cell.2011.07.014. PMC 3235919. PMID 21802130.
- ^ Kumar S, Reddy PH (September 2016). "Are circulating microRNAs peripheral biomarkers for Alzheimer's disease?". Biochim Biophys Acta. 1862 (9): 1617–27. doi:10.1016/j.bbadis.2016.06.001. PMC 5343750. PMID 27264337.
- van den Berg MM, Krauskopf J, Ramaekers JG, et al. (February 2020). "Circulating microRNAs as potential biomarkers for psychiatric and neurodegenerative disorders". Prog Neurobiol. 185: 101732. doi:10.1016/j.pneurobio.2019.101732. PMID 31816349.
- Cuman C, Van Sinderen M, Gantier MP, Rainczuk K, Sorby K, Rombauts L, et al. (October 2015). "Human Blastocyst Secreted microRNA Regulate Endometrial Epithelial Cell Adhesion". eBioMedicine. 2 (10): 1528–1535. doi:10.1016/j.ebiom.2015.09.003. PMC 4634783. PMID 26629549.
- Zhou L, Miller C, Miraglia LJ, Romero A, Mure LS, Panda S, et al. (January 2021). "A genome-wide microRNA screen identifies the microRNA-183/96/182 cluster as a modulator of circadian rhythms". Proceedings of the National Academy of Sciences of the United States of America. 118 (1): e2020454118. Bibcode:2021PNAS..11820454Z. doi:10.1073/pnas.2020454118. PMC 7817116. PMID 33443164. S2CID 230713808.
- "MicroRNAs Play Key Role in Regulation of Circadian Rhythms". Science News. 6 January 2021.
- Axtell MJ, Bartel DP (June 2005). "Antiquity of microRNAs and their targets in land plants". The Plant Cell. 17 (6): 1658–73. doi:10.1105/tpc.105.032185. PMC 1143068. PMID 15849273.
- Tanzer A, Stadler PF (May 2004). "Molecular evolution of a microRNA cluster". Journal of Molecular Biology. 339 (2): 327–35. CiteSeerX 10.1.1.194.1598. doi:10.1016/j.jmb.2004.03.065. PMID 15136036.
- Chen K, Rajewsky N (February 2007). "The evolution of gene regulation by transcription factors and microRNAs". Nature Reviews Genetics. 8 (2): 93–103. doi:10.1038/nrg1990. PMID 17230196. S2CID 174231.
- Lee CT, Risom T, Strauss WM (April 2007). "Evolutionary conservation of microRNA regulatory circuits: an examination of microRNA gene complexity and conserved microRNA-target interactions through metazoan phylogeny". DNA and Cell Biology. 26 (4): 209–18. doi:10.1089/dna.2006.0545. PMID 17465887.
- ^ Peterson KJ, Dietrich MR, McPeek MA (July 2009). "MicroRNAs and metazoan macroevolution: insights into canalization, complexity, and the Cambrian explosion". BioEssays. 31 (7): 736–47. doi:10.1002/bies.200900033. PMID 19472371. S2CID 15364875.
- Shabalina SA, Koonin EV (October 2008). "Origins and evolution of eukaryotic RNA interference". Trends in Ecology & Evolution. 23 (10): 578–87. Bibcode:2008TEcoE..23..578S. doi:10.1016/j.tree.2008.06.005. PMC 2695246. PMID 18715673.
- Axtell MJ, Westholm JO, Lai EC (2011). "Vive la différence: biogenesis and evolution of microRNAs in plants and animals". Genome Biology. 12 (4): 221. doi:10.1186/gb-2011-12-4-221. PMC 3218855. PMID 21554756.
- ^ Wheeler BM, Heimberg AM, Moy VN, Sperling EA, Holstein TW, Heber S, et al. (2009). "The deep evolution of metazoan microRNAs". Evolution & Development. 11 (1): 50–68. doi:10.1111/j.1525-142X.2008.00302.x. PMID 19196333. S2CID 14924603.
- Pashkovskiy PP, Ryazansky SS (June 2013). "Biogenesis, evolution, and functions of plant microRNAs". Biochemistry. Biokhimiia. 78 (6): 627–37. doi:10.1134/S0006297913060084. PMID 23980889. S2CID 12025420.
- ^ Heimberg AM, Sempere LF, Moy VN, Donoghue PC, Peterson KJ (February 2008). "MicroRNAs and the advent of vertebrate morphological complexity". Proceedings of the National Academy of Sciences of the United States of America. 105 (8): 2946–50. Bibcode:2008PNAS..105.2946H. doi:10.1073/pnas.0712259105. PMC 2268565. PMID 18287013.
- ^ Nozawa M, Miura S, Nei M (July 2010). "Origins and evolution of microRNA genes in Drosophila species". Genome Biology and Evolution. 2: 180–9. doi:10.1093/gbe/evq009. PMC 2942034. PMID 20624724.
- Allen E, Xie Z, Gustafson AM, Sung GH, Spatafora JW, Carrington JC (December 2004). "Evolution of microRNA genes by inverted duplication of target gene sequences in Arabidopsis thaliana". Nature Genetics. 36 (12): 1282–90. doi:10.1038/ng1478. PMID 15565108. S2CID 11997028.
- Warthmann N, Das S, Lanz C, Weigel D (May 2008). "Comparative analysis of the MIR319a microRNA locus in Arabidopsis and related Brassicaceae". Molecular Biology and Evolution. 25 (5): 892–902. doi:10.1093/molbev/msn029. PMID 18296705.
- Fahlgren N, Jogdeo S, Kasschau KD, Sullivan CM, Chapman EJ, Laubinger S, et al. (April 2010). "MicroRNA gene evolution in Arabidopsis lyrata and Arabidopsis thaliana". The Plant Cell. 22 (4): 1074–89. doi:10.1105/tpc.110.073999. PMC 2879733. PMID 20407027.
- Caravas J, Friedrich M (June 2010). "Of mites and millipedes: recent progress in resolving the base of the arthropod tree". BioEssays. 32 (6): 488–95. doi:10.1002/bies.201000005. PMID 20486135. S2CID 20548122.
- Kenny NJ, Namigai EK, Marlétaz F, Hui JH, Shimeld SM (December 2015). "Draft genome assemblies and predicted microRNA complements of the intertidal lophotrochozoans Patella vulgata (Mollusca, Patellogastropoda) and Spirobranchus (Pomatoceros) lamarcki (Annelida, Serpulida)". Marine Genomics. 24 (2): 139–46. Bibcode:2015MarGn..24..139K. doi:10.1016/j.margen.2015.07.004. PMID 26319627.
- Cock JM, Sterck L, Rouzé P, Scornet D, Allen AE, Amoutzias G, et al. (June 2010). "The Ectocarpus genome and the independent evolution of multicellularity in brown algae". Nature. 465 (7298): 617–21. Bibcode:2010Natur.465..617C. doi:10.1038/nature09016. PMID 20520714.
- Cuperus JT, Fahlgren N, Carrington JC (February 2011). "Evolution and functional diversification of MIRNA genes". The Plant Cell. 23 (2): 431–42. doi:10.1105/tpc.110.082784. PMC 3077775. PMID 21317375.
- Ryan JF, Pang K, Schnitzler CE, Nguyen AD, Moreland RT, Simmons DK, et al. (December 2013). "The genome of the ctenophore Mnemiopsis leidyi and its implications for cell type evolution". Science. 342 (6164): 1242592. doi:10.1126/science.1242592. PMC 3920664. PMID 24337300.
- Maxwell EK, Ryan JF, Schnitzler CE, Browne WE, Baxevanis AD (December 2012). "MicroRNAs and essential components of the microRNA processing machinery are not encoded in the genome of the ctenophore Mnemiopsis leidyi". BMC Genomics. 13 (1): 714. doi:10.1186/1471-2164-13-714. PMC 3563456. PMID 23256903.
- Dimond PF (15 March 2010). "miRNAs' Therapeutic Potential". Genetic Engineering & Biotechnology News. 30 (6): 1. Archived from the original on 19 July 2010. Retrieved 10 July 2010.
- Tjaden B, Goodwin SS, Opdyke JA, Guillier M, Fu DX, Gottesman S, et al. (2006). "Target prediction for small, noncoding RNAs in bacteria". Nucleic Acids Research. 34 (9): 2791–802. doi:10.1093/nar/gkl356. PMC 1464411. PMID 16717284.
- Liu CG, Calin GA, Volinia S, Croce CM (2008). "MicroRNA expression profiling using microarrays". Nature Protocols. 3 (4): 563–78. doi:10.1038/nprot.2008.14. PMID 18388938. S2CID 2441105.
- Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee DH, Nguyen JT, et al. (November 2005). "Real-time quantification of microRNAs by stem-loop RT-PCR". Nucleic Acids Research. 33 (20): e179. doi:10.1093/nar/gni178. PMC 1292995. PMID 16314309.
- Shingara J, Keiger K, Shelton J, Laosinchai-Wolf W, Powers P, Conrad R, et al. (September 2005). "An optimized isolation and labeling platform for accurate microRNA expression profiling". RNA. 11 (9): 1461–70. doi:10.1261/rna.2610405. PMC 1370829. PMID 16043497.
- Buermans HP, Ariyurek Y, van Ommen G, den Dunnen JT, 't Hoen PA (December 2010). "New methods for next generation sequencing based microRNA expression profiling". BMC Genomics. 11: 716. doi:10.1186/1471-2164-11-716. PMC 3022920. PMID 21171994.
- Kloosterman WP, Wienholds E, Ketting RF, Plasterk RH (2004). "Substrate requirements for let-7 function in the developing zebrafish embryo". Nucleic Acids Research. 32 (21): 6284–91. doi:10.1093/nar/gkh968. PMC 535676. PMID 15585662.
- Flynt AS, Li N, Thatcher EJ, Solnica-Krezel L, Patton JG (February 2007). "Zebrafish miR-214 modulates Hedgehog signaling to specify muscle cell fate". Nature Genetics. 39 (2): 259–63. doi:10.1038/ng1953. PMC 3982799. PMID 17220889.
- Meister G, Landthaler M, Dorsett Y, Tuschl T (March 2004). "Sequence-specific inhibition of microRNA- and siRNA-induced RNA silencing". RNA. 10 (3): 544–50. doi:10.1261/rna.5235104. PMC 1370948. PMID 14970398.
- Kloosterman WP, Lagendijk AK, Ketting RF, Moulton JD, Plasterk RH (August 2007). "Targeted inhibition of miRNA maturation with morpholinos reveals a role for miR-375 in pancreatic islet development". PLOS Biology. 5 (8): e203. doi:10.1371/journal.pbio.0050203. PMC 1925136. PMID 17676975. (This paper currently has an expression of concern, see doi:10.1371/journal.pbio.3001631, PMID 35486652. If this is an intentional citation to a such a paper, please replace
{{expression of concern|...}}with{{expression of concern|...|intentional=yes}}.) - Gebert LF, Rebhan MA, Crivelli SE, Denzler R, Stoffel M, Hall J (January 2014). "Miravirsen (SPC3649) can inhibit the biogenesis of miR-122". Nucleic Acids Research. 42 (1): 609–21. doi:10.1093/nar/gkt852. PMC 3874169. PMID 24068553.
- Choi WY, Giraldez AJ, Schier AF (October 2007). "Target protectors reveal dampening and balancing of Nodal agonist and antagonist by miR-430". Science. 318 (5848): 271–4. Bibcode:2007Sci...318..271C. doi:10.1126/science.1147535. PMID 17761850. S2CID 30461594.
- You Y, Moreira BG, Behlke MA, Owczarzy R (May 2006). "Design of LNA probes that improve mismatch discrimination". Nucleic Acids Research. 34 (8): e60. doi:10.1093/nar/gkl175. PMC 1456327. PMID 16670427.
- Lagendijk AK, Moulton JD, Bakkers J (June 2012). "Revealing details: whole mount microRNA in situ hybridization protocol for zebrafish embryos and adult tissues". Biology Open. 1 (6): 566–9. doi:10.1242/bio.2012810. PMC 3509442. PMID 23213449.
- Kaur H, Arora A, Wengel J, Maiti S (June 2006). "Thermodynamic, counterion, and hydration effects for the incorporation of locked nucleic acid nucleotides into DNA duplexes". Biochemistry. 45 (23): 7347–55. doi:10.1021/bi060307w. PMID 16752924.
- Nielsen JA, Lau P, Maric D, Barker JL, Hudson LD (August 2009). "Integrating microRNA and mRNA expression profiles of neuronal progenitors to identify regulatory networks underlying the onset of cortical neurogenesis". BMC Neuroscience. 10: 98. doi:10.1186/1471-2202-10-98. PMC 2736963. PMID 19689821.
- Grimson A, Farh KK, Johnston WK, Garrett-Engele P, Lim LP, Bartel DP (July 2007). "MicroRNA targeting specificity in mammals: determinants beyond seed pairing". Molecular Cell. 27 (1): 91–105. doi:10.1016/j.molcel.2007.06.017. PMC 3800283. PMID 17612493.
- Griffiths-Jones S, Saini HK, van Dongen S, Enright AJ (January 2008). "miRBase: tools for microRNA genomics". Nucleic Acids Research. 36 (Database issue): D154–8. doi:10.1093/nar/gkm952. PMC 2238936. PMID 17991681.
- Nam S, Li M, Choi K, Balch C, Kim S, Nephew KP (July 2009). "MicroRNA and mRNA integrated analysis (MMIA): a web tool for examining biological functions of microRNA expression". Nucleic Acids Research. 37 (Web Server issue): W356–62. doi:10.1093/nar/gkp294. PMC 2703907. PMID 19420067.
- Artmann S, Jung K, Bleckmann A, Beissbarth T (2012). Provero P (ed.). "Detection of simultaneous group effects in microRNA expression and related target gene sets". PLOS ONE. 7 (6): e38365. Bibcode:2012PLoSO...738365A. doi:10.1371/journal.pone.0038365. PMC 3378551. PMID 22723856.
- Jiang Q, Wang Y, Hao Y, Juan L, Teng M, Zhang X, et al. (January 2009). "miR2Disease: a manually curated database for microRNA deregulation in human disease". Nucleic Acids Research. 37. 37 (Database issue): D98–104. doi:10.1093/nar/gkn714. PMC 2686559. PMID 18927107.
- Mencía A, Modamio-Høybjør S, Redshaw N, Morín M, Mayo-Merino F, Olavarrieta L, et al. (May 2009). "Mutations in the seed region of human miR-96 are responsible for nonsyndromic progressive hearing loss". Nature Genetics. 41 (5): 609–13. doi:10.1038/ng.355. PMID 19363479. S2CID 11113852.
- Hughes AE, Bradley DT, Campbell M, Lechner J, Dash DP, Simpson DA, et al. (November 2011). "Mutation altering the miR-184 seed region causes familial keratoconus with cataract". American Journal of Human Genetics. 89 (5): 628–33. doi:10.1016/j.ajhg.2011.09.014. PMC 3213395. PMID 21996275.
- de Pontual L, Yao E, Callier P, Faivre L, Drouin V, Cariou S, et al. (September 2011). "Germline deletion of the miR-17~92 cluster causes skeletal and growth defects in humans". Nature Genetics. 43 (10): 1026–30. doi:10.1038/ng.915. PMC 3184212. PMID 21892160.
- Calin GA, Liu CG, Sevignani C, Ferracin M, Felli N, Dumitru CD, et al. (August 2004). "MicroRNA profiling reveals distinct signatures in B cell chronic lymphocytic leukemias". Proceedings of the National Academy of Sciences of the United States of America. 101 (32): 11755–11760. Bibcode:2004PNAS..10111755C. doi:10.1073/pnas.0404432101. PMC 511048. PMID 15284443.
- Velle A, Pesenti C, Grassi T, Beltrame L, Martini P, Jaconi M, et al. (May 2023). "A comprehensive investigation of histotype-specific microRNA and their variants in Stage I epithelial ovarian cancers". International Journal of Cancer. 152 (9): 1989–2001. doi:10.1002/ijc.34408. hdl:11577/3478062. PMID 36541726. S2CID 255034585.
- Kyriakidis I, Kyriakidis K, Tsezou A (August 2022). "MicroRNAs and the Diagnosis of Childhood Acute Lymphoblastic Leukemia: Systematic Review, Meta-Analysis and Re-Analysis with Novel Small RNA-Seq Tools". Cancers. 14 (16): 3976. doi:10.3390/cancers14163976. PMC 9406077. PMID 36010971.
- Võsa U, Vooder T, Kolde R, Fischer K, Välk K, Tõnisson N, et al. (October 2011). "Identification of miR-374a as a prognostic marker for survival in patients with early-stage nonsmall cell lung cancer". Genes, Chromosomes & Cancer. 50 (10): 812–22. doi:10.1002/gcc.20902. PMID 21748820. S2CID 9746594.
- Akçakaya P, Ekelund S, Kolosenko I, Caramuta S, Ozata DM, Xie H, et al. (August 2011). "miR-185 and miR-133b deregulation is associated with overall survival and metastasis in colorectal cancer". International Journal of Oncology. 39 (2): 311–8. doi:10.3892/ijo.2011.1043. PMID 21573504.
- ^ Eyking A, Reis H, Frank M, Gerken G, Schmid KW, Cario E (2016). "MiR-205 and MiR-373 Are Associated with Aggressive Human Mucinous Colorectal Cancer". PLOS ONE. 11 (6): e0156871. Bibcode:2016PLoSO..1156871E. doi:10.1371/journal.pone.0156871. PMC 4894642. PMID 27271572.
- MicroRNA-21 promotes hepatocellular carcinoma HepG2 cell proliferation through repression of mitogen-activated protein kinase-kinase 3. Guangxian Xu et al., 2013
- Jones K, Nourse JP, Keane C, Bhatnagar A, Gandhi MK (January 2014). "Plasma microRNA are disease response biomarkers in classical Hodgkin lymphoma". Clinical Cancer Research. 20 (1): 253–64. doi:10.1158/1078-0432.CCR-13-1024. PMID 24222179.
- Hosseinahli N, Aghapour M, Duijf PH, Baradaran B (August 2018). "Treating cancer with microRNA replacement therapy: A literature review". Journal of Cellular Physiology. 233 (8): 5574–5588. doi:10.1002/jcp.26514. PMID 29521426. S2CID 3766576.
- Liu G, Sun Y, Ji P, Li X, Cogdell D, Yang D, et al. (July 2014). "MiR-506 suppresses proliferation and induces senescence by directly targeting the CDK4/6-FOXM1 axis in ovarian cancer". The Journal of Pathology. 233 (3): 308–18. doi:10.1002/path.4348. PMC 4144705. PMID 24604117.
- Wen SY, Lin Y, Yu YQ, Cao SJ, Zhang R, Yang XM, et al. (February 2015). "miR-506 acts as a tumor suppressor by directly targeting the hedgehog pathway transcription factor Gli3 in human cervical cancer". Oncogene. 34 (6): 717–25. doi:10.1038/onc.2014.9. PMID 24608427. S2CID 20603801.
- Peking University (13 September 2022). "Scientists Develop a New, Powerful Cancer-Fighting Weapon". Cell. 185 (11). SciTech Daily: 1888–1904.e24. doi:10.1016/j.cell.2022.04.030. PMID 35623329. S2CID 249070106. Retrieved 15 September 2022.
- Loeb KR, Loeb LA (March 2000). "Significance of multiple mutations in cancer". Carcinogenesis. 21 (3): 379–385. doi:10.1093/carcin/21.3.379. PMID 10688858.
- Lodish H, Berk A, Kaiser CA, Krieger M, Bretscher A, Ploegh H, et al. (2016). Molecular Cell Biology (8th ed.). New York: W. H. Freeman and Company. p. 203. ISBN 978-1-4641-8339-3.
- Hu H, Gatti RA (June 2011). "MicroRNAs: new players in the DNA damage response". Journal of Molecular Cell Biology. 3 (3): 151–158. doi:10.1093/jmcb/mjq042. PMC 3104011. PMID 21183529.
- Jasperson KW, Tuohy TM, Neklason DW, Burt RW (June 2010). "Hereditary and familial colon cancer". Gastroenterology. 138 (6): 2044–58. doi:10.1053/j.gastro.2010.01.054. PMC 3057468. PMID 20420945.
- Truninger K, Menigatti M, Luz J, Russell A, Haider R, Gebbers JO, et al. (May 2005). "Immunohistochemical analysis reveals high frequency of PMS2 defects in colorectal cancer". Gastroenterology. 128 (5): 1160–71. doi:10.1053/j.gastro.2005.01.056. PMID 15887099.
- Valeri N, Gasparini P, Fabbri M, Braconi C, Veronese A, Lovat F, et al. (April 2010). "Modulation of mismatch repair and genomic stability by miR-155". Proceedings of the National Academy of Sciences of the United States of America. 107 (15): 6982–7. Bibcode:2010PNAS..107.6982V. doi:10.1073/pnas.1002472107. PMC 2872463. PMID 20351277.
- ^ Zhang W, Zhang J, Hoadley K, Kushwaha D, Ramakrishnan V, Li S, et al. (June 2012). "miR-181d: a predictive glioblastoma biomarker that downregulates MGMT expression". Neuro-Oncology. 14 (6): 712–9. doi:10.1093/neuonc/nos089. PMC 3367855. PMID 22570426.
- Spiegl-Kreinecker S, Pirker C, Filipits M, Lötsch D, Buchroithner J, Pichler J, et al. (January 2010). "O6-Methylguanine DNA methyltransferase protein expression in tumor cells predicts outcome of temozolomide therapy in glioblastoma patients". Neuro-Oncology. 12 (1): 28–36. doi:10.1093/neuonc/nop003. PMC 2940563. PMID 20150365.
- Sgarra R, Rustighi A, Tessari MA, Di Bernardo J, Altamura S, Fusco A, et al. (September 2004). "Nuclear phosphoproteins HMGA and their relationship with chromatin structure and cancer". FEBS Letters. 574 (1–3): 1–8. Bibcode:2004FEBSL.574....1S. doi:10.1016/j.febslet.2004.08.013. PMID 15358530. S2CID 28903539.
- Xu Y, Sumter TF, Bhattacharya R, Tesfaye A, Fuchs EJ, Wood LJ, et al. (May 2004). "The HMG-I oncogene causes highly penetrant, aggressive lymphoid malignancy in transgenic mice and is overexpressed in human leukemia". Cancer Research. 64 (10): 3371–5. doi:10.1158/0008-5472.CAN-04-0044. PMID 15150086.
- Borrmann L, Schwanbeck R, Heyduk T, Seebeck B, Rogalla P, Bullerdiek J, et al. (December 2003). "High mobility group A2 protein and its derivatives bind a specific region of the promoter of DNA repair gene ERCC1 and modulate its activity". Nucleic Acids Research. 31 (23): 6841–51. doi:10.1093/nar/gkg884. PMC 290254. PMID 14627817.
- Facista A, Nguyen H, Lewis C, Prasad AR, Ramsey L, Zaitlin B, et al. (April 2012). "Deficient expression of DNA repair enzymes in early progression to sporadic colon cancer". Genome Integrity. 3 (1): 3. doi:10.1186/2041-9414-3-3. PMC 3351028. PMID 22494821.
- Gibert B, Delloye-Bourgeois C, Gattolliat CH, Meurette O, Le Guernevel S, Fombonne J, et al. (November 2014). "Regulation by miR181 family of the dependence receptor CDON tumor suppressive activity in neuroblastoma". Journal of the National Cancer Institute. 106 (11). doi:10.1093/jnci/dju318. PMID 25313246.
- Chen JF, Murchison EP, Tang R, Callis TE, Tatsuguchi M, Deng Z, et al. (February 2008). "Targeted deletion of Dicer in the heart leads to dilated cardiomyopathy and heart failure". Proceedings of the National Academy of Sciences of the United States of America. 105 (6): 2111–6. Bibcode:2008PNAS..105.2111C. doi:10.1073/pnas.0710228105. PMC 2542870. PMID 18256189.
- ^ Zhao Y, Ransom JF, Li A, Vedantham V, von Drehle M, Muth AN, et al. (April 2007). "Dysregulation of cardiogenesis, cardiac conduction, and cell cycle in mice lacking miRNA-1-2". Cell. 129 (2): 303–17. doi:10.1016/j.cell.2007.03.030. PMID 17397913.
- Thum T, Galuppo P, Wolf C, Fiedler J, Kneitz S, van Laake LW, et al. (July 2007). "MicroRNAs in the human heart: a clue to fetal gene reprogramming in heart failure". Circulation. 116 (3): 258–67. doi:10.1161/CIRCULATIONAHA.107.687947. PMID 17606841.
- van Rooij E, Sutherland LB, Liu N, Williams AH, McAnally J, Gerard RD, et al. (November 2006). "A signature pattern of stress-responsive microRNAs that can evoke cardiac hypertrophy and heart failure". Proceedings of the National Academy of Sciences of the United States of America. 103 (48): 18255–60. Bibcode:2006PNAS..10318255V. doi:10.1073/pnas.0608791103. PMC 1838739. PMID 17108080.
- Tatsuguchi M, Seok HY, Callis TE, Thomson JM, Chen JF, Newman M, et al. (June 2007). "Expression of microRNAs is dynamically regulated during cardiomyocyte hypertrophy". Journal of Molecular and Cellular Cardiology. 42 (6): 1137–41. doi:10.1016/j.yjmcc.2007.04.004. PMC 1934409. PMID 17498736.
- Zhao Y, Samal E, Srivastava D (July 2005). "Serum response factor regulates a muscle-specific microRNA that targets Hand2 during cardiogenesis". Nature. 436 (7048): 214–20. Bibcode:2005Natur.436..214Z. doi:10.1038/nature03817. PMID 15951802. S2CID 4340449.
- Xiao J, Luo X, Lin H, Zhang Y, Lu Y, Wang N, et al. (April 2007). "MicroRNA miR-133 represses HERG K+ channel expression contributing to QT prolongation in diabetic hearts". The Journal of Biological Chemistry. 282 (17): 12363–7. doi:10.1074/jbc.C700015200. PMC 3151106. PMID 17344217. (Retracted, see doi:10.1074/jbc.A111.700015, PMID 21961173, Retraction Watch. If this is an intentional citation to a retracted paper, please replace
{{retracted|...}}with{{retracted|...|intentional=yes}}.) - Yang B, Lin H, Xiao J, Lu Y, Luo X, Li B, et al. (April 2007). "The muscle-specific microRNA miR-1 regulates cardiac arrhythmogenic potential by targeting GJA1 and KCNJ2". Nature Medicine. 13 (4): 486–91. doi:10.1038/nm1569. PMID 17401374. S2CID 1935811.
- Carè A, Catalucci D, Felicetti F, Bonci D, Addario A, Gallo P, et al. (May 2007). "MicroRNA-133 controls cardiac hypertrophy". Nature Medicine. 13 (5): 613–8. doi:10.1038/nm1582. PMID 17468766. S2CID 10097893.
- van Rooij E, Sutherland LB, Qi X, Richardson JA, Hill J, Olson EN (April 2007). "Control of stress-dependent cardiac growth and gene expression by a microRNA". Science. 316 (5824): 575–9. Bibcode:2007Sci...316..575V. doi:10.1126/science.1139089. PMID 17379774. S2CID 1927839.
- Keller T, Boeckel JN, Groß S, Klotsche J, Palapies L, Leistner D, et al. (July 2017). "Improved risk stratification in prevention by use of a panel of selected circulating microRNAs". Scientific Reports. 7 (1): 4511. Bibcode:2017NatSR...7.4511K. doi:10.1038/s41598-017-04040-w. PMC 5495799. PMID 28674420.
- Insull W (January 2009). "The pathology of atherosclerosis: plaque development and plaque responses to medical treatment". The American Journal of Medicine. 122 (1 Suppl): S3–S14. doi:10.1016/j.amjmed.2008.10.013. PMID 19110086.
- ^ Son DJ, Kumar S, Takabe W, Kim CW, Ni CW, Alberts-Grill N, et al. (2013). "The atypical mechanosensitive microRNA-712 derived from pre-ribosomal RNA induces endothelial inflammation and atherosclerosis". Nature Communications. 4: 3000. Bibcode:2013NatCo...4.3000S. doi:10.1038/ncomms4000. PMC 3923891. PMID 24346612.
- ^ Basu R, Fan D, Kandalam V, Lee J, Das SK, Wang X, et al. (December 2012). "Loss of Timp3 gene leads to abdominal aortic aneurysm formation in response to angiotensin II". The Journal of Biological Chemistry. 287 (53): 44083–96. doi:10.1074/jbc.M112.425652. PMC 3531724. PMID 23144462.
- Libby P (2002). "Inflammation in atherosclerosis". Nature. 420 (6917): 868–74. Bibcode:2002Natur.420..868L. doi:10.1038/nature01323. PMID 12490960. S2CID 407449.
- ^ Phua YL, Chu JY, Marrone AK, Bodnar AJ, Sims-Lucas S, Ho J (October 2015). "Renal stromal miRNAs are required for normal nephrogenesis and glomerular mesangial survival". Physiological Reports. 3 (10): e12537. doi:10.14814/phy2.12537. PMC 4632944. PMID 26438731.
- Cao DD, Li L, Chan WY (May 2016). "MicroRNAs: Key Regulators in the Central Nervous System and Their Implication in Neurological Diseases". International Journal of Molecular Sciences. 17 (6): 842. doi:10.3390/ijms17060842. PMC 4926376. PMID 27240359.
- Lang MF, Shi Y (2012). "Dynamic Roles of microRNAs in Neurogenesis". Frontiers in Neuroscience. 6: 71. doi:10.3389/fnins.2012.00071. PMC 3356852. PMID 22661924.
- Schratt G (December 2009). "microRNAs at the synapse". Nature Reviews. Neuroscience. 10 (12): 842–849. doi:10.1038/nrn2763. PMID 19888283. S2CID 3507952.
- Luo M, Li L, Ding M, Niu Y, Xu X, Shi X, et al. (19 January 2023). "Long-term potentiation and depression regulatory microRNAs were highlighted in Bisphenol A induced learning and memory impairment by microRNA sequencing and bioinformatics analysis". PLOS ONE. 18 (1): e0279029. Bibcode:2023PLoSO..1879029L. doi:10.1371/journal.pone.0279029. PMC 9851566. PMID 36656826.
- Cuellar TL, Davis TH, Nelson PT, Loeb GB, Harfe BD, Ullian E, et al. (April 2008). "Dicer loss in striatal neurons produces behavioral and neuroanatomical phenotypes in the absence of neurodegeneration". Proceedings of the National Academy of Sciences of the United States of America. 105 (14): 5614–5619. Bibcode:2008PNAS..105.5614C. doi:10.1073/pnas.0801689105. PMC 2291142. PMID 18385371.
- Hébert SS, Papadopoulou AS, Smith P, Galas MC, Planel E, Silahtaroglu AN, et al. (October 2010). "Genetic ablation of Dicer in adult forebrain neurons results in abnormal tau hyperphosphorylation and neurodegeneration". Human Molecular Genetics. 19 (20): 3959–3969. doi:10.1093/hmg/ddq311. PMID 20660113.
- Roy B, Lee E, Li T, Rampersaud M (February 2022). "Role of miRNAs in Neurodegeneration: From Disease Cause to Tools of Biomarker Discovery and Therapeutics". Genes. 13 (3): 425. doi:10.3390/genes13030425. PMC 8951370. PMID 35327979.
- Henshall DC, Hamer HM, Pasterkamp RJ, Goldstein DB, Kjems J, Prehn JH, et al. (December 2016). "MicroRNAs in epilepsy: pathophysiology and clinical utility". The Lancet. Neurology. 15 (13): 1368–1376. doi:10.1016/S1474-4422(16)30246-0. PMID 27839653.
- Hommers LG, Domschke K, Deckert J (January 2015). "Heterogeneity and individuality: microRNAs in mental disorders" (PDF). Journal of Neural Transmission. 122 (1): 79–97. doi:10.1007/s00702-014-1338-4. PMID 25395183. S2CID 25088900. Archived (PDF) from the original on 23 May 2022.
- Feng J, Sun G, Yan J, Noltner K, Li W, Buzin CH, et al. (July 2009). Reif A (ed.). "Evidence for X-chromosomal schizophrenia associated with microRNA alterations". PLOS ONE. 4 (7): e6121. Bibcode:2009PLoSO...4.6121F. doi:10.1371/journal.pone.0006121. PMC 2699475. PMID 19568434.
- Beveridge NJ, Gardiner E, Carroll AP, Tooney PA, Cairns MJ (December 2010). "Schizophrenia is associated with an increase in cortical microRNA biogenesis". Molecular Psychiatry. 15 (12): 1176–1189. doi:10.1038/mp.2009.84. PMC 2990188. PMID 19721432.
- "Stroke Facts". Centers for Disease Control and Prevention. 15 March 2019. Retrieved 5 December 2019.
- Rink C, Khanna S (May 2011). "MicroRNA in ischemic stroke etiology and pathology". Physiological Genomics. 43 (10): 521–528. doi:10.1152/physiolgenomics.00158.2010. PMC 3110894. PMID 20841499.
- Ouyang YB, Stary CM, Yang GY, Giffard R (January 2013). "microRNAs: innovative targets for cerebral ischemia and stroke". Current Drug Targets. 14 (1): 90–101. doi:10.2174/138945013804806424. PMC 3673881. PMID 23170800.
- ^ Lewohl JM, Nunez YO, Dodd PR, Tiwari GR, Harris RA, Mayfield RD (November 2011). "Up-regulation of microRNAs in brain of human alcoholics". Alcoholism: Clinical and Experimental Research. 35 (11): 1928–37. doi:10.1111/j.1530-0277.2011.01544.x. PMC 3170679. PMID 21651580.
- Tapocik JD, Solomon M, Flanigan M, Meinhardt M, Barbier E, Schank JR, et al. (June 2013). "Coordinated dysregulation of mRNAs and microRNAs in the rat medial prefrontal cortex following a history of alcohol dependence". The Pharmacogenomics Journal. 13 (3): 286–96. doi:10.1038/tpj.2012.17. PMC 3546132. PMID 22614244.
- Gorini G, Nunez YO, Mayfield RD (2013). "Integration of miRNA and protein profiling reveals coordinated neuroadaptations in the alcohol-dependent mouse brain". PLOS ONE. 8 (12): e82565. Bibcode:2013PLoSO...882565G. doi:10.1371/journal.pone.0082565. PMC 3865091. PMID 24358208.
- Tapocik JD, Barbier E, Flanigan M, Solomon M, Pincus A, Pilling A, et al. (March 2014). "microRNA-206 in rat medial prefrontal cortex regulates BDNF expression and alcohol drinking". The Journal of Neuroscience. 34 (13): 4581–8. doi:10.1523/JNEUROSCI.0445-14.2014. PMC 3965783. PMID 24672003.
- Lippai D, Bala S, Csak T, Kurt-Jones EA, Szabo G (2013). "Chronic alcohol-induced microRNA-155 contributes to neuroinflammation in a TLR4-dependent manner in mice". PLOS ONE. 8 (8): e70945. Bibcode:2013PLoSO...870945L. doi:10.1371/journal.pone.0070945. PMC 3739772. PMID 23951048.
- ^ Li J, Li J, Liu X, Qin S, Guan Y, Liu Y, et al. (September 2013). "MicroRNA expression profile and functional analysis reveal that miR-382 is a critical novel gene of alcohol addiction". EMBO Molecular Medicine. 5 (9): 1402–14. doi:10.1002/emmm.201201900. PMC 3799494. PMID 23873704.
- Romao JM, Jin W, Dodson MV, Hausman GJ, Moore SS, Guan LL (September 2011). "MicroRNA regulation in mammalian adipogenesis". Experimental Biology and Medicine. 236 (9): 997–1004. doi:10.1258/ebm.2011.011101. PMID 21844119. S2CID 30646787.
- Skårn M, Namløs HM, Noordhuis P, Wang MY, Meza-Zepeda LA, Myklebost O (April 2012). "Adipocyte differentiation of human bone marrow-derived stromal cells is modulated by microRNA-155, microRNA-221, and microRNA-222". Stem Cells and Development. 21 (6): 873–83. doi:10.1089/scd.2010.0503. hdl:10852/40423. PMID 21756067.
- Zuo Y, Qiang L, Farmer SR (March 2006). "Activation of CCAAT/enhancer-binding protein (C/EBP) alpha expression by C/EBP beta during adipogenesis requires a peroxisome proliferator-activated receptor-gamma-associated repression of HDAC1 at the C/ebp alpha gene promoter". The Journal of Biological Chemistry. 281 (12): 7960–7. doi:10.1074/jbc.M510682200. PMID 16431920.
- Jun-Hao ET, Gupta RR, Shyh-Chang N (March 2016). "Lin28 and let-7 in the Metabolic Physiology of Aging". Trends in Endocrinology and Metabolism. 27 (3): 132–141. doi:10.1016/j.tem.2015.12.006. PMID 26811207. S2CID 3614126.
- Zhu H, Shyh-Chang N, Segrè AV, Shinoda G, Shah SP, Einhorn WS, et al. (September 2011). "The Lin28/let-7 axis regulates glucose metabolism". Cell. 147 (1): 81–94. doi:10.1016/j.cell.2011.08.033. PMC 3353524. PMID 21962509.
- Frost RJ, Olson EN (December 2011). "Control of glucose homeostasis and insulin sensitivity by the Let-7 family of microRNAs". Proceedings of the National Academy of Sciences of the United States of America. 108 (52): 21075–80. Bibcode:2011PNAS..10821075F. doi:10.1073/pnas.1118922109. PMC 3248488. PMID 22160727.
- Teruel-Montoya R, Rosendaal FR, Martínez C (February 2015). "MicroRNAs in hemostasis". Journal of Thrombosis and Haemostasis. 13 (2): 170–181. doi:10.1111/jth.12788. PMID 25400249.
- ^ Nourse J, Braun J, Lackner K, Hüttelmaier S, Danckwardt S (November 2018). "Large-scale identification of functional microRNA targeting reveals cooperative regulation of the hemostatic system". Journal of Thrombosis and Haemostasis. 16 (11): 2233–2245. doi:10.1111/jth.14290. PMID 30207063.
- Nourse J, Danckwardt S (February 2021). "A novel rationale for targeting FXI: Insights from the hemostatic microRNA targetome for emerging anticoagulant strategies". Pharmacology & Therapeutics. 218: 107676. doi:10.1016/j.pharmthera.2020.107676. PMID 32898547.
- Berardi E, Pues M, Thorrez L, Sampaolesi M (October 2012). "miRNAs in ESC differentiation". American Journal of Physiology. Heart and Circulatory Physiology. 303 (8): H931–H939. doi:10.1152/ajpheart.00338.2012. PMID 22886416. S2CID 6402014.
- ^ Borchers A, Pieler T (November 2010). "Programming pluripotent precursor cells derived from Xenopus embryos to generate specific tissues and organs". Genes. 1 (3): 413–426. doi:10.3390/ijms232314755. PMC 9740008. PMID 36499082.
- Curaba J, Spriggs A, Taylor J, Li Z, Helliwell C (July 2012). "miRNA regulation in the early development of barley seed". BMC Plant Biology. 12 (1): 120. doi:10.1186/1471-2229-12-120. PMC 3443071. PMID 22838835.
- Choudhary A, Kumar A, Kaur H, Kaur N (May 2021). "MiRNA: the taskmaster of plant world". Biologia. 76 (5): 1551–1567. Bibcode:2021Biolg..76.1551C. doi:10.1007/s11756-021-00720-1. ISSN 1336-9563. S2CID 233685660.
- Waheed S, Zeng L (March 2020). "The Critical Role of miRNAs in Regulation of Flowering Time and Flower Development". Genes. 11 (3): 319. doi:10.3390/genes11030319. PMC 7140873. PMID 32192095.
- Wong CE, Zhao YT, Wang XJ, Croft L, Wang ZH, Haerizadeh F, et al. (May 2011). "MicroRNAs in the shoot apical meristem of soybean". Journal of Experimental Botany. 62 (8): 2495–2506. doi:10.1093/jxb/erq437. hdl:10536/DRO/DU:30106047. PMID 21504877.
- Qureshi A, Thakur N, Monga I, Thakur A, Kumar M (1 January 2014). "VIRmiRNA: a comprehensive resource for experimentally validated viral miRNAs and their targets". Database. 2014: bau103. doi:10.1093/database/bau103. PMC 4224276. PMID 25380780.
- Kumar M. "VIRmiRNA". Resource for experimental viral miRNA and their targets. Bioinformatics center, CSIR-IMTECH.
- Tuddenham L, Jung JS, Chane-Woon-Ming B, Dölken L, Pfeffer S (February 2012). "Small RNA deep sequencing identifies microRNAs and other small noncoding RNAs from human herpesvirus 6B". Journal of Virology. 86 (3): 1638–49. doi:10.1128/JVI.05911-11. PMC 3264354. PMID 22114334.
- Zheng H, Fu R, Wang JT, Liu Q, Chen H, Jiang SW (April 2013). "Advances in the techniques for the prediction of microRNA targets". International Journal of Molecular Sciences. 14 (4): 8179–87. doi:10.3390/ijms14048179. PMC 3645737. PMID 23591837.
- Agarwal V, Bell GW, Nam JW, Bartel DP (August 2015). "Predicting effective microRNA target sites in mammalian mRNAs". eLife. 4: e05005. doi:10.7554/eLife.05005. PMC 4532895. PMID 26267216.
Further reading
- miRNA definition and classification: Ambros V, Bartel B, Bartel DP, Burge CB, Carrington JC, Chen X, et al. (March 2003). "A uniform system for microRNA annotation". RNA. 9 (3): 277–9. doi:10.1261/rna.2183803. PMC 1370393. PMID 12592000.
- Science review of small RNA: Baulcombe D (September 2002). "DNA events. An RNA microcosm". Science. 297 (5589): 2002–3. doi:10.1126/science.1077906. PMID 12242426. S2CID 82531727.
- Discovery of lin-4, the first miRNA to be discovered: Lee RC, Feinbaum RL, Ambros V (December 1993). "The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14". Cell. 75 (5): 843–54. doi:10.1016/0092-8674(93)90529-Y. PMID 8252621.
External links
- The miRBase database
- miRTarBase, the experimentally validated microRNA-target interactions database.
- semirna, Web application to search for microRNAs in a plant genome.
- ONCO.IO: Integrative resource for microRNA and transcription factors analysis in cancer.
- MirOB Archived 4 March 2014 at the Wayback Machine: MicroRNA targets database and data analysis and dataviz tool.
- ChIPBase database: An open access database for decoding the transcription factors that were involved in or affected the transcription of microRNAs from ChIP-seq data.
- An animated video of the microRNA biogenesis process.
- miRNA modulation reagents to enable up-regulation or suppression of endogenous mature microRNA function
| miRNA precursor families | |
|---|---|
| 1-100 | |
| 101-200 | |
| 201-300 | |
| 301-400 | |
| 401-500 | |
| 501-600 | |
| 601+ | |
| Other | |
| Types of RNA | |
|---|---|
| Protein synthesis | |
| RNA processing | |
| Gene regulation | |
| Cis-regulatory elements | |
| Parasites | |
| Other | |
| Types of nucleic acids | |||||||
|---|---|---|---|---|---|---|---|
| Constituents | |||||||
| Ribonucleic acids (coding, non-coding) |
| ||||||
| Deoxyribonucleic acids | |||||||
| Analogues | |||||||
| Cloning vectors | |||||||