 | |
| Clinical data | |
|---|---|
| Trade names | Mavyret, Maviret (combination with glecaprevir) |
| Other names | ABT-530 |
| Pharmacokinetic data | |
| Protein binding | >99.9% |
| Elimination half-life | 13 hours |
| Excretion | 96.6% in faeces |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
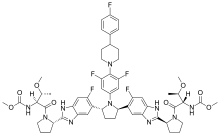
| Formula | C57H65F5N10O8 |
| Molar mass | 1113.201 g·mol |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Pibrentasvir is an NS5A inhibitor antiviral agent. In the United States and Europe, it is approved for use with glecaprevir as the combination drug glecaprevir/pibrentasvir (trade name Mavyret in the US and Maviret in the EU) for the treatment of hepatitis C. It is sold by Abbvie.
References
- Ng TI, Krishnan P, Pilot-Matias T, Kati W, Schnell G, Beyer J, et al. (May 2017). "In Vitro Antiviral Activity and Resistance Profile of the Next-Generation Hepatitis C Virus NS5A Inhibitor Pibrentasvir". Antimicrobial Agents and Chemotherapy. 61 (5): e02558–16. doi:10.1128/AAC.02558-16. PMC 5404558. PMID 28193664.
- Johnson LA (August 3, 2017). "FDA OKs new drug to treat all forms of hepatitis C". Fox Business.
- "Maviret: EPAR – Summary for the public" (PDF). European Medicines Agency. 2017-08-17. Archived from the original (PDF) on 2017-10-19. Retrieved 2017-10-19.
| RNA virus antivirals (primarily J05, also S01AD and D06BB) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Hepatitis C |
| ||||||||
| Hepatitis D | |||||||||
| Picornavirus | |||||||||
| Anti-influenza agents | |||||||||
| Multiple/general |
| ||||||||
| |||||||||
This antiinfective drug article is a stub. You can help Misplaced Pages by expanding it. |