 | |
| Clinical data | |
|---|---|
| Routes of administration | Topical (ointment, eye drops) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.217.969 |
| Chemical and physical data | |
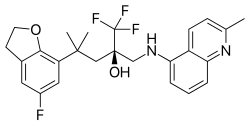
| Formula | C25H26F4N2O2 |
| Molar mass | 462.489 g·mol |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (what is this?) (verify) | |
Mapracorat (INN, code names BOL-303242-X, ZK-245186) is an anti-inflammatory drug belonging to the experimental class of selective glucocorticoid receptor agonists (SEGRAs). It is in clinical trials for the topical treatment of atopic dermatitis, inflammation following cataract surgery, and allergic conjunctivitis. Preliminary investigation for the treatment of keratoconjunctivitis sicca has been conducted in cellular models.
Clinical trials
Phase II clinical trials with mapracorat started in summer 2009. One trial was a double blind dose finding study for an ointment against atopic dermatitis. It tested concentrations of 0.01%, 0.03% and 0.1% versus placebo over four weeks in around 64 patients. This trial was conducted by Intendis, a part of Bayer HealthCare Pharmaceuticals specialized on dermatology, and completed in September or October 2010. The other trial, also with a double blind design, evaluated an ophthalmic suspension for the treatment of inflammation following cataract surgery. Various concentrations and dosing schemes were tested versus placebo in about 550 patients. The study was conducted by Bausch & Lomb and completed in September 2010. Its successor study, a phase III trial, started in November 2010 and completed in August 2011.
As of January 2017 no study results are available.
See also
References
- ^ Cavet ME, Harrington KL, Ward KW, Zhang JZ (September 2010). "Mapracorat, a novel selective glucocorticoid receptor agonist, inhibits hyperosmolar-induced cytokine release and MAPK pathways in human corneal epithelial cells". Molecular Vision. 16: 1791–800. PMC 2932489. PMID 20824100.
- ^ Clinical trial number NCT00944632 for "Dose Escalation of Different Concentrations of ZK 245186 in Atopic Dermatitis" at ClinicalTrials.gov
- ^ Clinical trial number NCT00905450 for "Evaluation of BOL-303242-X Versus Vehicle for the Treatment of Inflammation Following Cataract Surgery" at ClinicalTrials.gov
- Clinical trial number NCT01289431 for "Mapracorat Ophthalmic Formulation in Subjects With Allergic Conjunctivitis" at ClinicalTrials.gov
- Clinical trial number NCT01230125 for "Mapracorat Ophthalmic Suspension for the Treatment of Ocular Inflammation Following Cataract Surgery" at ClinicalTrials.gov
| Glucocorticoids and antiglucocorticoids (D07, H02) | |
|---|---|
| Glucocorticoids | |
| Antiglucocorticoids |
|
| Synthesis modifiers | |
| |
This drug article relating to the musculoskeletal system is a stub. You can help Misplaced Pages by expanding it. |
This antineoplastic or immunomodulatory drug article is a stub. You can help Misplaced Pages by expanding it. |
This dermatologic drug article is a stub. You can help Misplaced Pages by expanding it. |